Use The Bond Energies Provided To Estimate
So, have you ever wondered how much oomph it takes to break a chemical bond? Like, the sticky stuff that holds atoms together? It’s not just magic, you know. We can actually estimate it!
Think of it like LEGO bricks. Atoms are our bricks, and chemical bonds are the little connectors. Some connectors are super strong, like the ones holding your favorite toy together. Others are a bit wobbly, ready to pop off with a little nudge.
And guess what? Scientists have a cool way to measure this “stickiness.” It’s called bond energy. Basically, it’s the amount of energy needed to break a specific type of bond. Pretty neat, right?
Must Read
We’re going to use some of these handy bond energies to play a little game. It’s like being a detective, but instead of fingerprints, we’re looking for energy clues!
Imagine you have a molecule. It’s like a tiny, intricate sculpture made of atoms. To figure out the total energy needed to smash that sculpture into its individual atom parts, we can add up the energy needed to break each little connector, or bond.
It's a bit like taking apart a really complex puzzle. You know how much effort it takes to pull apart each individual piece. If you add up all those efforts, you get a pretty good idea of how much work it took to put the whole thing together in the first place!
And here's where it gets fun. We're not going to do super-duper complex calculations. We're just going to estimate. Think of it as a scientific approximation, a really educated guess. Because sometimes, the exact answer isn't as important as understanding the general idea.
Why is this even a thing?
Well, understanding bond energies helps us predict how chemical reactions will happen. Will a reaction release a ton of energy, like a tiny explosion? Or will it need a big energy boost to even get started? It’s like knowing if a recipe is going to be spicy or require a lot of stirring.

Plus, it’s just cool to know that we can quantify these invisible forces. The forces that hold everything around us together. Your coffee mug, your phone, even you! All held together by these amazing little bonds.
Think about water, H₂O. It’s two hydrogen atoms chilling with one oxygen atom. There are two O-H bonds in there. If we know the bond energy for an O-H bond, we can get a ballpark figure for how much energy it would take to rip that water molecule apart into individual hydrogen and oxygen atoms.
It’s like knowing the price of a single LEGO brick and then estimating the cost of a whole castle by counting the bricks. Simple, right? Well, almost!
The Quirky Side of Bonds
Did you know that the same type of bond can have slightly different energies depending on the molecule it’s in? It’s like how a hug from your best friend might feel a little different than a hug from your grandma. They're both hugs, but the context matters!
This is because the other atoms attached to a bond can influence how strong it is. They can either pull on the bond, making it tighter, or push on it, making it a bit looser.

And sometimes, when we break bonds, they don't just break cleanly. They might do a little dance, rearrange themselves, and form new bonds! Chemical reactions are a bit like a chaotic party where the guests (atoms) swap dance partners (bonds).
So, when we’re estimating bond energies, we’re using average values. These are like the "average" temperature for a particular city. It's a useful number, but some days it'll be hotter, and some days it'll be colder.
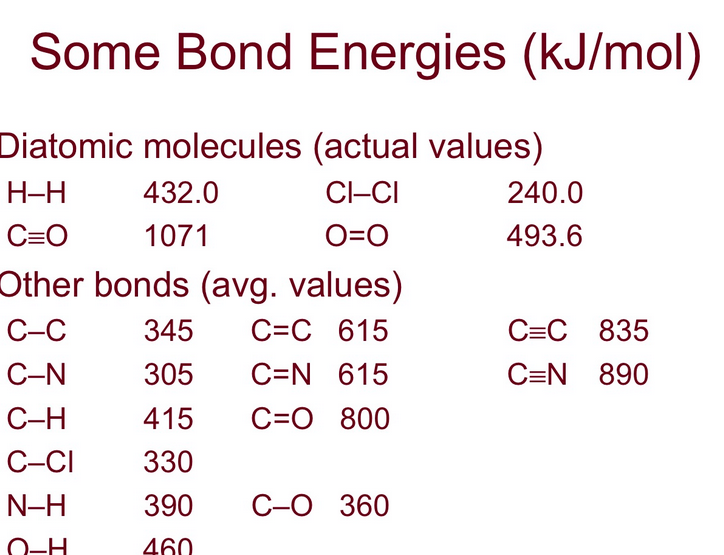
Our bond energy table is our cheat sheet. It’s got all these numbers, like a secret codebook for the molecular world. We'll be looking at things like C-H bonds, O-H bonds, and maybe even some fancy double bonds, like C=O.
Let’s Get Estimating!
Okay, ready to put on our detective hats? Imagine we have a molecule of methane, CH₄. It’s basically a carbon atom with four hydrogen atoms clinging to it. There are four C-H bonds.
If we look up the average bond energy for a C-H bond, let’s say it’s around 413 kilojoules per mole (kJ/mol). That's just a unit of energy, don't get bogged down by it!

So, to break all four C-H bonds in methane, we’d take that 413 kJ/mol and multiply it by four. 413 * 4 = 1652 kJ/mol. Boom! That’s our estimated energy to break methane into a carbon atom and four hydrogen atoms.
See? We’re not doing rocket science here. We're just doing molecular science estimation!
Now, what about a slightly more complex molecule, like ethanol? That's the stuff in your fizzy drink (the non-alcoholic kind, of course!). Ethanol has a formula like C₂H₅OH. It's got some C-C bonds, some C-H bonds, and an O-H bond.
We'd need to know the bond energies for all those different types. So, let's say we have our handy table:
- C-C bond: ~347 kJ/mol
- C-H bond: ~413 kJ/mol
- O-H bond: ~463 kJ/mol
Now, we need to count how many of each bond are in ethanol. It's got one C-C bond, five C-H bonds (two on one carbon, three on the other), and one O-H bond. So, we do the math:

- (1 * 347) + (5 * 413) + (1 * 463) = 347 + 2065 + 463 = 2875 kJ/mol.
So, it would take about 2875 kJ/mol to break all the bonds in an ethanol molecule. Pretty cool, huh? It's like deconstructing a miniature molecular toy!
Why It's More Than Just Numbers
This whole bond energy thing isn't just about memorizing numbers. It's about understanding the relationships between atoms. It’s about seeing the hidden forces that govern our universe.
And when you start to see these patterns, you can begin to predict things. Like, if a reaction involves breaking a lot of strong bonds and forming weaker ones, it might release a lot of energy. If it’s the other way around, it’ll need energy to happen.
It’s like understanding the ebb and flow of a river. You can’t control it, but you can learn to predict its movements. And that’s powerful!
So, next time you look at a chemical formula, don’t just see a jumble of letters and numbers. See a tiny, intricate structure held together by invisible forces. And know that with a little bit of information – those handy bond energies – you can start to unravel the secrets of its strength!
It’s a fun way to peek behind the curtain of chemistry and see the fundamental building blocks of our world in action. And who knows? You might just discover a new appreciation for the invisible glue that holds everything together!

