What Is The Ph Of A Neutral Solution At 25c
Hey there, science curious friends! Ever found yourself staring at a bottle of water and suddenly wondering... what’s its pH? And not just any pH, but the special pH of a neutral solution at a cozy 25°C? Sounds a bit like a secret handshake in the world of chemistry, right? Well, buckle up, because we're about to unlock this little mystery, and I promise, it's more fun than it sounds!
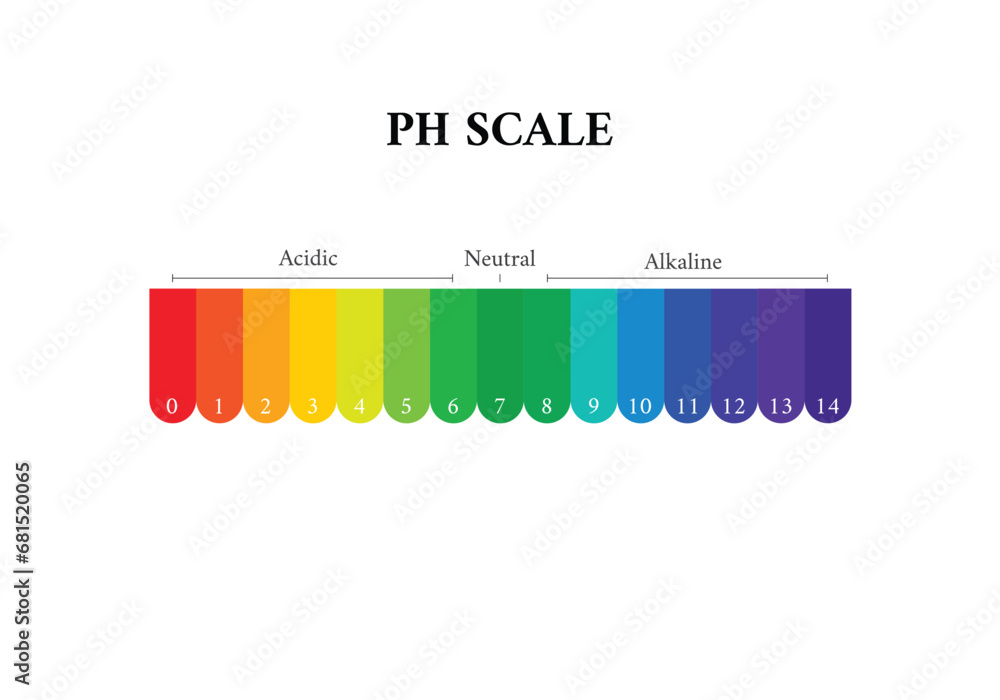
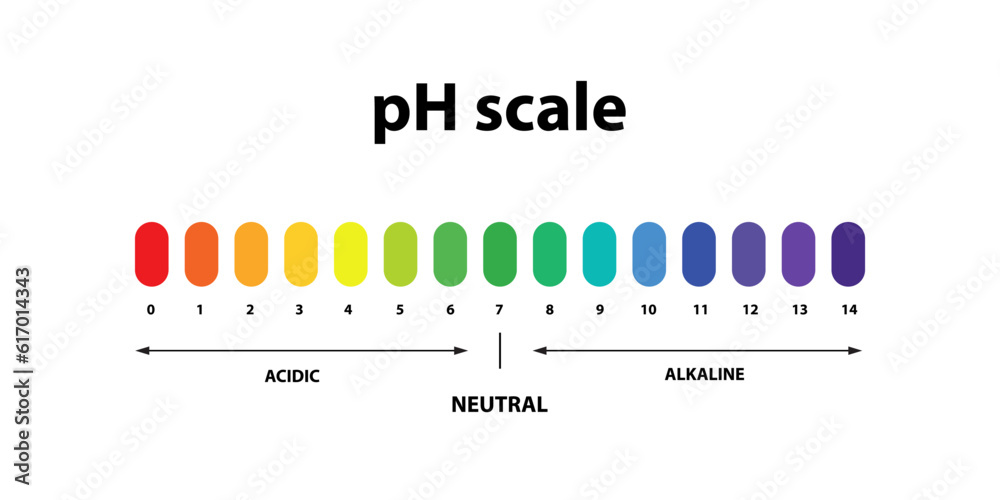
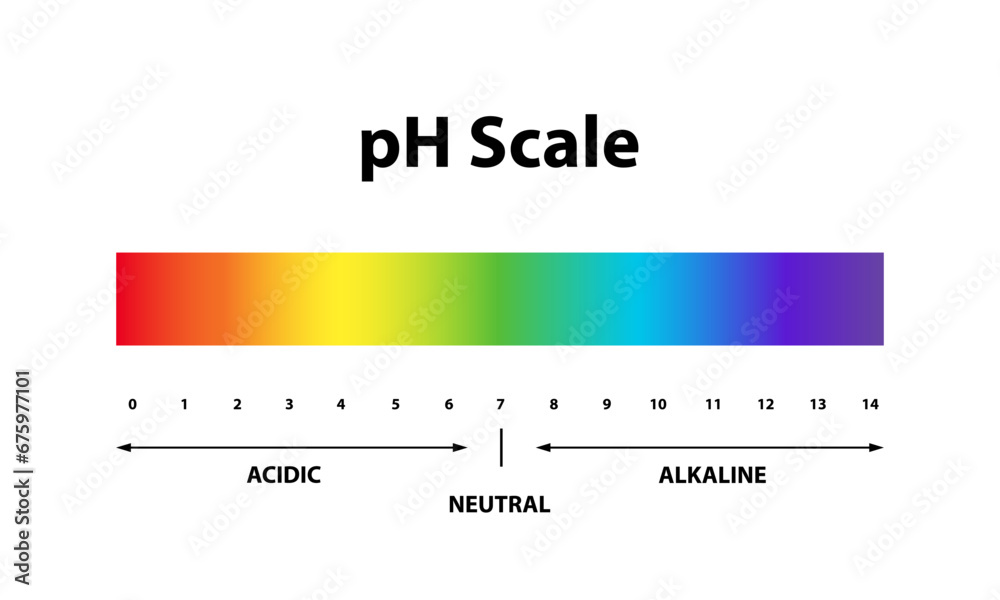
So, let's dive right in. What exactly is pH? Think of it as a scale, a way for scientists (and now, for you!) to measure how acidic or basic something is. It’s like a sourness meter or a slipperiness gauge, all rolled into one. The scale runs from 0 to 14. Anything below 7 is considered acidic – think lemon juice or vinegar. Anything above 7 is basic (or alkaline, if you want to sound fancy) – like baking soda or, well, some of your favorite cleaning products.
But here’s the magic number, the VIP of the pH world: 7. This is our golden ticket to neutrality. A pH of 7 means a solution is neither acidic nor basic. It's perfectly balanced, like a harmonious choir hitting all the right notes. And guess what? When we’re talking about a neutral solution chilling out at 25 degrees Celsius (that’s a comfortable room temperature, by the way – not too hot, not too cold, just right!), its pH is precisely, wonderfully, gloriously 7.
Must Read
Why 25°C, you ask? Good question! Temperature has a tiny little bit of influence on how water molecules behave. At 25°C, water is doing its standard, happy thing. As the temperature changes, the balance between acidic and basic components in water shifts ever so slightly, meaning the neutral pH can drift away from 7. But for our everyday adventures in understanding the world around us, 25°C is our benchmark, our perfect pH party starter.
So, what does this pH of 7 actually mean in the grand scheme of things? It means that in a neutral solution, the concentration of hydrogen ions (H+, which make things acidic) and hydroxide ions (OH-, which make things basic) are exactly equal. It’s a state of perfect equilibrium. Imagine a seesaw perfectly balanced with equal weights on both sides. That’s our neutral solution!

Now, you might be thinking, "Okay, pH 7 is neutral. Why should I care?" Oh, my friend, this is where the fun really begins! Understanding pH is like gaining a superpower to interpret the world in a whole new way. Think about it:
Your Kitchen Science Playground
Why does that tomato taste a little tangy, while that banana is so sweet? pH! Why does adding baking soda make your cakes rise? pH! Why does milk go sour? You guessed it – a change in pH!
Even cooking becomes a science experiment where pH plays a starring role. The tenderness of meat, the color of your vegetables, the way your bread bakes – all influenced by the subtle or not-so-subtle pH of your ingredients and cooking environment.

And when you're making that perfect cup of coffee or brewing your favorite tea, the pH of the water can actually affect the flavor! Who knew a simple glass of water could hold so many secrets? A slightly acidic water might bring out different notes than a slightly alkaline one. Isn't that just fascinating?
Your Body's Inner Balance Act
Your own body is a marvel of pH regulation. Your blood, for instance, needs to stay within a very narrow pH range (around 7.35 to 7.45) to keep you healthy and energized. If it drifts too far in either direction, it’s a big problem. Your amazing body works tirelessly to maintain this delicate balance, and knowing about pH helps us appreciate just how incredible our internal systems are.
Even your skin has a natural "acid mantle," a protective layer with a slightly acidic pH that helps keep out nasty bacteria. That’s why using the wrong skincare products, which might be too alkaline, can disrupt this natural defense. So, the next time you’re choosing a face wash, you’ll have a little extra scientific ammunition!
Gardening Glory
Love to garden? pH is your secret weapon for happy plants! Different plants thrive in different soil pH levels. Some love it a little acidic, while others prefer it more alkaline. Understanding the pH of your soil helps you choose the right plants for your patch of earth and even amend the soil to make it perfect for your green companions. Imagine nurturing your garden to its fullest potential, all thanks to a little bit of pH knowledge!
Everyday Wonders
Think about swimming pools. Why do they add chemicals? To keep the water’s pH just right for comfort and to prevent it from becoming a breeding ground for bacteria. Or consider aquariums – maintaining the correct pH is crucial for the health of your aquatic friends. It’s everywhere!
The world around us is constantly engaged in a pH dance, and now you’re invited to the ballroom! It’s not just about memorizing numbers; it’s about appreciating the intricate chemical processes that make life as we know it possible. It’s about seeing the science in the mundane and finding wonder in the everyday.
So, the next time you encounter something that’s neutral, or acidic, or basic, don’t just pass it by. Give it a nod of recognition. You understand its fundamental nature! You’ve peered behind the curtain and seen a glimpse of the elegant chemistry that shapes our world.
And this is just the beginning! The world of chemistry is vast and full of amazing discoveries waiting to be made. From the smallest atoms to the grandest galaxies, there’s always something new and exciting to learn. So, don't stop here! Let this little peek into the pH of a neutral solution spark your curiosity. Keep asking questions, keep exploring, and you’ll find that learning about science can be not only incredibly rewarding but also genuinely fun. Go forth and be scientifically curious – the universe is waiting for you to discover its secrets!