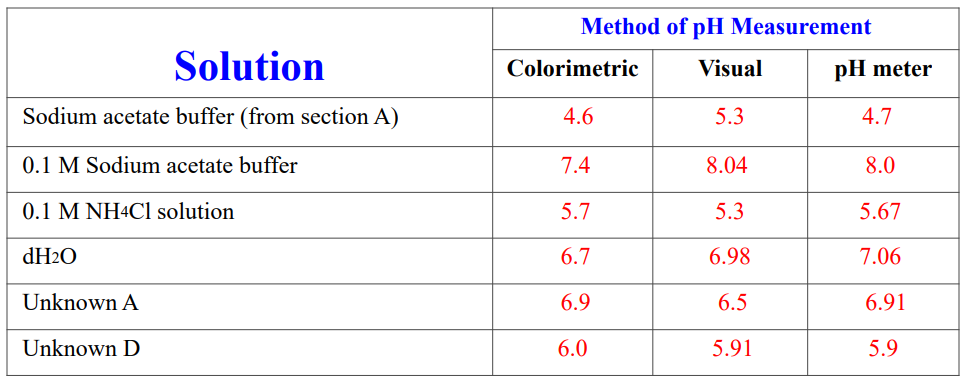
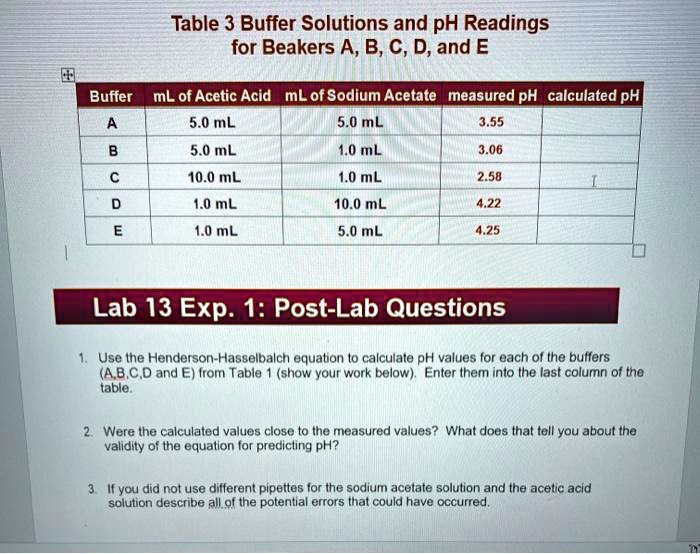
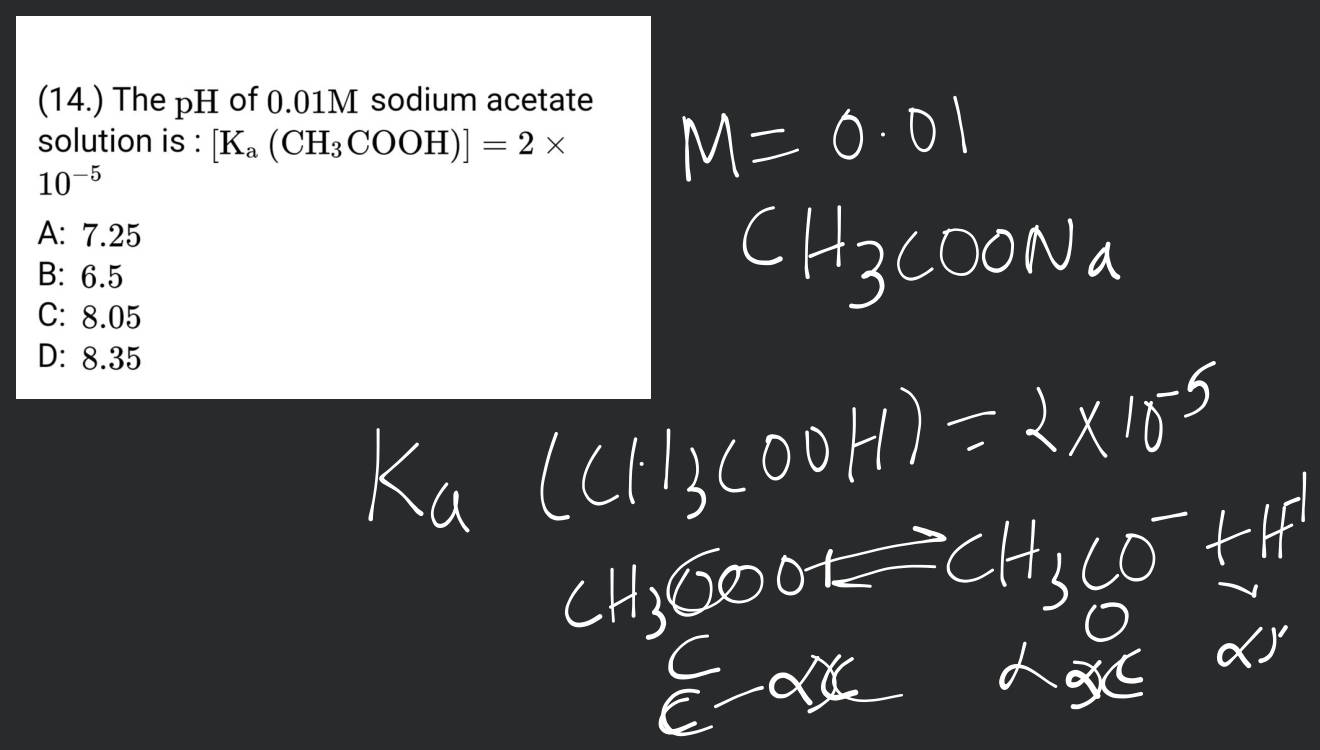The Ph Of A 1.0 M Sodium Acetate Solution Is

Let's talk about something that sounds a bit fancy, but is actually quite down-to-earth. We're diving into the world of sodium acetate. Think of it as a salt, but with a bit more personality. And when we talk about a "1.0 M" solution, it just means we've mixed a good, solid amount of it in water. Not too little, not too much, just a nice, respectable concentration.
Now, the big question that might be keeping you up at night (or not, which is totally fine too!) is about its pH. This is the number that tells us if something is acidic, neutral, or basic. You know, like lemon juice is acidic and baking soda is basic. So, where does our friend, sodium acetate, land on this scale?
Here's where things get a little juicy. Many people might assume that because it's a salt, it's just going to chill in the middle, a perfect pH of 7. That's the neutral zone, the Switzerland of pH. But nope! Our 1.0 M sodium acetate solution has a little secret.
Must Read
It leans towards the basic side. Yep, you heard me. It's not aggressively basic like bleach, but it's definitely not neutral. So, if you were to measure it with a pH strip, you'd see a number higher than 7. Maybe around 8 or even a little more, depending on the exact conditions.
This is kind of like discovering your quiet neighbor actually has a secret passion for karaoke. It's unexpected, a little surprising, and makes them all the more interesting. And honestly, I kind of like that about sodium acetate. It doesn't just do what you expect.
Why does this happen? Well, it's a little chemistry party going on. Sodium acetate is formed from a strong base (sodium hydroxide) and a weak acid (acetic acid). When they get together in water, the little bits and pieces start to play. The acetate part of the sodium acetate is a bit like a water-lover, but it also likes to hold onto its hydrogen atoms.

When it interacts with water, it can actually "borrow" a hydrogen ion from the water molecule. Water, bless its heart, is pretty happy to let go of one of its hydrogens. And when water loses a hydrogen, it becomes hydroxide. More hydroxide ions in solution means a higher pH. See? It's not magic, it's just chemistry being a little mischievous.
So, the next time you hear someone talking about the pH of a 1.0 M sodium acetate solution, you can confidently nod and say, "Ah, yes, it's a little bit basic, isn't it?" You'll sound like you've got all the answers, when really, you just understand that salts can be more complex than they seem.
It's the same feeling you get when you realize your favorite comfy sweater is actually a designer piece. You knew it was good, but now you know it's got a bit of that extra oomph. Sodium acetate is like that sweater. Reliable, useful, and with a subtle, pleasant surprise.
Think about it this way: If water is a perfectly calm lake, a neutral pH is like a peaceful boat ride right in the middle. An acidic solution is like paddling towards a waterfall – exciting, but maybe a bit too much action for some. A basic solution, like our sodium acetate, is more like drifting towards a gentle, sunny shore. It's a bit warmer, a bit more welcoming, and just feels a touch more relaxing.
![Acetic acid concentration, [HA], at equilibrium Dissociation constant](https://cdn.numerade.com/ask_images/6eccfa07da68434384d5ce378a8a67be.jpg)
And in a world that can sometimes feel too acidic, a little bit of gentle basicity from a 1.0 M sodium acetate solution sounds pretty appealing, right? It’s like a calm sigh after a long day. It’s not trying to be something it's not. It’s just… sodium acetate.
I mean, who decided that "neutral" had to be the ultimate goal all the time? Sometimes, a little bit of "leaning" is exactly what you need. It's the difference between a polite nod and a warm hug. Sodium acetate, in its 1.0 M glory, offers that gentle, embracing hug. It’s not a dramatic pH shift, but a subtle, noticeable one.
This is also why buffers are so cool. Sodium acetate is a key player in buffer solutions. Buffers are like the peacekeepers of the chemistry world. They resist changes in pH. So, if you add a little bit of acid or base to a buffer solution, it doesn't go haywire. It stays relatively stable.
A 1.0 M sodium acetate solution, by itself, isn't a strong buffer, but it's a building block for them. It's like the sturdy foundation of a house. You need that solid base to create something that can withstand the elements. And what are the elements in this case? Little splashes of acid or base!

So, while you might not be mixing up a 1.0 M sodium acetate solution in your kitchen anytime soon (unless you're a culinary adventurer!), understanding its pH is like knowing a fun fact about a celebrity. It makes them a little more relatable, a little more interesting. You can casually drop it into conversation at your next science-themed (or just general) gathering.
"Did you know," you might say, with a twinkle in your eye, "that a 1.0 M sodium acetate solution isn't actually neutral? It's a tad bit basic!" And then, you can watch their eyes widen, or perhaps they'll just nod along, secretly impressed by your pH prowess.
It’s that satisfying feeling of knowing something a little bit niche, a little bit unexpected. It’s the opposite of an unpopular opinion, really. It’s more like a well-kept, easily explained secret. And who doesn't love a good secret?
Ultimately, the pH of a 1.0 M sodium acetate solution is just a number, sure. But it's a number that tells a story. A story about how things that seem simple can have hidden depths. It’s a reminder that in chemistry, as in life, things aren’t always as straightforward as they appear on the surface. And that, my friends, is something worth smiling about.

So next time you see the words "sodium acetate," don't just think "salt." Think "mildly basic," think "useful in buffers," think "a little bit of a chemist's quiet triumph." It’s a small thing, but sometimes, the small things are the most delightful. It’s not a loud, boisterous statement, but a gentle, knowing wink from the world of molecules.
And if anyone tells you it should be pH 7, you can just smile and say, "Ah, but the acetate ion likes to hug those water molecules just a little too much!" It’s a charming explanation for a charming property.
So there you have it. The pH of a 1.0 M sodium acetate solution. It’s not neutral. It’s a little bit basic. And that’s perfectly okay. In fact, I’d argue it's rather excellent.