What Occurs When Nacl S Is Added To Water

Ever been in the kitchen, maybe rustling around for that late-night snack, and you’ve grabbed a spoonful of something white, thinking it’s sugar, but BAM! Salty surprise? Yeah, we’ve all been there. Or perhaps you’re the diligent chef, carefully measuring out ingredients for that perfect stew, and you reach for the ol’ shaker. That white stuff, the one that makes our fries sing and our popcorn pop with flavor? That, my friends, is often NaCl, or as we mere mortals know it, table salt.
Now, let’s talk about its partner in crime, the universally present water. It’s in our coffee, our tea, our tears (sometimes from chopping onions, sometimes from watching a rom-com), and it’s basically the elixir of life. What happens when these two, NaCl and water, decide to have a little mingle? It’s a story as old as time, or at least as old as the first person who accidentally dropped their salt shaker near a puddle.
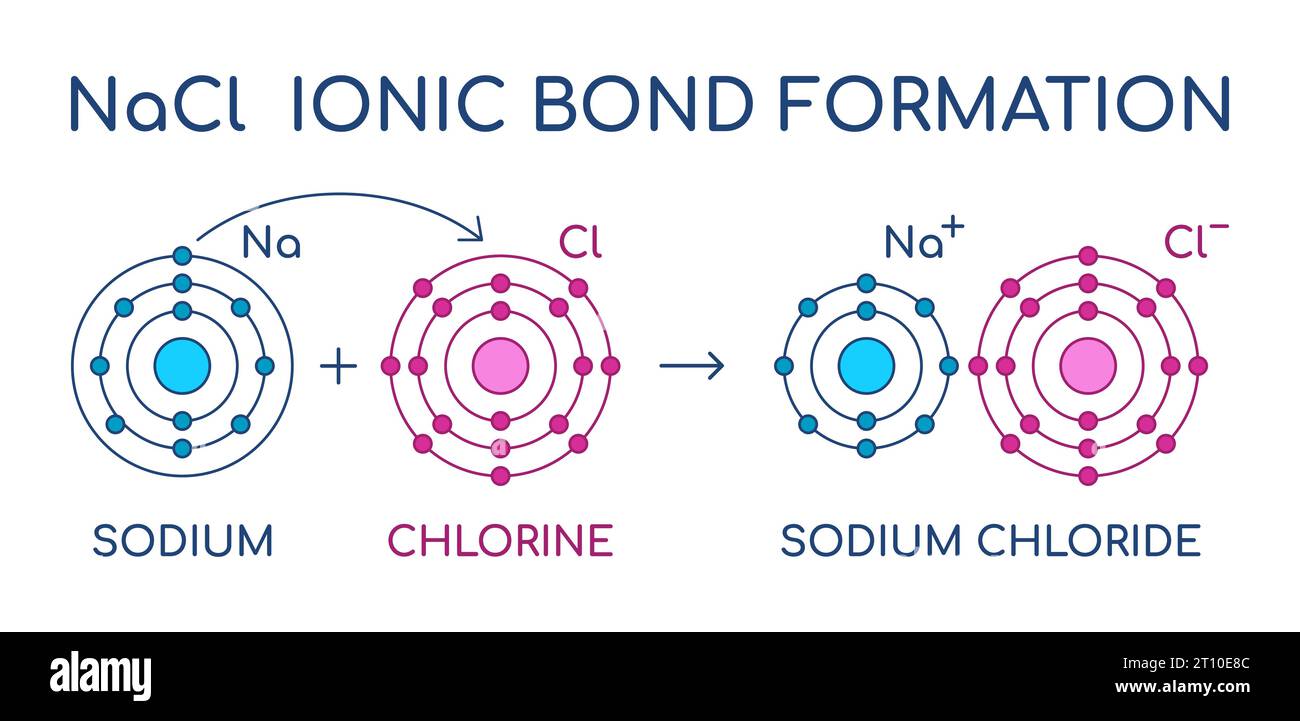
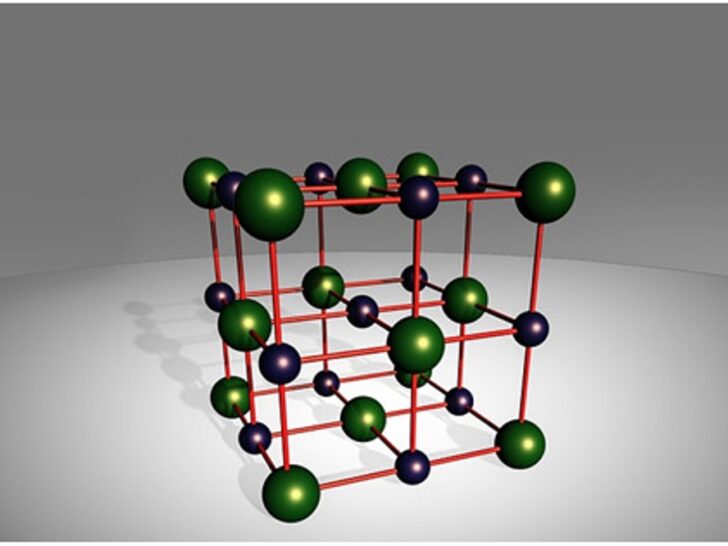
Imagine NaCl as a tiny, perfectly organized little ballroom dancer. It's got this neat, crystalline structure, like a miniature skyscraper built by tiny, very orderly aliens. Each NaCl unit is made up of two very different characters: a sodium ion (Na+) and a chloride ion (Cl-).
Must Read
Now, the sodium ion is a bit like a hyperactive kid who’s had too much sugar. It’s positively charged, meaning it’s got a little bit of an "I'm feeling energetic and a bit needy" vibe. On the flip side, the chloride ion is more like a cool, laid-back teenager. It’s negatively charged, carrying a bit of a "whatever, man" attitude. These two are pretty attracted to each other, like magnets that just can't get enough of each other. They stick together in that crystal lattice, forming the solid, grainy salt we know and love (or hate, depending on how much you’ve just eaten).
Then comes water. Ah, water. It’s a fascinating molecule, and its secret weapon in this whole NaCl dissolving saga is something called polarity. You see, a water molecule (H2O) isn’t perfectly symmetrical. It’s a bit like a Mickey Mouse head, with the oxygen atom being the nose and the two hydrogen atoms being the ears. The oxygen end of the molecule has a slightly negative charge, and the hydrogen ends have slightly positive charges. It’s not a full-blown electric shock, mind you, but it’s enough to make it a bit of a flirt.
When you toss that salty dancer into the watery dance floor, things start to get interesting. The water molecules, with their charmingly charged ends, are like the club’s social butterflies. They start swarming around the NaCl crystal.
The slightly positive hydrogen ends of the water molecules are drawn to the negatively charged chloride ions. They start to hug them, and pull at them, trying to pry them away from their buddies. It’s like a bunch of really friendly strangers saying, "Hey, come dance with us!"
At the same time, the slightly negative oxygen ends of the water molecules are doing the same thing to the positively charged sodium ions. They’re like, "You look lonely over there, let’s hang out!"
This energetic water party continues, with the water molecules essentially surrounding each sodium ion and chloride ion individually. It’s like each salt crystal gets completely mobbed by enthusiastic dancers. They break apart the happy (or maybe not-so-happy, depending on their relationship) ionic bond that held the NaCl together.
Once they’re all separated and individually surrounded by water molecules, the NaCl is considered dissolved. It’s no longer a solid chunk of salt; it’s now tiny, invisible ions zipping around in the water. This is why your soup looks clear even after you’ve added salt – the salt hasn't disappeared, it's just become so dispersed that you can't see it anymore. It's like your favorite celebrity showing up at a huge concert; they're there, but you can't pick them out of the crowd without a magnifying glass and a very dedicated fan club.
This whole process is called dissociation. The ions dissociate from each other. It's a fancy word for "they broke up and went their separate ways, but they're still in the same room."

And the resulting mixture? It’s called a solution. Specifically, a saltwater solution. It's the basis for so many things. Think about the ocean. It’s just a massive, ancient saltwater solution. All those tiny sodium and chloride ions, chilling in the water for eons.
Or consider when you’re feeling a bit under the weather and your doctor suggests saline solution for your eyes or nose. That’s basically a carefully balanced saltwater solution designed to be gentle on your body. It’s like the water molecules decided to be super hospitable and make everyone feel welcome.
What’s really neat is that this ability of water to dissolve things is a big deal in the natural world. It’s how plants get their nutrients from the soil. The minerals in the soil get dissolved by rainwater, and then the plant roots can slurp them up. Without water’s dissolving superpowers, life as we know it would be a lot more… dry.
Let's go back to that ballroom analogy. The NaCl crystal is the VIP section. The water molecules are the enthusiastic partygoers. When the water molecules arrive, they’re like, "Let’s get this party started!" The sodium and chloride ions, initially holding hands tightly, find themselves being pulled apart by the sheer force of all these energetic water molecules.
Each sodium ion, being positive, feels a strong attraction towards the negative oxygen end of a water molecule. Imagine a very popular kid (the sodium ion) being surrounded by a huge fan club (the water molecules), each wanting a piece of them. The water molecules essentially surround the sodium ion in a protective, watery embrace.
Similarly, the chloride ion, being negative, gets cozy with the positive hydrogen ends of the water molecules. It’s like the slightly more reserved but equally popular friend (the chloride ion) also finding themselves the center of attention. They too are completely enveloped by water molecules.
This phenomenon is called hydration. The ions are being hydrated by the water molecules. It’s like they’ve been given a nice, refreshing drink and are now ready to mingle freely in the liquid environment.
The crystal structure of NaCl is quite strong, like a meticulously built LEGO castle. But the polarity of water molecules provides the gentle, persistent force needed to dismantle it, piece by piece. It’s not a violent demolition; it’s more like a friendly invitation to a more spread-out, less structured gathering.
Think about dissolving sugar in water. It’s a similar principle, though sugar molecules are held together differently than salt ions. But the water molecules’ ability to interact with and surround other molecules is what makes it such a universal solvent. It’s the ultimate social lubricant of the molecular world.
So, the next time you’re adding a pinch of salt to your meal, or you’re sipping on some electrolyte drink, you can think about this tiny, molecular dance party happening. It's a constant, silent ballet of ions and molecules, all thanks to the amazing properties of water and the inherent charm of NaCl.
It's fascinating to consider how this simple interaction underpins so much of what we experience. From the taste of our food to the very functioning of our bodies, the ability of NaCl to dissolve in water is a fundamental chemical event that’s happening all around us, all the time. It’s the invisible magic that makes things taste better, and life itself possible.
The key takeaway here is the polarity of water. Without it, salt would just sit at the bottom of your glass, looking all smug and unmoving. But because water molecules are a bit like tiny, energetic little magnets, they can gently pull apart the ionic bonds that hold NaCl together. It’s a beautiful example of how even seemingly simple substances have complex and fascinating interactions.
And don't even get me started on what happens when you add too much salt. That's a whole other story, usually involving a desperate scramble for a glass of fresh water and a newfound appreciation for moderation. But for now, let's just appreciate the wonder of NaCl and water doing their thing, creating solutions that make our world a little tastier and a lot more functional.
It's a testament to the elegant simplicity of chemistry. No complicated machinery, no flashing lights, just molecules doing what they do best. It’s a reminder that even in the mundane act of seasoning our dinner, there’s a whole universe of molecular activity at play. Pretty cool, right?