What Is The Purpose Of Catalytic Converter
Alright, gather 'round, grab a cuppa, and let's talk about something that’s probably living under your car right now, looking all mysterious and important. I'm talking about the humble, yet utterly crucial, catalytic converter. Ever wondered what this chunky metal box of tricks actually does? Is it secretly plotting world domination? Does it have a tiny barista inside brewing espresso for your engine? (Spoiler alert: it doesn't. Though, wouldn't that be neat?).
Let's be honest, most of us just know it's "that thing" the mechanic mentions when it's acting up and your wallet starts to weep. You might have even heard it described as a "money pit," which, while sometimes accurate when it's on its last legs, totally misses the point of its real purpose. Think of it as the unsung hero of your exhaust system, the bouncer at the club of your car's emissions, politely but firmly telling the bad guys to get lost.
The "Bad Guys" Your Catalytic Converter Kicks Out
So, what kind of hooligans are we talking about? When your engine does its thing – combusting fuel, making that satisfying rumble (or not-so-satisfying rattle) – it creates some pretty nasty byproducts. We're talking about a cocktail of pollutants that, if left unchecked, would make the air we breathe about as pleasant as licking a bus exhaust pipe. These are your primary offenders:
Must Read
First up, we have carbon monoxide (CO). This stuff is bad news bears. It's colorless, odorless, and can knock you out cold faster than a poorly timed dad joke. Imagine your engine exhaling pure, unadulterated suffocation. Not ideal, right?
Then there are hydrocarbons (HC). This is basically unburnt fuel. Think of it as your car spitting out expensive gasoline vapor into the atmosphere. Talk about a waste of money and air quality! It’s like going to a fancy restaurant and just… not eating your food. What a crime!

And finally, the notorious nitrogen oxides (NOx). These guys are the architects of smog and acid rain. They’re the grumpy old neighbors of air pollution, always complaining and making everything worse. They contribute to respiratory problems and generally make the sky look like a faded denim jacket.
Enter Our Hero: The Catalytic Converter!
Now, imagine all these nasty characters trying to escape your car's tailpipe, ready to wreak havoc on our beautiful planet. But wait! There's a guardian at the gate! Our trusty catalytic converter steps in, like a traffic cop with a very stern expression and a whole lot of scientific know-how.
Its main gig, its raison d'être, its raison de caca (if you will, for the less sophisticated gasses), is to perform a little chemical magic. It’s not actually burning anything, despite the name. It’s more about changing things. It’s a chemical alchemist, transforming these toxic troublemakers into much more agreeable substances.

The Chemical Cocktail Party
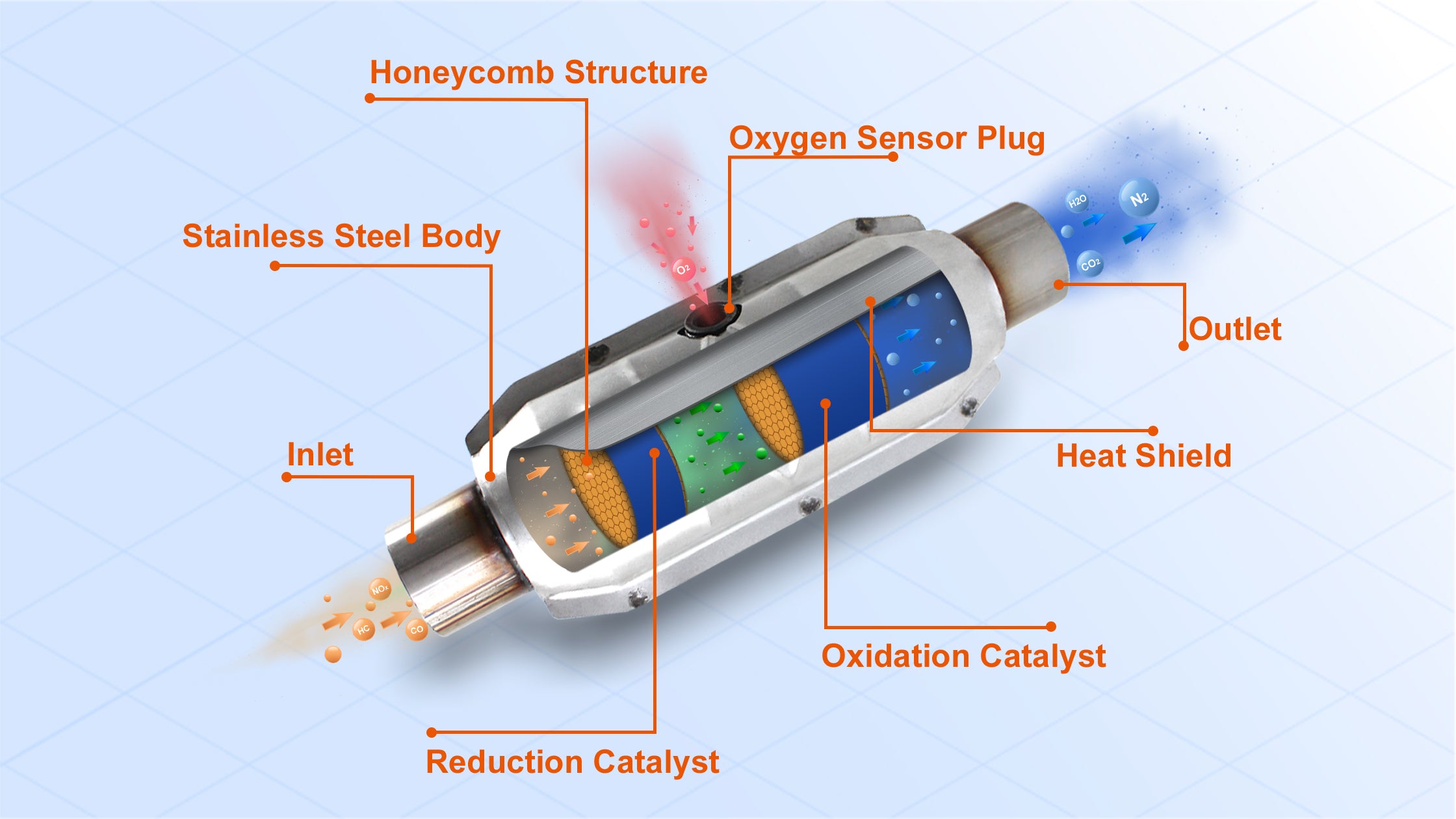
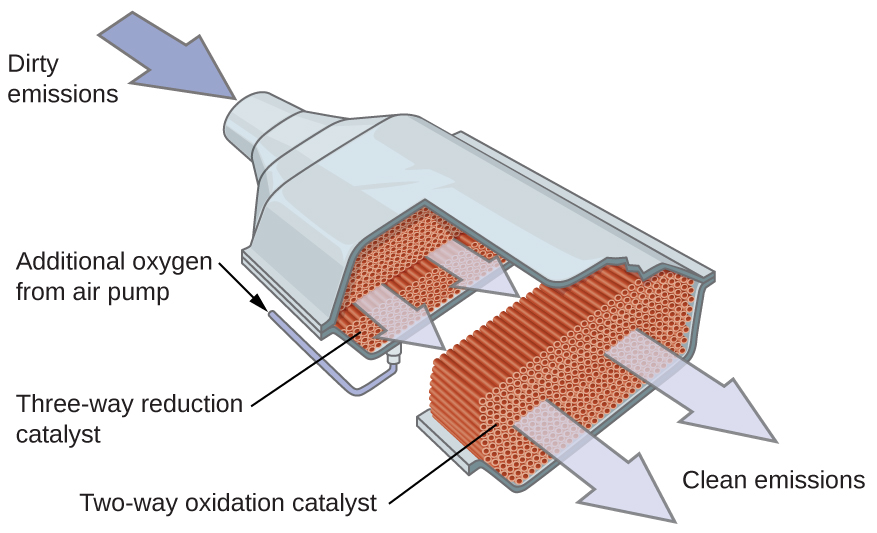
Inside that unassuming metal box, there's a honeycomb-like structure, usually made of ceramic. This isn't for storing honey, sadly. This honeycomb is coated with precious metals – think platinum, palladium, and rhodium. These aren't just for bling; they're the hardworking catalysts for this whole operation. They provide a massive surface area for the exhaust gases to interact with.
So, when the hot, grumpy exhaust gases rush through these channels, they come into contact with these precious metals. It's like a speed-dating event for molecules, but instead of awkward silences, there are chemical reactions happening at lightning speed. These reactions are what we call catalysis, hence the name!
Let's break down the transformations, shall we? It's less "Transformers" and more "Transistors" of toxicity:

- Carbon Monoxide (CO) meets Oxygen (O₂): Thanks to our metallic matchmakers, the CO gets together with O₂ and they happily form… carbon dioxide (CO₂). Yep, the same CO₂ that gets a bad rap for climate change. But here’s the kicker: CO₂ is way less toxic than CO. It’s like going from a mugger to just a slightly annoying acquaintance. Plus, your car already produces tons of CO₂ naturally from burning fuel. So, the converter is just making a bad situation a little less deadly. Think of it as upgrading from poisonous gas to… well, still a greenhouse gas, but a less immediately poisonous one. It's all about context!
- Hydrocarbons (HC) meet Oxygen (O₂): These greedy unburnt fuel molecules also get a makeover. With the help of platinum and palladium, they combine with O₂ to become… carbon dioxide (CO₂) and water (H₂O). Boom! Your car is now producing water vapor and CO₂ instead of spitting out raw gasoline. It’s like turning a leaky faucet into a pleasant mist. And honestly, who doesn't love a bit of mist on a hot day?
- Nitrogen Oxides (NOx) get… reformed! This is where things get a bit more complex, and rhodium plays a starring role. NOx molecules are broken down, and their nitrogen and oxygen atoms are separated. The nitrogen atoms then bond with other nitrogen atoms to form nitrogen gas (N₂), which is the primary component of the air we breathe! The oxygen atoms are essentially recycled back into the mix to help with the oxidation of CO and HC. So, NOx, the smog-maker, is turned into 78% of the air you're currently inhaling. Pretty neat trick, huh?
Why All the Fuss?
So, why do we even care about this chemical sleight of hand? Well, remember those "bad guys" we talked about? If they were allowed to run wild, the air quality in our cities would be abysmal. We’d be living in a perpetual smog haze, and respiratory illnesses would skyrocket. It’s estimated that without catalytic converters, the U.S. would have released an extra 100 million tons of pollutants into the atmosphere annually since their widespread adoption in the 1970s. That’s enough to make a squirrel faint from a mile away!
Think of it as the unsung bodyguard for your lungs and the lungs of everyone around you. It's the reason you can roll down your car window in traffic without instantly tasting metallic despair. It's the silent guardian, the watchful protector, the… well, you get the idea.
A Little More Than Meets the Eye
Here’s a fun fact: these little marvels are not just for cars. They're also used in trucks, buses, motorcycles, and even some generators. Anywhere there’s an internal combustion engine burning fossil fuels, there’s a good chance a catalytic converter is doing its noble duty.

Another surprising fact? They can get incredibly hot. We're talking temperatures that could fry an egg in seconds. So, while they’re busy cleaning up your car's act, they're also basically acting as tiny, extremely efficient car furnaces. Just don't go touching them after a long drive, unless you fancy a free, painful skin graft.
And if your check engine light comes on and the mechanic mentions a "clogged" or "failed" catalytic converter, it usually means one of a few things. It could be old age, like your grandpa after a marathon. It could be contaminated by bad fuel or oil, which is like feeding your athlete junk food before a big race. Or, and this is the sad part, it could have been stolen. Yep, the precious metals inside are valuable, making them targets for thieves. So, the very thing that protects our air can also become a victim of crime. The irony is almost as thick as the smog it prevents.
So, the next time you hear that familiar rumble of an engine, spare a thought for the unsung hero under your car. It’s a small piece of engineering with a massive impact, working tirelessly to make our air a little cleaner, one chemical transformation at a time. It’s not just a part; it’s a pact with the planet. And that, my friends, is a purpose worth talking about.