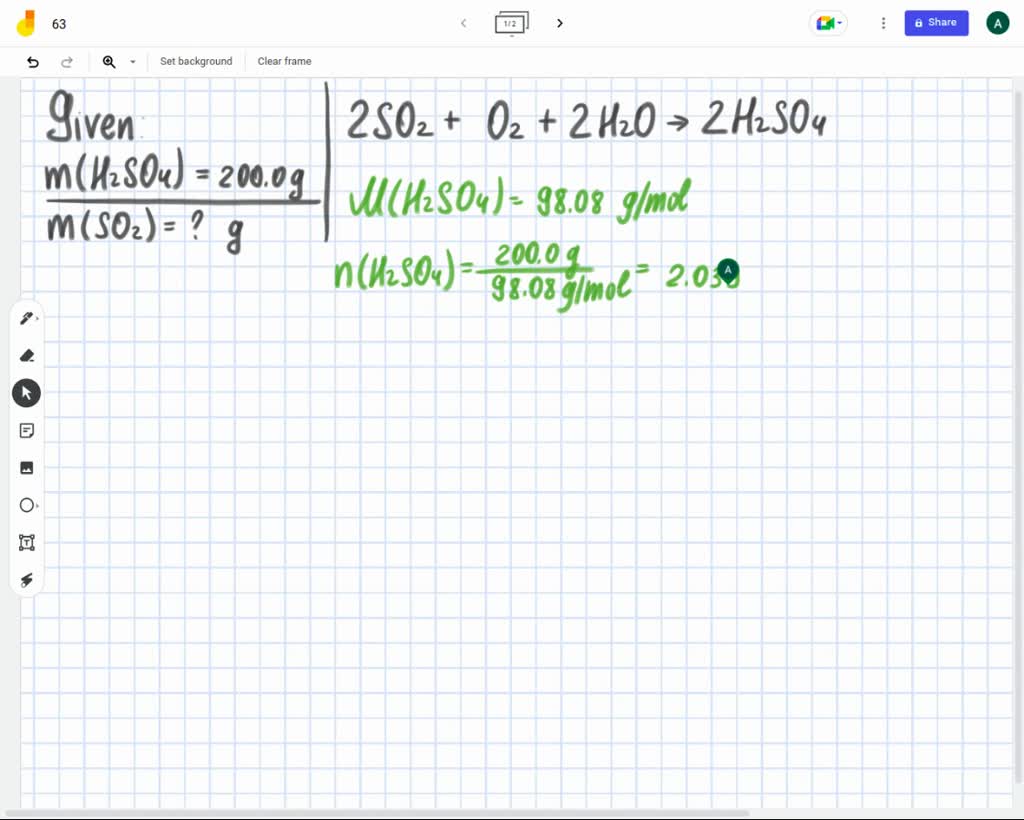
What Minimum Mass Of H2so4 Would You Need

Hey there, coffee buddy! So, you're wondering about sulfuric acid, huh? Fancy stuff. Or, well, maybe not fancy, but definitely... potent. Like, really potent. Ever spilled a tiny bit on your jeans and seen that immediate little fizz and then, poof, a hole? Yeah, that's H2SO4 for ya. It's basically the superhero of acids, or maybe the supervillain, depending on how you look at it. And today, we're diving into the nitty-gritty: what's the minimum mass of this stuff you'd even need?
It sounds like a question straight out of a chemistry textbook, doesn't it? But hey, we're here for a casual chat, so let's pretend we're not actually doing homework. We're just curious. Maybe you saw it in a movie, or you're just a naturally inquisitive soul. Either way, you're here, and I'm here, and we're going to figure this out. No need to break out your calculator just yet, though, we'll get there.
So, what is "minimum mass"? It's not like you can just grab a single molecule and say, "Yep, that's the minimum." Life, and chemistry, are a bit more… substantial than that. We're talking about needing a certain amount for something. But what is that something? That’s the real question, isn't it? It’s like asking, "What’s the minimum amount of coffee I need to function?" It depends on the day, right? And the person!
Must Read
Let's break it down. Sulfuric acid, H2SO4. Got it. The H is for hydrogen, the S is for sulfur, and the O is for oxygen. Four atoms in total per molecule. Simple enough, right? But when you start talking about mass, things get a little more interesting. We can't just count molecules, can we? That would take forever. Imagine trying to count all the coffee beans in a giant sack. No thanks.
Instead, chemists use this handy-dandy thing called the mole. It's like a chemist's dozen, but way bigger. One mole is approximately 6.022 x 10^23 particles. Yeah, that's a 6 followed by 23 zeros. It's an astronomically huge number. So huge, in fact, it's almost impossible to visualize. Think about it: if you had that many grains of sand, you could cover the entire Earth in a layer several miles thick. Wild, right?
And this mole concept is directly linked to the atomic mass of elements. You know those numbers on the periodic table? Like, hydrogen is about 1.008, sulfur is about 32.06, and oxygen is about 15.999. Those are essentially the mass of one mole of those elements in grams. So, for our H2SO4 molecule, we can just add up the masses of all the atoms in it. Easy peasy, lemon squeezy… almost.
Two hydrogens (2 x 1.008) plus one sulfur (32.06) plus four oxygens (4 x 15.999). Let's do some quick (mental) math here. 2.016 + 32.06 + 63.996. Drumroll, please… it comes out to roughly 98.07 grams per mole. So, one mole of sulfuric acid weighs about 98.07 grams. That's our molar mass. It's the weight of that humongous number of molecules.

But the question isn't about the mass of one mole, is it? It's about the minimum mass. And that, my friend, is where the fun (and the potential for disaster) begins. What are we trying to do with this sulfuric acid? Are we trying to dissolve a small pebble? Neutralize a tiny amount of a base? Or maybe just make a really, really, really potent cleaning solution for that one stubborn stain you've been ignoring? The "minimum mass" is entirely context-dependent. It’s like asking, "What's the minimum amount of water I need to drink?" Well, are you running a marathon or just watching TV?
Let's consider some scenarios. If you were doing a simple lab experiment, say, titrating a known amount of a base, you might need a specific amount of sulfuric acid. But that's a calculated amount, not necessarily a "minimum" in the abstract sense. It's the minimum required for that particular reaction. It's not like there's a universal "minimum sulfuric acid you should ever have on hand" rule. Although, if there was, I suspect it would be a ridiculously small number, probably less than a drop.
Think about it this way: what's the smallest amount of something that can still be considered something? For sulfuric acid, we're dealing with molecules. So, technically, one molecule of H2SO4 is the minimum amount of sulfuric acid. But can you even measure one molecule? Not with our everyday tools, that's for sure. Even in a super-advanced lab, isolating and measuring a single molecule is… well, it's pushing the limits of what's practical.
So, when we talk about "mass," we're generally talking about something we can weigh. And for practical purposes, you can't really weigh a single molecule. It's too light, too fleeting. We need a macroscopic amount. Something you can see, something you can hold (carefully, very carefully, with gloves and goggles!).
Let's say you wanted to perform a reaction that requires a certain concentration of sulfuric acid. Concentration is basically how much of your stuff is dissolved in how much of your solvent. Like, if you make lemonade, the concentration of lemon juice determines how tart it is. With sulfuric acid, you often have it in solution, usually with water. A common concentration is 98% sulfuric acid, meaning 98% of the mass is H2SO4 and the other 2% is mostly water.

If you needed, let's say, to react with 1 gram of sodium hydroxide (NaOH), which is a common base. To neutralize that, you'd need a specific amount of sulfuric acid. Using our molar masses (NaOH is about 40 g/mol, H2SO4 is about 98 g/mol), and the balanced chemical equation (H2SO4 + 2NaOH -> Na2SO4 + 2H2O), we can figure out the exact stoichiometry. It tells us that one mole of sulfuric acid reacts with two moles of sodium hydroxide.
So, to react with 1 gram of NaOH, which is roughly 1/40th of a mole, you'd need half of that, or 1/80th of a mole of H2SO4. And 1/80th of 98 grams is… about 1.225 grams. See? Even for a relatively small amount of another substance, you still need a measurable amount of sulfuric acid. 1.225 grams is not a tiny speck you can't see. That's something you can definitely weigh on a lab scale. It’s about the weight of a small paperclip. Still not huge, but definitely there.
But is 1.225 grams the minimum? Only in the context of that specific reaction. What if you just wanted to, I don't know, make a tiny bubble of gas? Or slightly etch a piece of metal? The minimum mass would change. It’s all about the purpose. It’s the classic "what’s the minimum amount of fuel for a rocket?" question. It depends on how far you want to go, doesn't it?
Let's think about the absolute smallest practically measurable amount. On a very sensitive analytical balance, you can weigh down to fractions of a milligram (0.001 grams). So, theoretically, you could measure out, say, 0.001 grams of sulfuric acid if you had a sufficiently pure sample and a very, very accurate scale. That's a speck. Like, a really, really tiny speck. You probably wouldn't even see it.

But would that speck do anything? Probably not much. It might react with a few stray molecules on the surface of something, causing a minuscule, imperceptible change. It's the difference between having a single grain of salt on your tongue and tasting the ocean. The grain is there, but it doesn't define the experience. The ocean… that's something else entirely.
So, if we're being super literal and asking for the minimum measurable mass of H2SO4, it’s probably going to be in the milligram range, assuming you have the right equipment. It's the smallest amount you can reliably quantify. But that’s more of a measurement question than a chemical reaction question.
If we're talking about the minimum mass needed to cause a noticeable effect, that's a whole different ballgame. What’s "noticeable"? A visible reaction? A change in pH? A burning sensation? All of these require different amounts. A tiny drop of concentrated sulfuric acid on your skin? Ouch! That’s definitely noticeable, and that's a very small mass. We’re talking fractions of a gram, maybe even less, depending on how concentrated it is and how long it’s in contact.
Let’s consider the pure, undiluted stuff. It’s dense, about 1.84 grams per milliliter. So, a single drop? A standard drop is about 0.05 milliliters. That would be roughly 0.092 grams of sulfuric acid. That’s still… well, it’s about a tenth of a gram. That’s enough to cause a pretty nasty burn. So, for causing harm, the minimum mass is surprisingly small!
But for a chemical reaction? To actually participate in a reaction in a meaningful way? You generally need a certain number of moles. And to get a certain number of moles, you need a certain mass. The smallest amount of H2SO4 that's commonly used in a lab setting for a simple reaction might be in the hundreds of milligrams, or maybe a few grams. It depends on the scale of the experiment, of course.

Think about a very dilute solution, like 0.1 M (molar). If you wanted 1 milliliter of that, you'd have 0.0001 moles of H2SO4. And 0.0001 moles * 98 g/mol = 0.0098 grams, or 9.8 milligrams. That’s a tiny amount! That’s about the weight of a few grains of sugar. So, if you’re diluting it down, the actual mass of H2SO4 needed gets really, really small. That's where the "minimum mass" really starts to shrink.
The key takeaway here is that there's no single, universal "minimum mass" of sulfuric acid. It's like asking "what's the minimum amount of water to swim?" Well, are you trying to do the butterfly stroke or just float? It depends on the activity, the scale, and the desired outcome. If you want to cause a dramatic chemical change, you'll need more. If you just want to observe a minuscule interaction, or if you're working with highly dilute solutions, the mass can be incredibly small.
So, next time someone asks you about the minimum mass of sulfuric acid you'd need, you can confidently say, "It depends on what you're doing with it!" And then maybe, just maybe, you can offer to explain the mole concept over another cup of coffee. Just remember to keep that H2SO4 far away from both the coffee and the listener!
It's fascinating, isn't it? How something so powerful can be measured in such minuscule amounts, or in such vast quantities. It’s all about context. The universe of chemistry, like the universe itself, is full of extremes. And sulfuric acid? It definitely occupies its own special, potent corner.
So, there you have it. No single number. Just a whole lot of "it depends." Kind of like life, really. Now, about that coffee refill…