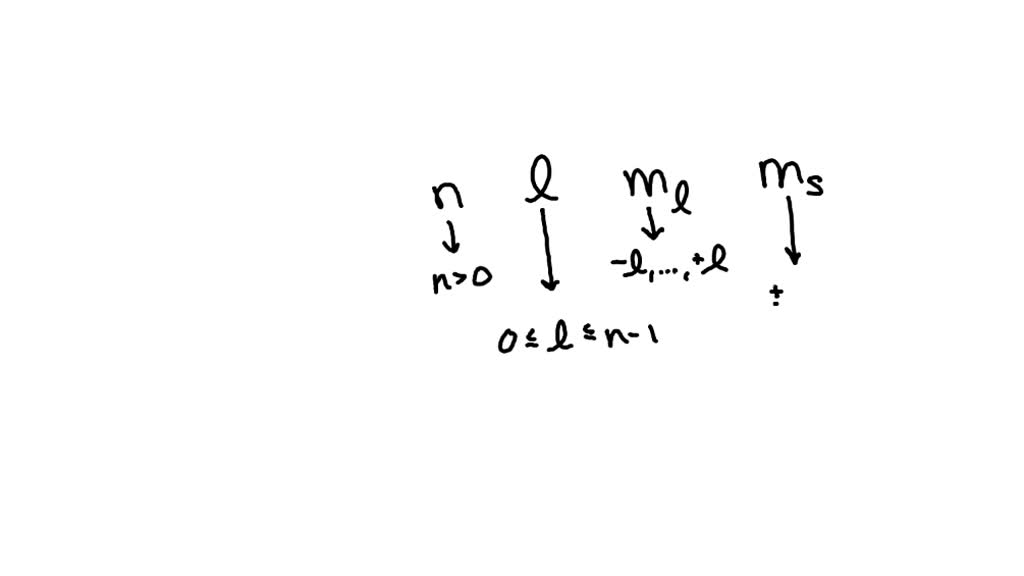
The Quantum Number Ml Can Have Values From

Imagine you're at a really fancy, exclusive party, like, the kind where only the coolest atoms get invited. These atoms have tiny little "rooms" where their electrons hang out. Think of these rooms as being like little apartments within the atom's overall "building."
Now, not all these rooms are the same. Some are shaped like cozy little spheres, others are like elegant dumbbells, and some get a bit more adventurous with their shapes. It's like a neighborhood with all sorts of architectural styles!
But here's where it gets interesting, and a little quirky. Within each of these "rooms," the electrons can orient themselves in different directions. It’s like how you can park your car facing north, south, east, or west, even if you're in the same parking spot.
Must Read
This particular little quirk is described by something scientists call the magnetic quantum number, or ml for short. It's a fancy name for a surprisingly simple idea: how the electron's "space" is angled or pointed in the atom's universe.
So, what values can this ml take? Well, it depends entirely on the "room" the electron is in. Think of it like this: a simple, spherical room doesn't offer many directional choices. It's pretty much just... there.
For those neat, spherical rooms (which scientists call s orbitals, bless their hearts), there's only one way to point: straight ahead, no matter which way you turn. So, for these s rooms, ml can only be 0. It’s like having a single parking space that’s just a circle – you can’t really angle it any other way.
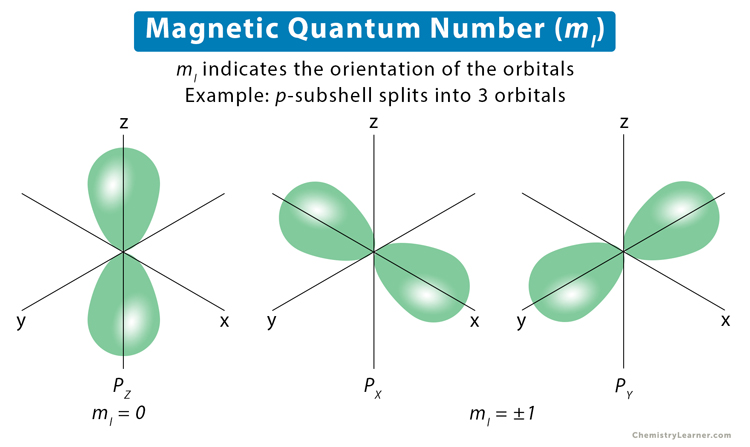
But then you have the dumbbell-shaped rooms, the p orbitals. These are much more fun! Imagine three of these dumbbell rooms all clustered together, like a little trio of excited puppies.
These p orbitals can point along the x-axis, the y-axis, or the z-axis. It's like having three distinct parking spots, each perfectly aligned with a different street in your atomic town.

Because of these three distinct directions, the ml value for a p orbital can be -1, 0, or +1. It's a little spectrum of orientations, allowing for three different ways an electron can "sit" within that cluster of dumbbell rooms.
It's like the atom is saying, "Okay, you're in the p room area. You can be parked along this line, or this line, or this one!" It gives the electrons a bit of freedom in how they arrange themselves.
Now, let's crank up the complexity (and the fun!) with the d orbitals. These are the real show-offs of the atomic world, with shapes that look like clovers, or even just strange, squiggly lines.
There are typically five of these d orbitals in a given energy level. And these five orbitals have a whole lot more ways to point and twist and turn.
For the d orbitals, the ml values can range from -2, through -1, 0, +1, all the way to +2. That's a total of five different possible orientations for the electrons in these rooms!
It's like having five different movie theaters, each showing a slightly different version of the same blockbuster. The electrons can pick their preferred viewing angle, so to speak.
Think of the atom as a cosmic apartment complex. Each floor is a different energy level, and each apartment on a floor has a certain shape (the orbital). The ml number is like the specific orientation of that apartment's furniture within its allotted space.
A spherical apartment (s orbital) only has one way to be arranged – it’s pretty minimalist. So, ml is just 0. There’s no room for variation.
The dumbbell-shaped apartments (p orbitals) are a bit more stylish. They can be arranged along the x, y, or z direction. So, ml gets to be -1, 0, or +1. They’re offering you different views from your window!
And then there are the deluxe, multi-room apartments (d orbitals). These have intricate layouts, allowing for many more orientations. The ml values expand to -2, -1, 0, +1, +2. It's like having a penthouse with a panoramic view and rotating furniture!
The beauty of this system is that it's not just arbitrary. These orientations have real-world consequences, influencing how atoms interact with light and with each other. It's the subtle details that make all the difference in the grand atomic dance.
The range of these ml values is always determined by a number called the angular momentum quantum number, or l. If you have an l value of 0 (that's the spherical s orbital), ml can only be 0. Simple and sweet.
If you have an l value of 1 (that's the dumbbell p orbitals), ml can be -1, 0, or +1. It's a set of three, perfectly coordinated.
And if you have an l value of 2 (those fancy d orbitals), ml stretches to -2, -1, 0, +1, +2. A whole family of possibilities!
It's like having a set of nesting dolls, but instead of dolls, they are possibilities. For each "size" of orbital shape (determined by l), there's a specific set of directional choices (determined by ml).

The number of possible ml values is always 2l + 1. This formula neatly sums up all those possible orientations. It's a little mathematical secret that unlocks the full directional palette for each orbital shape.
So, the next time you think about atoms and electrons, remember that it's not just about them zipping around randomly. They're actually residing in very specific, spatially oriented "rooms," and the ml quantum number tells us exactly how those rooms are angled.
It’s a surprisingly organized and elegant system, governing the unseen architecture of matter. And it all starts with the simple idea of how an electron's space can be pointed in the universe.
These quantum numbers, including ml, are like the hidden instructions for building the universe, one atom at a time. They’re the secret handshake that allows electrons to find their designated spots and interact in predictable, yet wonderfully complex, ways.
So, while the names might sound a bit intimidating, the concept is quite charming. It's the universe's way of ensuring that even the tiniest particles have their own personal space and preferred orientation.
And that, in a nutshell, is how the magnetic quantum number ml can have values from a simple 0 to a more complex spread of integers, all depending on the shape and elegance of the electron's atomic abode. It's a tiny detail with enormous implications for the world we see!