What Is The Average Atomic Mass Of Rubidium
Hey there, science curious folks! Ever wondered about the hidden, teeny-tiny building blocks that make up everything around us? Well, get ready to have your mind blown by a element that's almost as cool as a rockstar and twice as sparkly: Rubidium! Seriously, this stuff is amazing, and today we're going to tackle one of its most fascinating quirks – its average atomic mass.
Now, before you start picturing a giant scale in a super-secret lab, let's break this down. Think of atoms like tiny LEGO bricks that make up our universe. Each element has its own unique type of LEGO brick, and Rubidium is one of those special ones. It’s got its own distinct personality, and that personality includes how much it weighs, on an atomic scale, of course!
So, what exactly is this “average atomic mass” thing? Imagine you have a bag full of different kinds of apples. Some are big and juicy, some are smaller and tarter, and some are just… well, average. If you wanted to know the "average weight" of an apple in your bag, you'd weigh a bunch of them and then divide the total weight by the number of apples. That's kind of what scientists do with atoms!
Must Read
But here’s the super-duper, extra-special twist with elements like Rubidium. It’s not like all the Rubidium atoms are exactly the same size and weight, like perfectly manufactured identical twins. Nope, nature loves to keep things interesting! For Rubidium, there are a couple of different "versions" of its atom hanging around, and these versions have slightly different weights.
These different weight versions are called isotopes. Think of them like siblings in the same family. They're all fundamentally Rubidium, but they have a slightly different number of tiny particles inside their core. It's like having a brother who's a little taller and a sister who’s a little shorter, but they're still undeniably part of the same family.
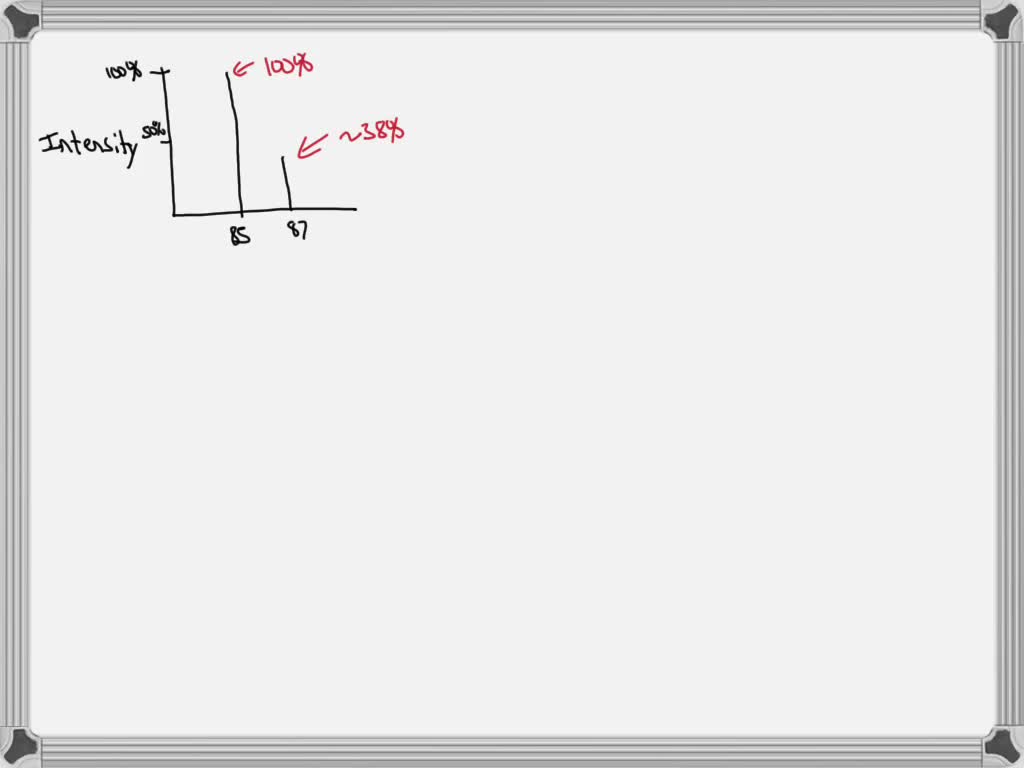
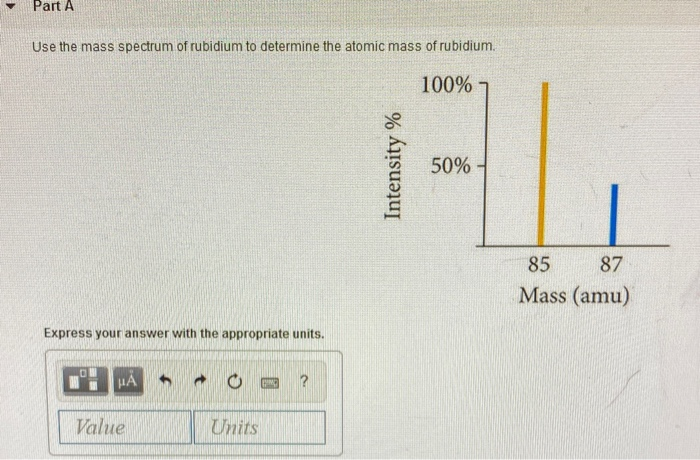
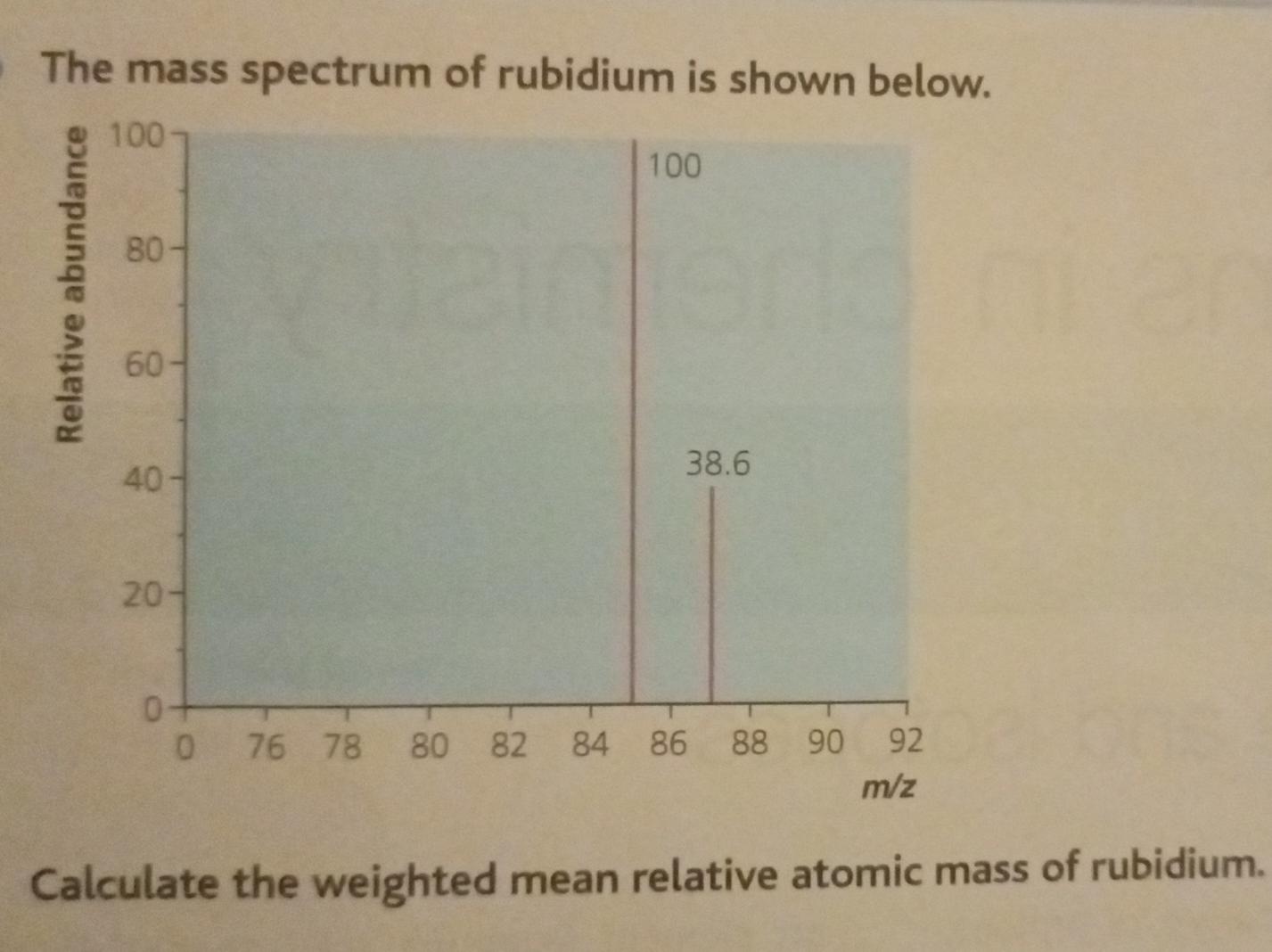
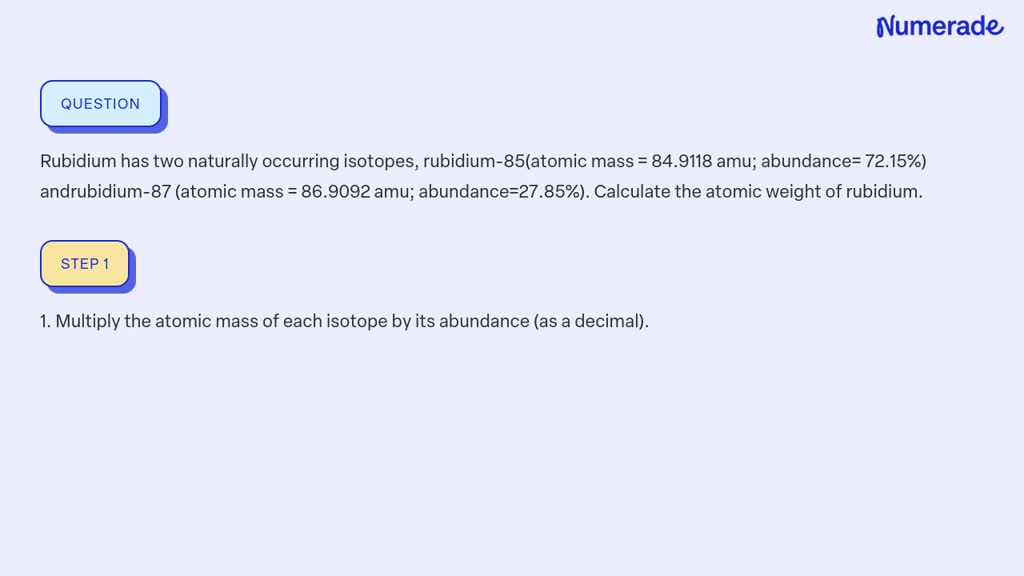
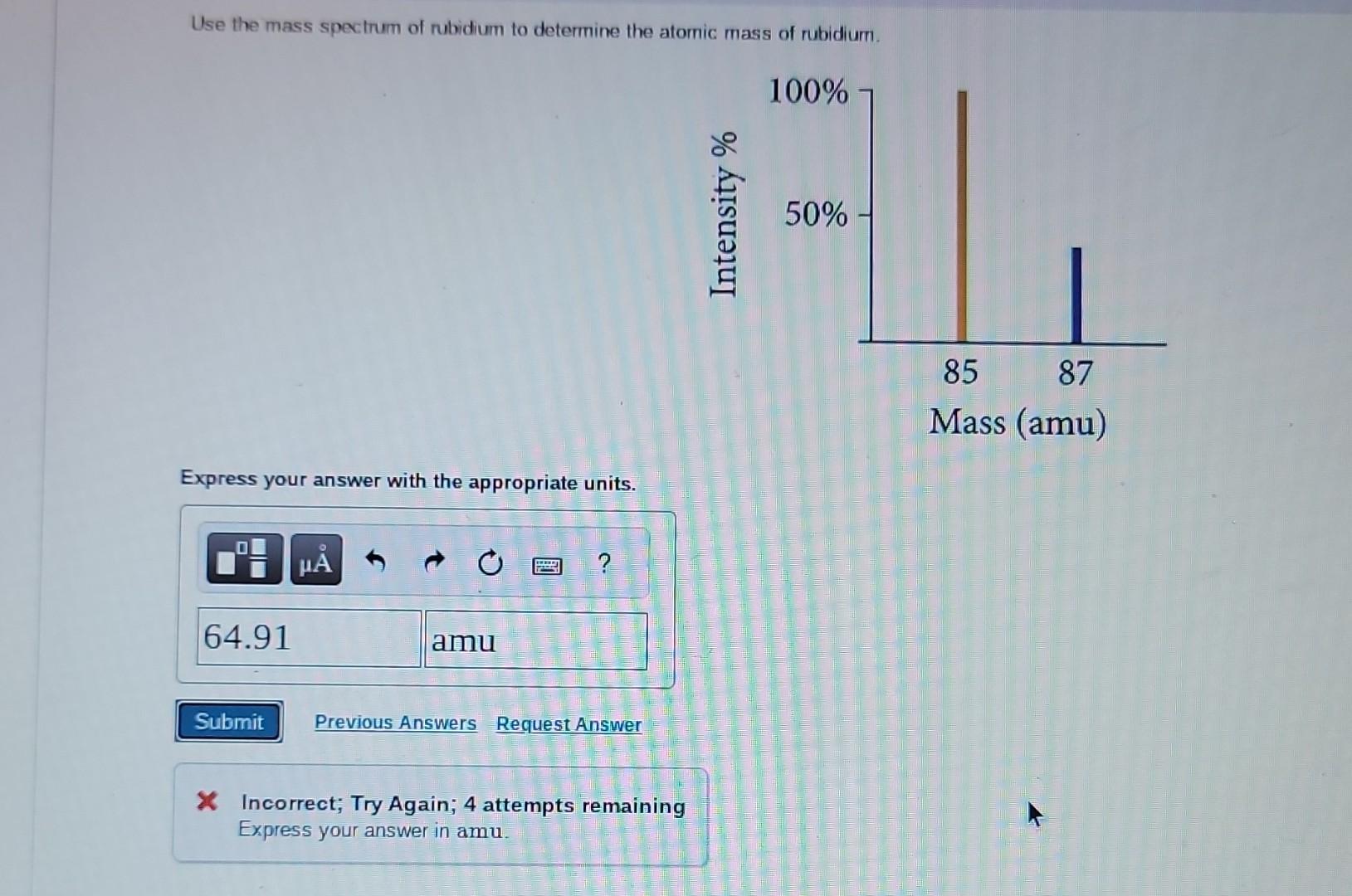
So, for Rubidium, we have two main siblings, two main isotopes. One is a bit more common, like the super popular sibling everyone knows. The other is a little rarer, like the sibling who’s always off on some quiet, fascinating adventure. These isotopes are Rubidium-85 and Rubidium-87.
Now, the names Rubidium-85 and Rubidium-87 sound super scientific, right? And they are! The "85" and "87" basically tell you the approximate weight of those particular Rubidium atoms. Think of it as their individual weight labels. The "85" one is a bit lighter, and the "87" one is a tad heavier. It’s like having two flavors of ice cream – one a little lighter on the palate, the other a bit richer.
Since Rubidium-85 is the more common sibling, it hangs out with us more often. It's like the class clown, always present and accounted for. Rubidium-87, being the more adventurous one, is a bit less frequent. It’s the one you might catch a glimpse of during a science experiment or on a rare geological find.
Because these two siblings, Rubidium-85 and Rubidium-87, exist in different amounts in nature, we can't just pick one and say, "This is the weight of Rubidium!" That wouldn't be fair to the other sibling, would it? It's like trying to describe the average height of all the people in your neighborhood by only measuring the tallest person. That's just not representative!
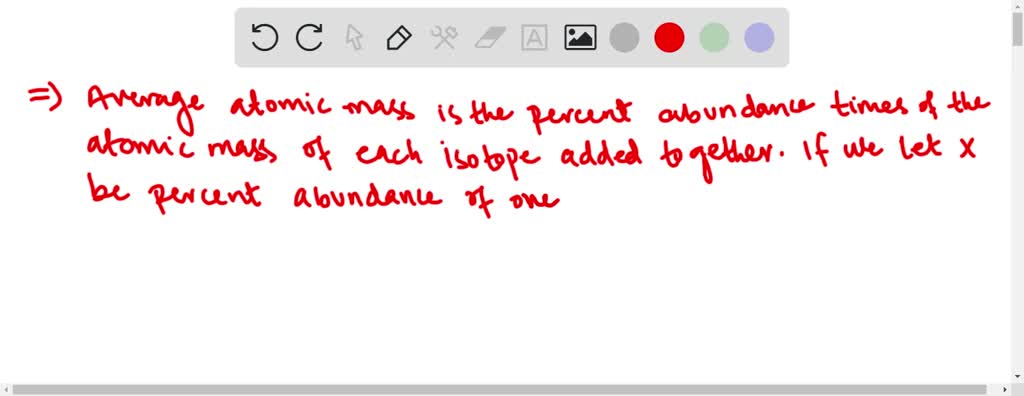
This is where the magic of the average atomic mass comes in. Scientists, with their incredible brains and fancy calculators, figure out how much of each sibling (isotope) is usually hanging out together in a natural sample of Rubidium. They then use this information to calculate a weighted average. It's like saying, "Okay, since Rubidium-85 is around 72% of the gang and Rubidium-87 is around 28%, we’ll give Rubidium-85 a bit more say in the final average weight."
So, after all that weighing and calculating and considering the popularity contest between siblings, we arrive at the average atomic mass of Rubidium. And guess what? It's approximately 85.468 atomic mass units! Yep, that’s the magic number!
Think of 85.468 as the super-duper, all-things-considered weight of a typical Rubidium atom. It’s not exactly 85, and it’s not exactly 87, but it's somewhere wonderfully in between. It's the representative weight that acknowledges both the common Rubidium-85 and the slightly rarer Rubidium-87. It's the compromise that tells the whole story!
This number, 85.468, is incredibly useful for scientists. It's like a universal ID card for Rubidium. When they're designing experiments, figuring out chemical reactions, or even looking for new materials, this average atomic mass is a crucial piece of information. It helps them predict how Rubidium will behave, almost like knowing your friend's personality helps you guess how they'll react to a surprise party!
So, the next time you hear the word "Rubidium," don't just think of a random element. Think of it as a fantastic family with two main siblings, Rubidium-85 and Rubidium-87. And their average atomic mass of 85.468 is the brilliant way we capture the essence of this whole fascinating family.
It's this kind of detail that makes the universe so incredibly intricate and beautiful. Even something as seemingly simple as the weight of an atom has its own fascinating story, a story of isotopes and averages and the cleverness of scientists.

Isn’t it amazing to think that something so small can have such a detailed and interesting "weight" profile? It's like discovering that your favorite cookie doesn't just taste good, it has a secret recipe of ingredients that make it perfectly chewy and perfectly crisp all at once!
So, give a little mental high-five to Rubidium and its impressive average atomic mass of 85.468. It’s a testament to the wonders of chemistry and the incredible diversity that exists, even at the atomic level. Keep exploring, keep wondering, and remember that even the most fundamental properties of matter can be a source of endless fun and discovery!
Who knew that numbers like 85.468 could be so exciting? Well, we do now! And that's the joy of diving into the world of elements. It's a journey that's always rewarding, and always, always interesting.
Just imagine – every time you see something made with Rubidium (and it’s used in some pretty cool stuff, like atomic clocks and even fireworks!), you can think, "Ah, there’s that element with the average atomic mass of 85.468!" It’s a little secret that makes you feel like a science superhero.

The beauty of it all is that this average mass is what makes Rubidium act the way it does in chemical reactions. It's this specific weight, this blend of its isotopic personalities, that determines how it bonds with other elements and what kinds of compounds it forms.
So, the next time you're looking at the periodic table, give Rubidium a special nod. It's not just another box on the chart; it's a testament to nature's cleverness and the power of scientific understanding to unravel its secrets, one atomic mass unit at a time. Isn't science just the coolest?
And there you have it! The average atomic mass of Rubidium, explained in a way that hopefully makes you feel a little bit smarter and a whole lot more entertained. Because learning about the universe should always be an adventure!
So, remember: Rubidium has siblings, and its average atomic mass of about 85.468 is the perfect way to describe the whole bunch!
It's this kind of curiosity that drives scientific discovery. We look at these numbers, we ask "why?", and we uncover the amazing stories hidden within. Rubidium's story is just one of many waiting to be told!