What Is The Empirical Formula For C6h12o6

Ever stared at a sugar cube, a juicy apple, or even a slice of bread and wondered, "What's really in this stuff?" We all know it's sweet, it's energy, it's... well, it's food. But sometimes, science likes to throw around fancy terms that sound like they belong in a Mad Scientist's lab rather than your kitchen. Today, we're going to tackle one of those terms: the empirical formula, specifically for that super common molecule, C6H12O6. Don't let the letters and numbers scare you; it's actually way less complicated than trying to assemble IKEA furniture on a Sunday afternoon.
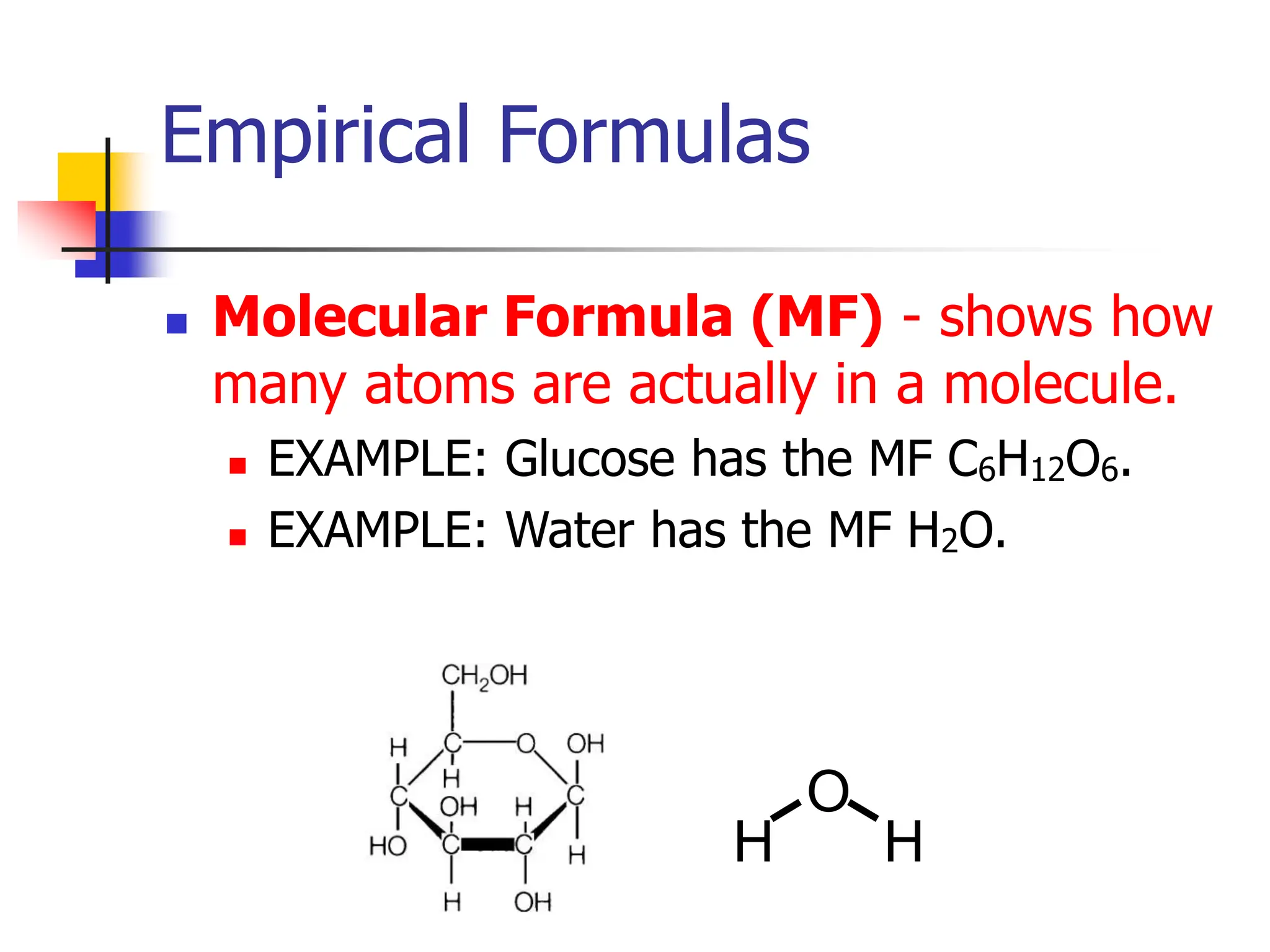
So, what's this C6H12O6 thing all about? Think of it as the molecular formula. It tells you the exact number of each type of atom in a single molecule. So, for this particular molecule, you've got 6 carbon atoms (that's the 'C'), 12 hydrogen atoms (the 'H'), and 6 oxygen atoms (the 'O'). It's like the full ingredient list for a specific recipe, down to the last grain of salt. You see this formula pop up for all sorts of yummy things, like glucose (that's your blood sugar, the stuff that gives you that immediate energy boost when you're about to faint from hunger), fructose (the sweet stuff in fruits, making that peach taste so darn good), and galactose (which you find in milk). They're all chemically identical at the molecular level, like identical triplets who somehow manage to look and act a little bit different depending on the situation.
Now, the empirical formula is like the simplified version of that ingredient list. Imagine you're making a big batch of cookies. Your molecular formula might be something like 10 cups of flour, 5 cups of sugar, 3 eggs, and 2 cups of chocolate chips. But if someone asks for the ratio of ingredients, you might say, "Well, for every 10 cups of flour, I use 5 cups of sugar." You're simplifying it, right? You're looking for the smallest whole-number ratio. The empirical formula does the same thing for molecules. It gives you the simplest ratio of atoms in a compound.
Must Read
Let's get back to our star molecule: C6H12O6. We've got carbons, hydrogens, and oxygens. Now, we need to find the smallest whole number that can divide all of these numbers evenly. Think of it like trying to split a pizza with your friends. If you have 6 slices, 12 slices, and 6 slices of different toppings, you want to divide them up so everyone gets the same proportional amount of each topping, and you want to use the fewest number of slices possible for each person's share. It's like saying, "Okay, for every 1 slice of topping A, I've got 2 slices of topping B and 1 slice of topping C."
In our case, the numbers are 6, 12, and 6. What's the biggest number that can go into all of those without leaving a remainder? Let's try a few. Can we divide by 2? Yes! 6 divided by 2 is 3, 12 divided by 2 is 6, and 6 divided by 2 is 3. So we get C3H6O3. That's a simpler ratio! But is it the simplest? Let's look at 3, 6, and 3. Can we divide all of these by another number? You guessed it! We can divide them all by 3.

So, 6 divided by 6 is 1. 12 divided by 6 is 2. And 6 divided by 6 is 1. This leaves us with a ratio of C1H2O1. We usually don't write the '1's in formulas, so it becomes CH2O. And that, my friends, is the empirical formula for C6H12O6! It's the simplest possible ratio of carbon to hydrogen to oxygen atoms.
Think about it like this: If you buy a big bag of M&Ms, the molecular formula is like the exact count of every single M&M in the bag. The empirical formula is like saying, "For every red M&M, there are two blue M&Ms and one green M&M." It tells you the proportion, the fundamental building blocks of the mix, without getting bogged down in the exact total number. It's the essence of the recipe, stripped down to its most basic, elegant form.

Why do scientists even bother with this "simplest ratio" thing? Well, sometimes they don't know the exact molecular formula right away. They might figure out the percentage of each element in a compound. For example, they might say, "This mysterious new substance is 40% carbon, 6.7% hydrogen, and 53.3% oxygen by mass." Sounds like a riddle, right? But with a little bit of chemical magic (and some handy calculations), they can use those percentages to find the empirical formula. It's like being a detective and figuring out the basic suspects and their roles before you know exactly how many of each were involved in the caper.
Once you have the empirical formula, you can then try to figure out the molecular formula if you know the compound's molar mass (which is like knowing the total weight of one "batch" of your recipe). You just see how many times the empirical formula's weight fits into the total weight. It's like saying, "My simplified recipe uses 1 cup of flour, 2 cups of sugar, and 1 cup of butter. If I know my entire batch of cookies needs to weigh 5 pounds, and each 'simplified recipe unit' weighs 1 pound, then I know I need to make 5 of those simplified recipes."
So, for C6H12O6, which is found in everything from that sugary rush you get from candy to the complex carbohydrates in your pasta, its empirical formula is CH2O. This simple ratio is actually the foundation for a whole class of compounds called carbohydrates. That's why they're called "carbo-hydrates" – it's basically carbon (carbo) with water (hydrate) attached in a specific way, and CH2O really captures that essence. It's like the "parent" structure, the fundamental blueprint.

Think of it like comparing a single, perfectly formed diamond (the molecule) to a pile of building blocks that can be assembled into that diamond in a specific ratio (the empirical formula). Or, think of the difference between a detailed blueprint of a skyscraper and a simple sketch of the basic shape. Both are useful, but they serve different purposes. The molecular formula tells you the precise structure, the exact "build." The empirical formula tells you the fundamental ratio, the essential "proportion."
It's kind of funny when you think about it. That sweet, delicious substance that powers your workouts or helps you through a tough afternoon? Its most basic chemical identity, its simplest proportional makeup, is just one carbon, two hydrogens, and one oxygen. It's like realizing that your favorite complex song can be broken down into a few simple, repeating notes. The magic is in how those simple notes are put together, just like the magic of sugars is in how those CH2O units are arranged in more complex ways to form C6H12O6 and other carbohydrates.

So, the next time you're enjoying a piece of fruit, a cookie, or even just sipping on some water (which has its own formula, H2O, and guess what? Its empirical formula is also H2O because it's already in its simplest form, like a perfectly balanced meal!), you can impress your friends (or just yourself) with the knowledge that you understand the difference between a molecular formula and an empirical formula. It’s like knowing the difference between the full ingredients list on a fancy gourmet dish and the simple, essential flavors that make it taste so good.
Remember, C6H12O6 is the exact recipe, the specific arrangement of atoms. But CH2O is the core ratio, the fundamental building block. It’s the difference between saying, "I have exactly 6 apples, 12 bananas, and 6 oranges," and saying, "For every apple, I have two bananas and one orange." Both are true, but the second one gives you a sense of the proportion in the simplest possible terms. It’s a bit like looking at a detailed painting and then understanding the basic color palette the artist used. The detail is amazing, but the foundation is also important.
And that, my friends, is the simple – and not-so-scary – truth about the empirical formula for C6H12O6. It's just a simplified ratio, a way to see the fundamental building blocks of something we encounter every single day. So, go forth and spread your newfound chemical wisdom! Just try not to overcomplicate things, and always remember the power of simplification, whether it's in chemistry or in figuring out what to make for dinner.