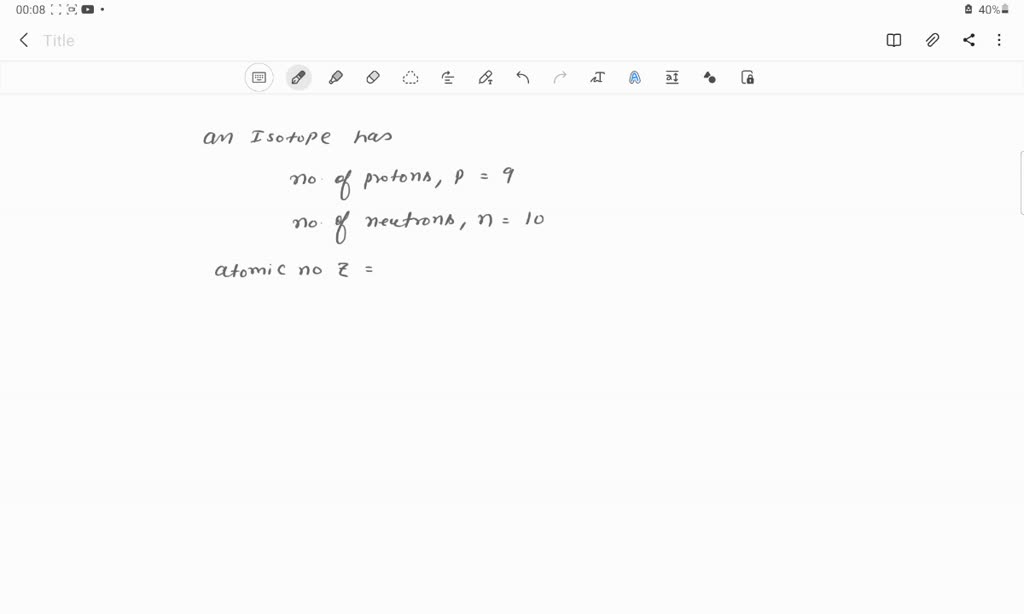
What Element Has 9 Protons And 10 Neutrons

Ever wondered what makes a masterpiece shimmer with that undeniable glow? Or perhaps you've marveled at the vibrant hues in stained glass windows, or the delicate beauty of antique jewelry. There's a certain element, a true crowd-pleaser in the artistic and scientific world, that often plays a starring role. It's an element known for its remarkable versatility and its ability to inspire creativity across a spectrum of endeavors. This popular substance, with a foundational identity of 9 protons, and a common variant boasting 10 neutrons, is none other than Fluorine! Wait, did we say Fluorine? Oh dear, a slight miscalculation. Let's rewind. The element with 9 protons is indeed Fluorine. However, when we're talking about an element with 9 protons and 10 neutrons, we're actually referring to the element Fluorine, specifically the isotope Fluorine-19. A fascinating quirk of atomic structure, isn't it? But let's pivot to an element that truly captures the imagination when it comes to its creative applications, an element that is often associated with its colorful compounds and its fascinating scientific journey. We're talking about the element that, when it has 9 protons and a stable atomic weight, is the dazzling Fluorine itself. Let's explore its creative potential!
For artists, hobbyists, and anyone with a curious mind, engaging with the world of Fluorine, or rather, its applications, can be incredibly rewarding. While you can't exactly sculpt with Fluorine gas (it's a bit too reactive!), its compounds are where the magic happens. Think about the brilliant blues and greens in certain ceramic glazes – that's often the work of fluorine compounds! For hobbyists building intricate models, the durability and clarity of certain fluorine-infused plastics offer fantastic possibilities. And for the casual learner, understanding how this element contributes to everyday objects, from non-stick cookware to the fluoride in your toothpaste, is a tiny spark of scientific discovery.
The examples of Fluorine's influence are widespread. In art, think of the luminous quality of certain glass types; fluorine compounds can enhance their refractive properties. Historically, the vibrant pigments used in ancient pottery often relied on metallic compounds that, when fired, interacted with trace elements to create stunning colors. While not directly Fluorine itself, its presence as an additive or impurity in materials can subtly alter the final aesthetic. For those interested in DIY projects, exploring the science behind non-stick coatings or even the chemistry of cleaning agents can offer a hands-on learning experience.
Must Read
Trying to explore the creative side of elements at home can be as simple as observing the world around you. Next time you're in the kitchen, examine your non-stick pan. That's a direct application of fluorine chemistry! Or, consider the safety glass in your car windows; it often incorporates fluorine-based materials for enhanced strength and clarity. For a more hands-on (and supervised!) approach, exploring educational chemistry kits can introduce you to safe reactions that showcase the principles behind various elemental compounds. Remember, the key is to appreciate the underlying science and how elements contribute to the materials we use and admire every day.
Ultimately, exploring the world of elements, even from a distance, is inherently enjoyable. It’s about understanding the building blocks of our universe and appreciating the ingenuity of both nature and human innovation. The element with 9 protons, Fluorine, may be reactive and gaseous in its pure form, but its widespread influence on the materials we encounter daily offers a constant source of wonder and creative inspiration. It’s a reminder that even the invisible can have a profound and beautiful impact on our world.