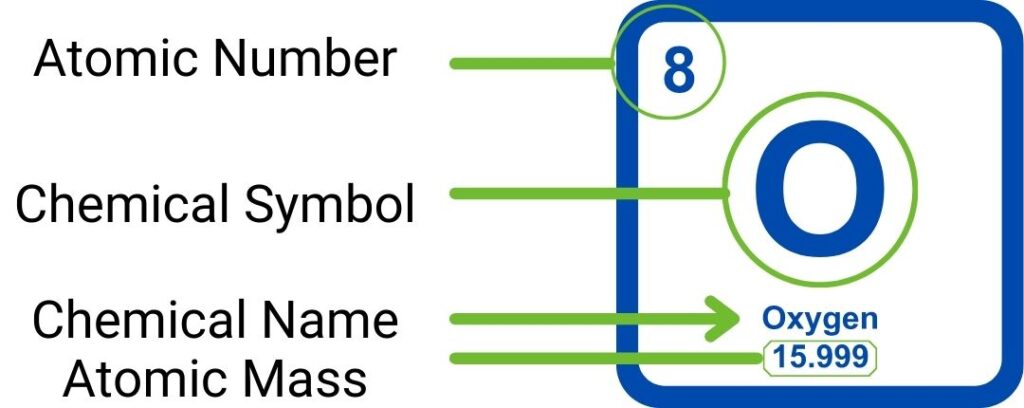
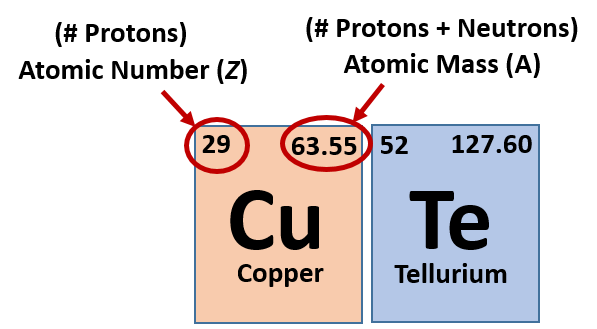
What Does The Number Next To The Ion Signify

So, you’re at the grocery store, right? You’re staring at this big ol’ tub of yogurt. Now, most of us are just looking for the flavor that screams “eat me now!” or maybe the one that promises to make our insides do a happy dance. But then you notice it. Tucked away, almost shyly, is a little number next to this thing called an “ion.” And your brain does that little whirr-click sound, like a vintage cash register trying to process a foreign coin. What in the name of all things creamy and cultured is this number doing here? Is it the yogurt’s age? Its IQ? The number of tiny dance parties happening inside?
Well, buckle up, buttercups, because we’re about to dive into the not-so-scary, surprisingly fascinating world of ions and their little number friends. Think of me as your friendly neighborhood science-explainer, armed with nothing but a slightly-too-strong latte and an unhealthy obsession with making chemistry sound less like a lecture and more like a particularly juicy gossip session.
The Case of the Missing Electrons (and the Extra Ones!)
Alright, let’s get down to brass tacks. What’s an ion? Imagine atoms are like tiny LEGO bricks. They’re pretty happy being neutral, minding their own business, with a perfectly balanced set of protons (the positively charged guys) and electrons (the negatively charged dudes). Everything’s chill. It’s like a perfect equilibrium, a teacup party where everyone’s got the right amount of sugar.
Must Read
But sometimes, these atoms get a little… antsy. They decide they need to either borrow an electron or lend one out. It’s like that friend who always forgets their wallet or the one who’s always offering to pay for everyone. When an atom either loses or gains an electron, it’s no longer neutral. It’s become an ion.
And that’s where the number comes in! This little digit is basically a report card on just how much electron-borrowing or lending has gone down. It tells you the charge of the ion. Think of it as the score in a cosmic game of electron tag.

The Minus Sign Crew: Gaining Electron Power!
Now, if you see a negative sign next to that number, like ‘Cl–’ (that’s chloride, by the way, you might see it in salt!), it means our atom has been a bit of a hoarder. It’s gained one or more electrons. Since electrons are negatively charged, when an atom gets more of them, it becomes negatively charged overall. It’s like stuffing your pockets with extra candy – you’re now carrying more sweet, sweet negativity (but in a good way!).
So, a ‘–1’ means it gained one electron. A ‘–2’ means it’s really gone wild and nabbed two. This is the anion club, the electron-rich rebels of the atomic world. They’re the ones who are always up for an extra snack.
The Plus Sign Posse: Lending a Helping Hand (or Electron)
On the flip side, you’ve got the positive sign. If you see something like ‘Na+’ (sodium, a star player in table salt and, surprisingly, your bananas!), that means our atom has been generous. It’s lost one or more electrons. When you lose those negative charges, you’re left with a surplus of positive protons. It’s like giving away your last cookie – you’re feeling a little lighter, a little more positively charged about the universe.
A ‘+1’ means it lost one electron. A ‘+2’ means it’s feeling extra benevolent and donated two. These are the cations, the electron-donating philanthropists. They’re the ones who’ll always offer you the last bite of their pizza, even though they secretly want it.
Why Should We Care About These Electron-y Numbers?
Okay, so we’ve got charged atoms running around. Big whoop, right? Well, actually, it’s a huge whoop! These charged little guys are the glue that holds so much of our world together. They’re involved in everything from the spark of lightning to the intricate workings of your brain.

Take your yogurt, for instance. Those little cultures doing their magical fermentation thing? They’re often working with ions. The tartness you taste? That can be due to hydrogen ions (H+). The way proteins fold and interact? Ions play a massive role. It’s like the unsung heroes of the dairy aisle, silently orchestrating deliciousness.
And it’s not just food! In your body, ions are doing a million things simultaneously. Sodium (Na+) and potassium (K+) ions are crucial for nerve signals. Calcium ions (Ca2+) are like the construction workers of your bones and teeth. Even the electrical charge that makes your muscles contract? Yep, you guessed it: ions!
A Splash of Unexpected Science
Here’s a fun fact to blow your mind: Did you know that the salty taste of tears is primarily due to sodium chloride (NaCl) dissociating into sodium ions (Na+) and chloride ions (Cl–)? So, when you cry tears of joy or sorrow, you’re literally tasting a chemical reaction. Science is everywhere, folks, even in your most dramatic moments!
Another cool tidbit: The vibrant colors of fireworks? Often created by heating metal salts, which causes their ions to emit specific wavelengths of light. So, that dazzling red or brilliant blue is actually the excited dance of ions showing off their electron-induced glow.
The Yogurt Finale (and a little bit of calcium)
So, the next time you’re at the supermarket, feeling a bit peckish for that creamy delight, don’t just glance at the yogurt. Peer closer. See that number next to the ion? It’s not just a random digit. It’s a tiny, powerful story about electrons gained or lost, a fundamental aspect of chemistry that’s literally keeping you alive, making your food tasty, and even painting the night sky with explosions of color. It's the secret handshake of the molecular world, and you, my friend, are now in on it.
So go forth, armed with your newfound knowledge. Impress your friends. Confuse your enemies. And maybe, just maybe, enjoy your yogurt a little bit more, knowing the complex dance of ions happening within. Who knew a simple number could be so… electrifying?





