What Ions Are Formed When Alkalis Dissolve In Water

Get ready for a little splash of science fun, because we're diving headfirst into the super cool world of what happens when those fizzy, bubbly alkali metals decide to take a dip in a nice, refreshing glass of water! Think of it like this: these metals are the life of the party, and water is their favorite dance floor. And when they get together, oh boy, do they create something special!
You know those metals that are like the energetic toddlers of the periodic table? The ones that can't sit still and are always eager to react? Yep, those are our alkali metals! We're talking about the crew that includes elements like lithium (the tiny but mighty one), sodium (you probably know this guy!), potassium (the real heavyweight champion of reactivity), and a few other fancy pals.
Now, imagine you’ve got a tiny speck of, say, sodium. It’s this shiny, silvery stuff that’s so eager to get going, it’s practically vibrating. And then you introduce it to water. Water, which is usually so calm and collected, suddenly gets a jolt of pure excitement! It's like the quiet librarian suddenly breaking out into a spontaneous tap dance.
Must Read
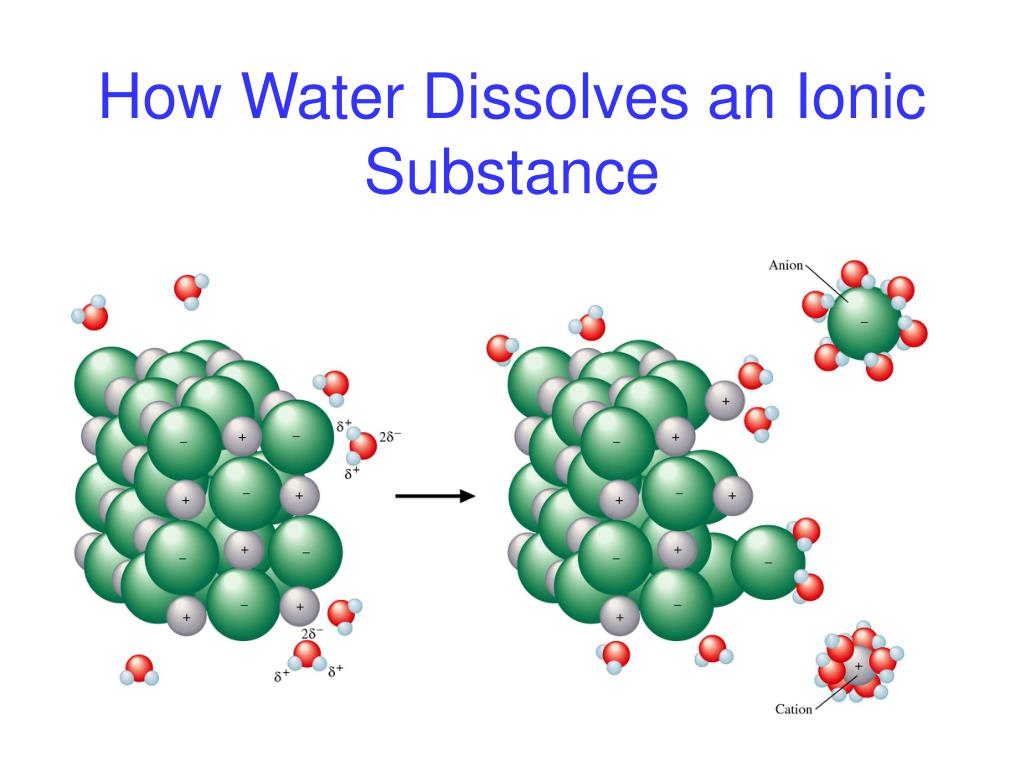
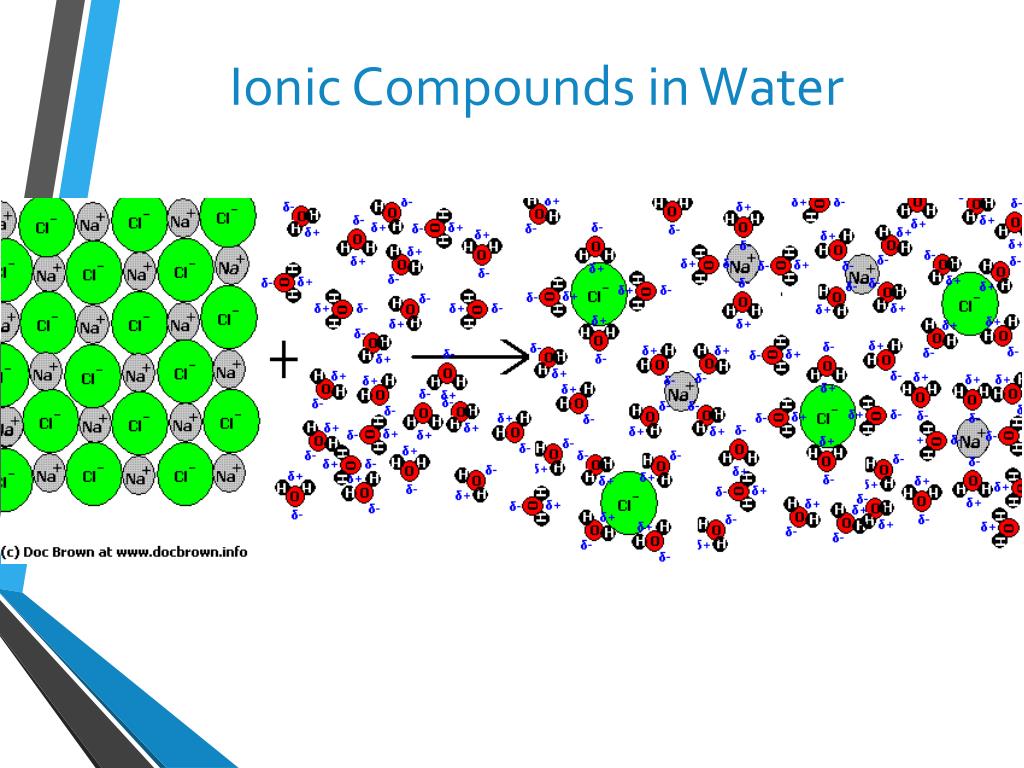
When these alkali metals hit the water, they don’t just sort of… dissolve. Oh no, that would be far too boring! They go through a magnificent transformation. They actually break apart, and in doing so, they release a brand new character onto the scene. This new character is called an ion. And not just any ion, but a very specific kind of ion that carries a little bit of a charge, like a tiny electric spark!
Specifically, when an alkali metal like sodium (which we’ll call Na for short, because scientists love their abbreviations!) dives into water, it sheds one of its electrons. This electron is like a tiny piece of luggage it decides it doesn’t need for its water adventure. And what’s left behind? A positively charged sodium ion! We write this as Na+. Imagine our sodium atom, which was minding its own business, suddenly going, "Whee! I'm lighter and I'm zipping around with a positive vibe!"

It's kind of like when you're at a party and you're feeling super happy and energetic – you've got that positive energy radiating off you! That's the alkali metal ion. It's the energetic, charged-up version of the original metal, ready to mingle and make things happen in the water.
And it’s not just sodium! Our friend lithium (Li) does the same thing. It jumps in, drops an electron, and becomes a lithium ion, or Li+. It’s like the mini-me version of the sodium ion, still positively charged and ready to party. Then there’s potassium (K), the grand poobah of these reactions. When potassium meets water, it gets so excited it practically explodes with enthusiasm, leaving behind a positively charged potassium ion, or K+. It’s like the rockstar of the alkali metal ions, leaving a trail of positive excitement wherever it goes.

Think of it like this: each alkali metal is a superhero with a cape. When they meet water, they shed their cape (that's the electron!) and become a super-charged hero with a positive aura! And these positive heroes are called cations. They're the positively charged ions that our friendly neighborhood alkali metals happily become.
So, what are these ions actually doing in the water? Well, they’re not just floating around looking pretty. They're now dissolved, dispersed, and ready to influence all sorts of things. They’re like the new enthusiastic attendees at the water party, making sure the vibe stays energetic and interesting. They can help other things dissolve, or they can change the properties of the water itself. It's a whole new world for them, and they embrace it with open arms – or should we say, with their positive charges!

The water itself also plays a starring role. It’s not just a passive observer. The water molecules are the super-welcoming hosts of this party. They surround these newly formed positive ions, kind of like giving them a warm, bubbly hug. This process is called hydration, and it’s what keeps these ions happily dissolved and floating around, continuing their energetic dance.
So, next time you hear about alkali metals and water, just remember the incredible transformation! It’s not just a simple dissolve; it’s a dazzling display of chemistry where these metals become super-charged, positively buzzing ions, ready to bring their unique brand of excitement to the aqueous world. They’re the happy, energetic, positively charged party starters, and the water is their perfect dance floor!