What Is The Average Atomic Mass Of Lithium

Ever wonder what makes things… well, things? It all comes down to tiny building blocks called atoms. And every element, from the stuff that makes you breathe to the metal in your phone, has its own special mix of these atoms.
Today, we're going on a little adventure into the world of Lithium. You might have heard of it, especially if you're into fancy batteries that power your gadgets. But Lithium has some secrets of its own, and one of them is its average atomic mass.
Now, "average atomic mass" might sound a bit like homework, but trust me, it's actually pretty neat! Think of it like this: not all atoms of the same element are exactly identical twins. They have cousins that are slightly heavier or lighter. That's where the "average" part comes in.
Must Read
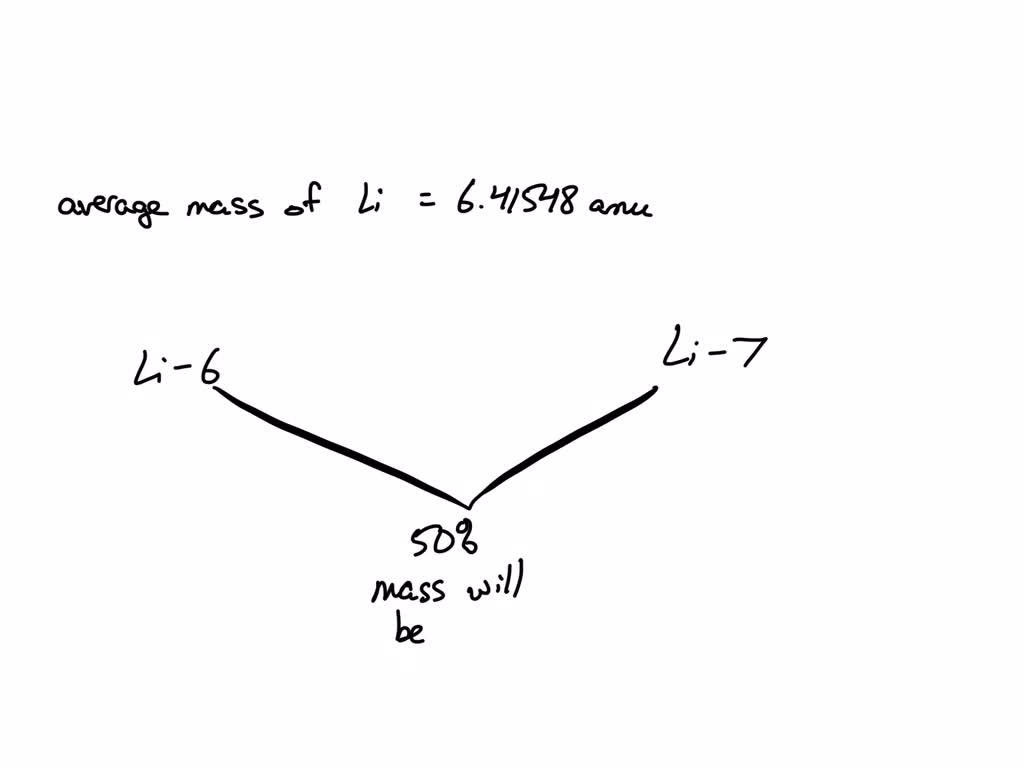
Lithium is a bit like a celebrity when it comes to its isotopes. Isotopes are those slightly different versions of an atom. For Lithium, the two main players are Lithium-6 and Lithium-7. They’re like the two members of a very popular band, each with their own personality (or rather, number of neutrons!).
These two isotopes are super important because they aren't present in equal amounts. It’s like one band member is way more famous than the other, so their "influence" on the band's overall sound is greater. For Lithium, Lithium-7 is the superstar, showing up way more often in nature than Lithium-6.
So, when scientists talk about the average atomic mass of Lithium, they're not just picking a random number. They’re taking into account how much of each isotope exists in the wild. It’s a weighted average, if you want to get fancy, but let’s just call it the "Lithium reality check."
This average is a specific number that tells us the typical weight of a Lithium atom you’d find floating around. It's a number that scientists use every single day, like a secret handshake in the chemistry club.
The average atomic mass of Lithium is approximately 6.94 atomic mass units (amu). Now, that number might not sound super exciting at first glance. But think about it – it’s the fingerprint of Lithium!

What makes this number special? Well, Lithium itself is pretty special. It's the lightest metal on the periodic table. That's like being the shortest person in a room full of giants, but still being incredibly strong and important.
And its average atomic mass reflects this lightness. It’s a relatively small number compared to, say, the average atomic mass of something like gold. It’s a testament to its small size and its fundamental nature.
Imagine you're collecting rare coins. Some coins might be worth a little, and some might be worth a lot. The average value of your coin collection would depend on how many of each type you have. The average atomic mass of Lithium is the same idea, but with atoms!
The exact value, 6.94, is a constant. It’s the answer to a long-standing question about Lithium’s identity. It’s not a made-up number; it’s derived from careful observations and measurements of how much Lithium-6 and Lithium-7 are out there.
Think about it like this: if you have a bag of apples, and most are small and a few are big, the average size of an apple in that bag won't be the size of the biggest apple. It will be closer to the size of the smaller, more common ones. That’s exactly what’s happening with Lithium's isotopes!
This specific number, 6.94, is crucial for chemists and physicists. When they’re working with Lithium in experiments or creating new materials, they need to know its typical weight. It helps them calculate how much of it they’re using, and how it will react with other elements.

It's like knowing the exact weight of a LEGO brick. If you're building a giant LEGO castle, knowing the weight of each individual brick helps you estimate the total weight of your masterpiece. The average atomic mass of Lithium is that precise LEGO brick weight for Lithium.
And the fact that it’s so close to 7 is no accident. It’s because Lithium-7 is so much more abundant. If Lithium-6 and Lithium-7 were equally common, the average would be exactly 6.5. But nature likes to keep things a bit more interesting!
This slight difference, this small deviation from a perfect 50/50 split, is what makes the average atomic mass of Lithium so fascinating. It’s a tiny clue about the history and formation of elements in the universe.
It tells us a story about how Lithium was created in the early days of the cosmos. The conditions back then favored the creation of more Lithium-7, and that imbalance has stuck around ever since.
So, next time you hear about Lithium, whether it's in a buzzing smartphone or a sleek electric car battery, remember its humble beginnings as a collection of atoms. And remember that even something as simple as its average atomic mass holds a universe of information.

It's a number that connects us to the very fabric of reality, a tiny piece of a grand cosmic puzzle. And it’s all thanks to the unique nature of Lithium and its isotopic friends!
The beauty of science is in these details. The seemingly small, specific numbers can unlock big understandings about how the world works. The average atomic mass of Lithium is one such number that quietly does its job, making sure our scientific calculations are spot on.
It's a reminder that even the smallest things have their own distinct characteristics. And learning about these characteristics can be a surprisingly engaging journey. Who knew an element's weight could be so intriguing?
So, if you ever find yourself curious about the building blocks of everything around you, don't shy away from numbers like "6.94 amu." They're not just abstract figures; they are the keys to unlocking the secrets of the universe, one atom at a time. And Lithium, with its lightweight charm and its specific average atomic mass, is a perfect starting point for such an exploration.
It’s a little bit like a detective story. Each isotope is a suspect, and their abundance is the evidence. Scientists put all that evidence together to come up with the overall profile, the average atomic mass, which tells us who Lithium really is.
The fact that this number is so consistent, so reliable, is what makes it so valuable. It’s a constant in a universe that is always changing. It’s a stable point of reference for all sorts of scientific endeavors.
And isn't that neat? That something so fundamental, so much a part of the world, can be described by a single, precise number? It's a testament to the order and beauty that exists in even the smallest scales of existence.
So, the next time you see that little "Li" on a periodic table, give it a nod. It’s not just an element; it’s a story, a history, and a number that tells a tale of cosmic proportions. The average atomic mass of Lithium is more than just a value; it's an invitation to explore the wonders of chemistry.
It's a fun fact waiting to be shared, a spark to ignite further curiosity about the unseen world that makes up everything we see and touch. Isn't that an awesome thought?
And it all boils down to Lithium having these two forms, these two isotopes, with slightly different weights. The way they're mixed together in nature gives us that magic number, that average atomic mass, that is so important.
It's a perfect example of how science takes complex phenomena and distills them into understandable, usable information. This average is what allows us to work with Lithium practically and understand its place in the grand scheme of things.
So, if you’re ever bored, remember the average atomic mass of Lithium. It's a little piece of the universe, waiting to be appreciated. It's a tiny number with a big story to tell. And it all adds up to something pretty extraordinary!

+and+92.58%25+exists+as+Li-7+(7.016+g/mol).jpg)
.jpg)


