What Is The Oxidation Number Of Cl In Cl2

Ever looked at a bottle of bleach and wondered about the tiny atoms having their own little drama inside? Well, today we're peeking behind the scenes of Chlorine, specifically when it's all by its lonesome, just like in that trusty bottle of Cl2. Think of it as a tiny, invisible party where everyone is a chlorine atom, and they're all holding hands.
Now, when elements decide to team up, they often have different roles, like in a play. One might be the hero, the other the sidekick, or maybe they're just equal partners. But what happens when everyone at the party is the exact same element? It's like a reunion of best friends who've known each other forever.
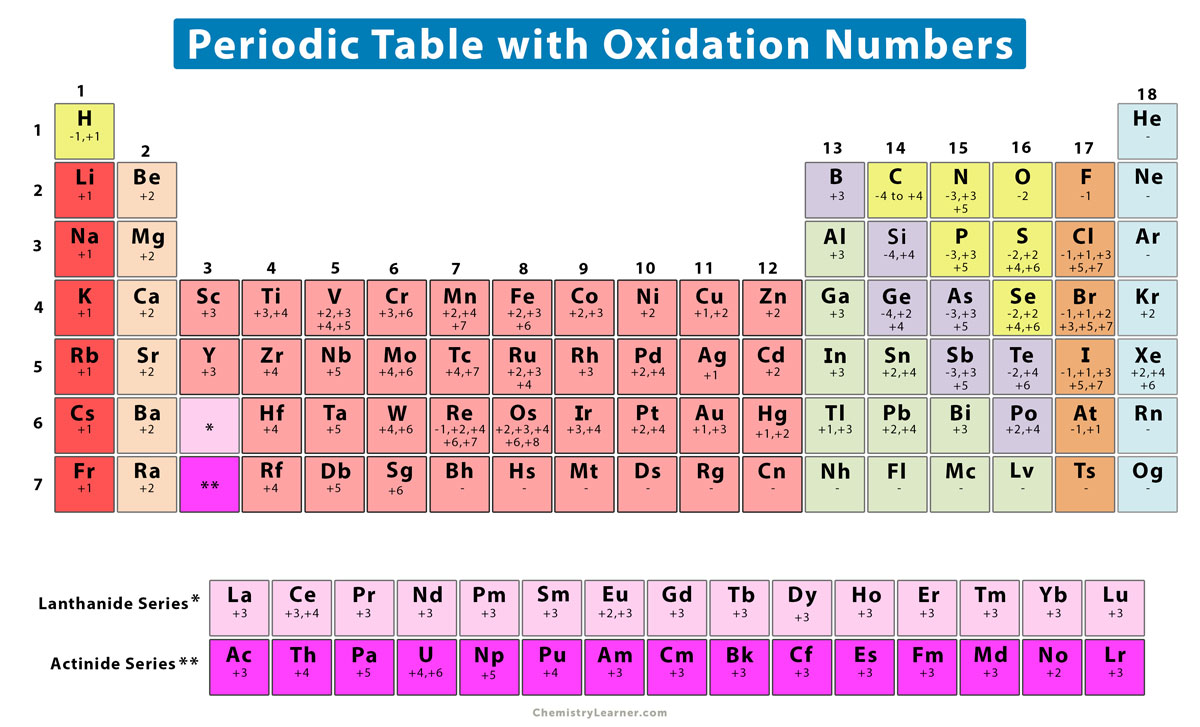
In the world of chemistry, these roles are sometimes described using something called an "oxidation number". It's a way to keep track of how electrons, those zippy little things, are shared or traded between atoms. Imagine it as an atom's "score" in a game of giving and taking.
Must Read
But here's where it gets interesting, and frankly, a little bit heartwarming. When two identical atoms, like two chlorine atoms in Cl2, decide to buddy up, they're essentially equals. They're not playing a game of "I'll take that electron!" versus "No, I want it!".
Instead, they share. They're like two kids on a seesaw, perfectly balanced, each holding on equally. Or maybe like two best friends sharing the last cookie – no one grabs it all for themselves.
So, when we talk about the oxidation number of Cl in Cl2, it's kind of like asking, "What's the score of a game where everyone ties perfectly?" The answer is beautifully, wonderfully simple.
Because they are sharing electrons equally, neither chlorine atom is "winning" or "losing" any electrons in that bond. They're just… together. And in the grand accounting system of oxidation numbers, this perfect equality translates to a very specific number.

This number isn't flashy or complex. It doesn't involve dramatic electron transfers or daring steals. It's a quiet, understated score that reflects a state of perfect equilibrium.
The oxidation number of Cl in Cl2 is zero. Yep, just plain old zero. It's like getting a perfect attendance award for not having to share or take anything unfairly.
Think about it this way: if you and your best friend are walking hand-in-hand, neither of you is pulling the other in a specific direction, right? You're just moving together, a unified pair. That's the Cl2 molecule.
The electrons in the bond between the two chlorine atoms are shared so evenly, so perfectly, that it's as if they're not being given or taken at all. They're just… there, in the middle, keeping the two atoms cozy.

This might seem a little anticlimactic after all that talk of electron drama. But in chemistry, sometimes the simplest answers are the most elegant. And there's a certain charm in the idea that even in the microscopic world, perfect togetherness can result in a score of nothing.
It’s a little reminder that not everything is about winning or losing. Sometimes, it’s about finding a partner, sharing equally, and existing in a state of peaceful, balanced union. And that’s exactly what our two chlorine friends are doing in Cl2.
They’re not striving for greatness by stealing electrons from someone else. They’re not being bullied into giving theirs away. They’ve found their perfect match, and they’re content to be a pair.
This applies to any time an element exists on its own, in its elemental form. If you find pure Oxygen (O2), its oxidation number is also zero. If you find pure Iron (Fe), it's zero. It’s a badge of honor for self-sufficiency and inner peace.
It's like each of these atoms has achieved a state of nirvana, where they are neither giving nor taking, but simply being. They are perfectly content in their own company, or with their identical twin.
So, the next time you see a bottle of bleach or a lab labeled with Cl2, you can smile knowing that within that seemingly ordinary substance, there’s a beautiful story of perfect partnership. Two chlorine atoms, side-by-side, sharing equally, and proudly sporting an oxidation number of zero.
It's a testament to the fact that sometimes, the most profound states of being are achieved through simple, equal sharing. No drama, no conflict, just pure, unadulterated molecular harmony.
And isn't that something to appreciate? The quiet strength of an element at peace with itself. The joy of finding a perfect, balanced bond.
It’s a little lesson from the atomic world, delivered with a wink and a smile. The oxidation number of Cl in Cl2 is zero because, in this case, Cl is just being Cl, with a friend who’s exactly the same. And that’s a beautiful thing indeed.
So, while other atoms might be busy with their complex electron exchanges and dramatic chemical reactions, remember the humble Cl2 molecule. It’s a symbol of perfect equality, a testament to the power of sharing, and a reminder that sometimes, zero is the most perfect number of all.
It's the atomic equivalent of a perfect high-five, or two people singing their favorite song in perfect harmony. They’re not trying to impress anyone; they’re just enjoying the moment, together.
And that, my friends, is the sweet, simple story of the oxidation number of Cl in Cl2. It’s less about complicated rules and more about the heartwarming reality of atoms finding their perfect, equal footing.