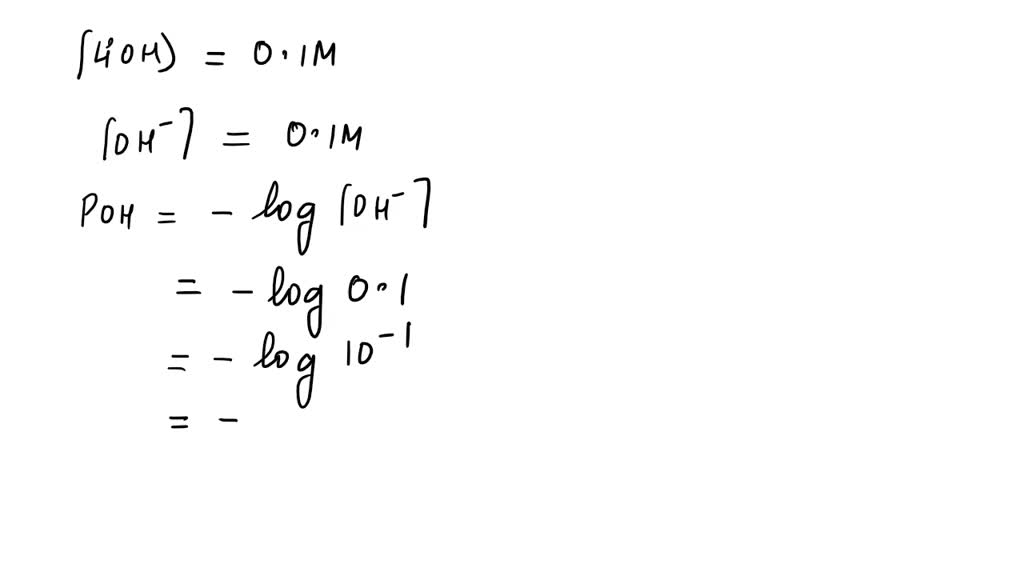What Is The Ph Of A 0.050 M Lioh Solution
Let's talk about something that might sound a bit… science-y. We're diving into the mysterious world of pH. Don't worry, we're keeping it super chill and fun. Think of it like a secret handshake for chemists, but way less awkward.
Today's star is a solution called LiOH. It's basically lithium hydroxide. Fancy name, right? It’s a compound that’s pretty good at what it does.
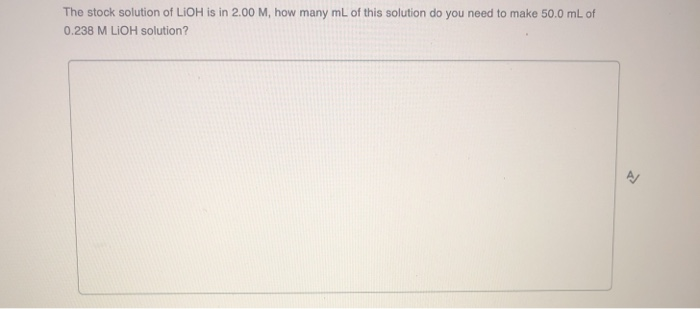
We've got a specific amount of this LiOH. It's 0.050 M. Now, "M" is just a shorthand for molarity. It tells us how much stuff is packed into a certain amount of liquid. It's like saying "a handful of jellybeans in this jar."
Must Read
The Big pH Question
So, the question on everyone’s (or at least, our imagined everyone’s) mind is: What’s the pH of this 0.050 M LiOH solution?
This is where things get a little exciting. pH is all about how acidic or basic something is. Think of it as a scale. On one end, you have things that make your tongue pucker, like lemons (acids). On the other end, you have things that feel slippery, like soap (bases).
Pure water is right in the middle. It’s neutral. It doesn't really pick a side. It’s the Switzerland of solutions, you could say.
LiOH: A Base of Operations
Now, LiOH? It's definitely not water. It's a base. A strong one, actually. This means it's ready to make things more alkaline. It’s the opposite of those sour lemons.
When LiOH dissolves in water, it does something special. It breaks apart. It releases ions. Two important ions are lithium ions (Li+) and hydroxide ions (OH-).
It's those hydroxide ions (OH-) that are the real MVPs when it comes to bases. They're the reason why a solution becomes basic. They’re the party starters for alkalinity.
It's like the LiOH is a small, powerful machine that churns out these OH- ions. More OH- means a higher pH. Simple as that, right?
Since LiOH is a strong base, it almost completely breaks apart. This is good news for us because it means we know pretty much exactly how many hydroxide ions are floating around.
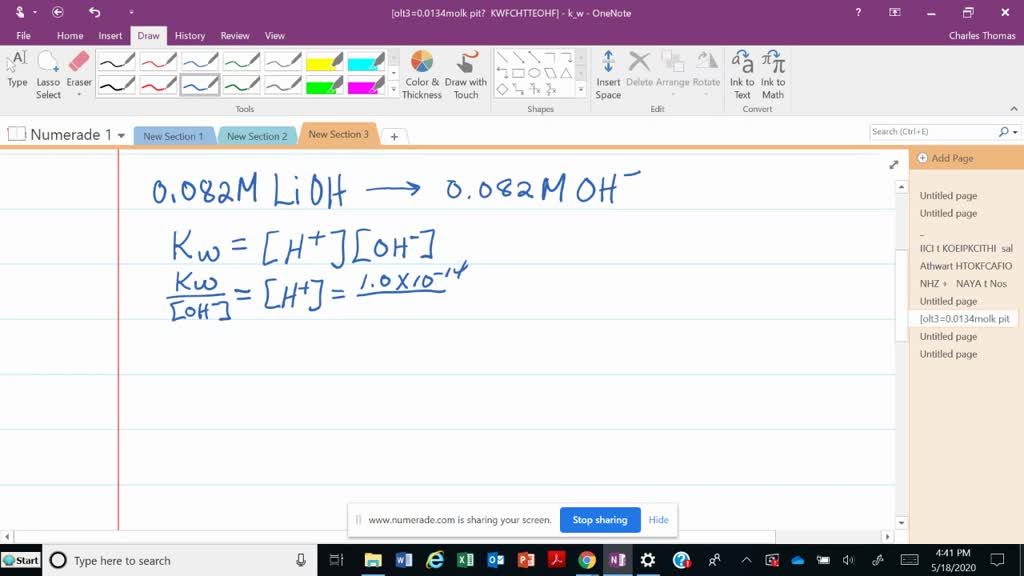
Our concentration of LiOH is 0.050 M. Because it's a strong base and releases one OH- for every LiOH molecule, the concentration of hydroxide ions is also 0.050 M.
So, we have a [OH-] of 0.050 M. This is the key number we need.
The pH Puzzle Pieces
Now, how do we get from [OH-] to pH? There's a little helper called pOH. It's basically the pH but for bases.
The relationship is super friendly: pH + pOH = 14. It's a constant, like the speed of light, but for our lab experiments.
To find pOH, we take the negative logarithm of the hydroxide ion concentration. It sounds fancy, but it’s just a mathematical tool. Think of it as a special calculator function.

So, pOH = -log[OH-].
Plugging in our number: pOH = -log(0.050).
If you punch that into a calculator (or, you know, just remember it from that one chemistry class you aced), you'll get a pOH of approximately 1.30.
Almost There!
We're so close, you can practically taste the alkaline goodness. We have our pOH, and we know that pH + pOH = 14.
So, to find the pH, we just rearrange that equation: pH = 14 - pOH.
Let's do the math:
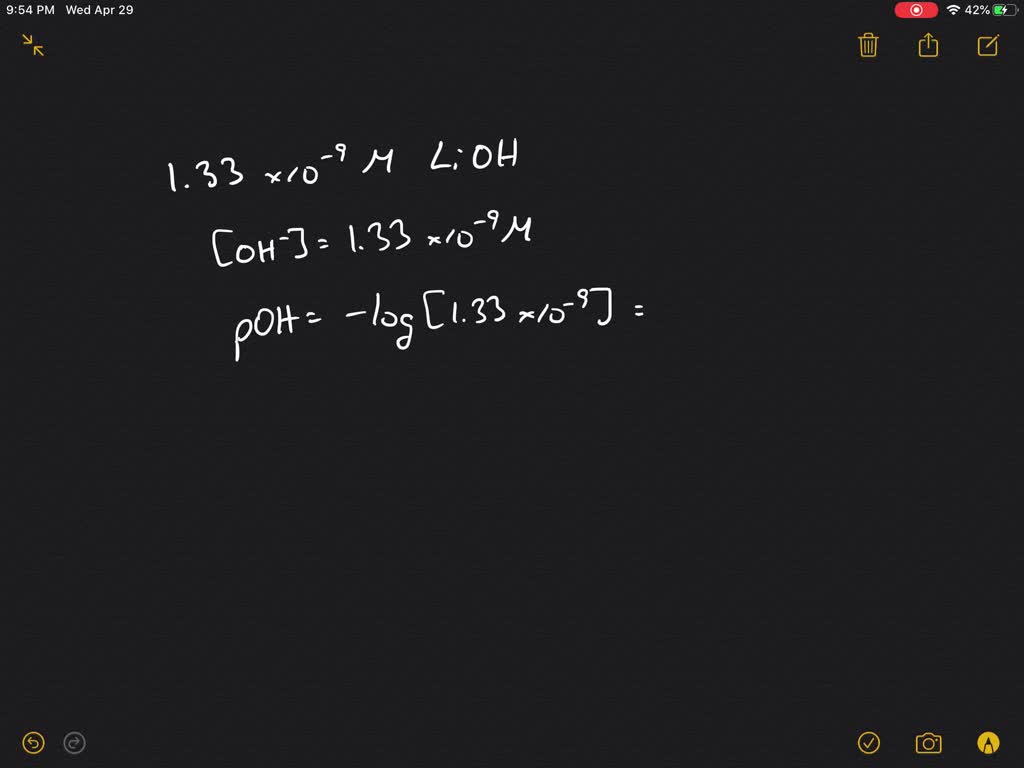
pH = 14 - 1.30.
And the grand reveal? The pH of our 0.050 M LiOH solution is approximately 12.70!
Yep, it’s a big, fat, happy number on the basic side of the scale. Definitely not something you’d want to drink with your breakfast cereal.
This pH value tells us our solution is very alkaline. It's a powerful base.
It’s a reminder that even simple-sounding solutions can have a significant impact. This little bit of LiOH packs a punch in terms of its pH.
Unpopular Opinion Time!
Okay, here’s my totally unpopular opinion: pH is actually pretty cool. I know, I know. Many people groan at the mention of acids and bases. They think it's all complicated formulas and lab coats.
But honestly, once you get past the jargon, it’s like a puzzle. You're figuring out the personality of a liquid. Is it grumpy (acidic)? Is it chill (neutral)? Or is it overly enthusiastic and slippery (basic)?
![[ANSWERED] What is the theoretical pH of a 0 0001 M LiOH solution 012](https://media.kunduz.com/media/sug-question-candidate/20220513044522218237-4440783.jpg?h=512)
And understanding pH helps us understand the world around us. From the food we eat to the cleaning products we use, pH is everywhere.
Think about how you choose your shampoo. Some are designed for oily hair (which might use ingredients that adjust pH). Others are for color-treated hair (where maintaining a specific pH is crucial).
Or consider gardening! Different plants thrive in different soil pH levels. It's like giving them the perfect dining environment.
So, this 0.050 M LiOH solution with its pH of 12.70? It’s just a friendly reminder of how diverse the chemical world is. It’s a bit like meeting a really, really energetic puppy. You know it’s strong, and you know it’s going to make a splash.
It’s a number, sure. But it’s a number that tells a story. A story about how much hydroxide is ready to mingle. A story about alkalinity.
And in my book, that's way more interesting than it sounds. So next time you hear about pH, don't run for the hills. Give it a little nod. It’s just the liquid world’s way of saying hello.
It’s a scale of personality, a spectrum of power. And this LiOH solution? It’s definitely leaning towards the “very, very alkaline” side of the personality spectrum. No apology needed!