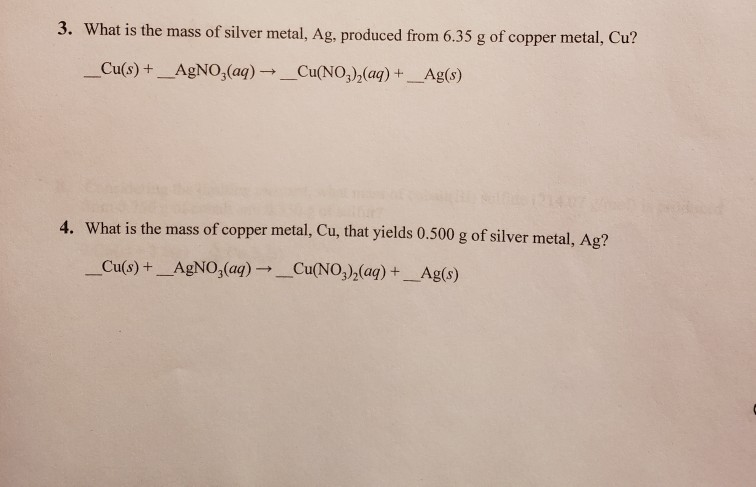What Mass Of Silver Is Produced From 100g Cu

Hey there, science-curious friend! So, you’ve been wondering, huh? Like, if you’ve got a hefty 100 grams of copper – a pretty common metal, right? – what kind of sparkly silver can you whip up from it? It sounds like a magic trick, a bit like turning lead into gold, but way more achievable and, let’s be honest, a lot shinier. We’re talking about a bit of chemistry here, but don’t worry, we’re going to keep it super chill and easy to digest. Think of it like following a recipe, but instead of cookies, we’re baking with elements!
Now, before we dive into the nitty-gritty, let’s get one thing straight: this isn't about literally transmuting copper into silver. That’s still in the realm of sci-fi or very, very expensive particle accelerators. What we're talking about is a chemical reaction where copper and silver are involved, usually in a solution, and one might displace the other. It’s a bit like a dance party, where one element (copper) decides to step aside and let another (silver) take its place in a chemical compound.
The most common way this happens is through something called a single displacement reaction. Imagine you have a bunch of copper atoms chilling in a solution, and then you introduce some silver ions. The copper, bless its heart, is a bit of a social butterfly. It’s more reactive than silver. This means it’s got more energy and is more eager to bond with other things. So, when it sees those silver ions hanging around, it thinks, "Ooh, new friends! I'd rather be bonded to that than whatever I'm doing now!"
Must Read
So, the copper atoms decide to ditch their current companions (whatever they were in the solution) and jump into a new partnership with the anions (the negatively charged bits) that the silver ions were originally with. In doing so, the copper atoms become ions themselves, and the silver ions, now free from their anion buddies, get a chance to buddy up with each other. And voilà! You get solid, shiny silver atoms precipitating out of the solution, like tiny silver glitter bombs going off!
The most common scenario where this plays out involves a copper wire or strip dipped into a solution containing silver nitrate. Silver nitrate sounds fancy, but it's basically a compound where silver ions (Ag+) and nitrate ions (NO3-) are happily partnered. When the copper (Cu) enters the picture, it’s like, "Hey, nitrate, you look like you could use a new dance partner!" The copper then kicks the silver out of the partnership, forming copper nitrate (Cu(NO3)2) and leaving the silver ions to clump together and form solid silver (Ag).
The chemical equation for this little drama looks something like this: Cu(s) + 2AgNO3(aq) → Cu(NO3)2(aq) + 2Ag(s). Don’t let the letters and numbers scare you! It’s just a shorthand way of saying that solid copper (Cu) reacts with silver nitrate dissolved in water (aq) to produce copper nitrate dissolved in water and solid silver. The little "(s)" means solid, and "(aq)" means dissolved in water. See? Not so scary!
Now, to figure out how much silver you’d get from 100 grams of copper, we need to bring in the big guns: stoichiometry. Don’t panic! Stoichiometry is just a fancy word for using the balanced chemical equation to figure out the relative amounts of reactants and products. It’s all about ratios, like making sure you have the right amount of flour for your cookies. Too much or too little, and things can get… well, a bit wonky.

The key to stoichiometry lies in the molar masses of the substances involved. Think of a mole as a baker’s dozen, but for atoms and molecules. It’s a specific number of particles (about 6.022 x 10^23 to be exact – that’s a lot of zeroes!). The molar mass tells you how many grams one mole of a substance weighs. It's usually found right on the periodic table. For our dance party, we need the molar masses of copper (Cu) and silver (Ag).
A quick peek at our trusty periodic table (or a quick Google search, no judgment here!) tells us that the atomic mass of copper (Cu) is approximately 63.55 grams per mole. That means 63.55 grams of copper is one mole of copper atoms. For silver (Ag), the atomic mass is around 107.87 grams per mole. So, 107.87 grams of silver is one mole of silver atoms.
Now, let’s look back at our balanced chemical equation: Cu(s) + 2AgNO3(aq) → Cu(NO3)2(aq) + 2Ag(s). This equation is like a recipe telling us that 1 mole of copper reacts to produce 2 moles of silver. The coefficients in front of the chemical formulas are our magic numbers!
So, if we start with 100 grams of copper, the first step is to figure out how many moles of copper that is. To do that, we divide the mass of copper by its molar mass:
Moles of Cu = Mass of Cu / Molar Mass of Cu

Moles of Cu = 100 g / 63.55 g/mol
Let’s do that math… drumroll please… approximately 1.57 moles of copper.
Great! Now we know we have about 1.57 moles of copper ready to party. According to our balanced equation, for every 1 mole of copper, we get 2 moles of silver. So, if we have 1.57 moles of copper, we’ll get twice that amount of silver!
Moles of Ag produced = Moles of Cu × (2 moles Ag / 1 mole Cu)

Moles of Ag produced = 1.57 moles Cu × 2
This gives us approximately 3.14 moles of silver. Woohoo! We’re getting closer to that sparkly silver goodness!
The final step is to convert these moles of silver back into grams, because we’re interested in the mass, the tangible, sparkly stuff. To do this, we multiply the moles of silver by its molar mass:
Mass of Ag produced = Moles of Ag produced × Molar Mass of Ag
Mass of Ag produced = 3.14 moles × 107.87 g/mol

And the grand total… with a little calculation… we get approximately 338.7 grams of silver!
Isn’t that neat? So, from a good old 100-gram chunk of copper, you can potentially end up with over 338 grams of pure, shimmering silver! It’s like the copper is saying, "Here, have some bling! I’m going to be happy as copper nitrate."
Of course, in the real world, things aren't always 100% perfect. There might be some side reactions, some copper that doesn't fully react, or some silver that doesn’t quite form solid crystals. These are called yield issues. Sometimes you get a theoretical yield (what the math says you should get), and then you have an actual yield (what you actually get in your experiment). Think of it like baking: sometimes your cookies turn out perfectly, and sometimes they’re a little… enthusiastic in spreading out. But the math gives us the ideal scenario, the ultimate potential!
This whole process is a fantastic example of how chemistry works – how different elements interact, how we can predict the outcomes of reactions, and how we can even use these reactions to our advantage. It’s not just about boring textbooks and equations; it’s about understanding the world around us and the amazing transformations that can happen, sometimes with just a little bit of water and a bit of ingenuity.
So, the next time you see a piece of copper, you can imagine it holding the potential for a significant amount of dazzling silver. It's a reminder that even common materials can hold hidden treasures and that the universe is constantly engaging in these fascinating chemical dances. It’s a little bit of science magic, just waiting to be understood. And hopefully, this little chat has made you feel a bit more enlightened and, dare I say, a little bit inspired by the wonders of chemistry! Keep that curiosity alive, and who knows what amazing discoveries you'll make!