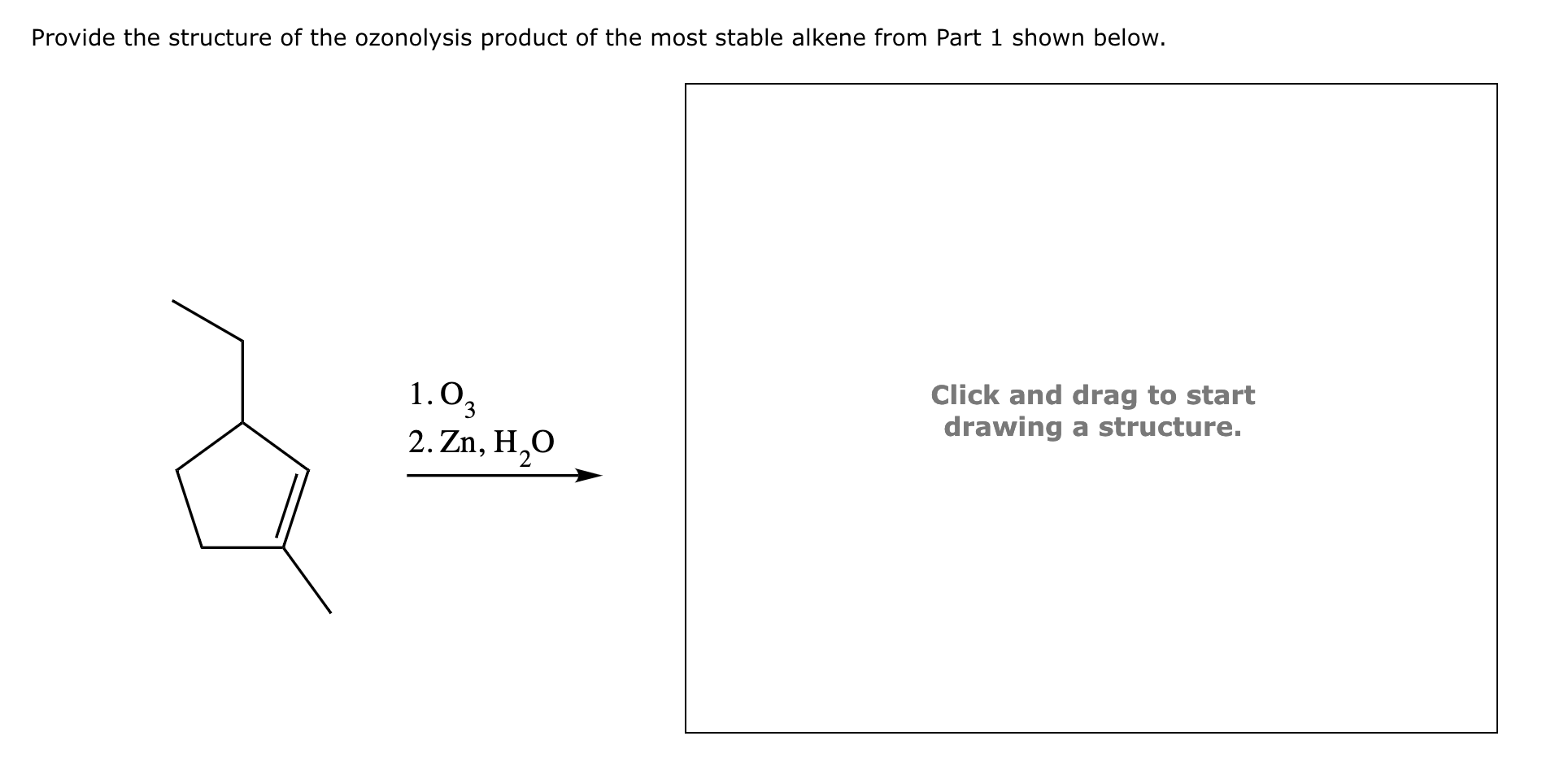
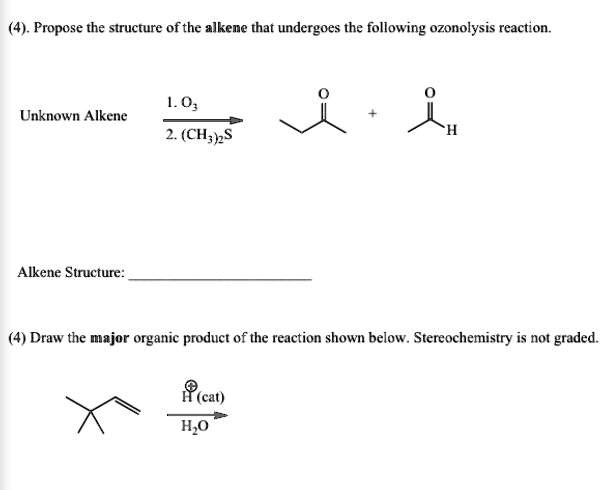
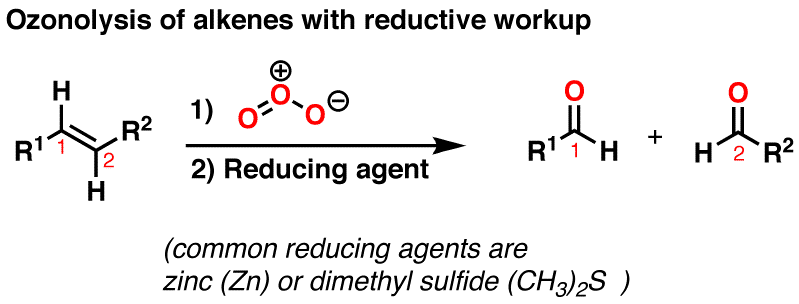
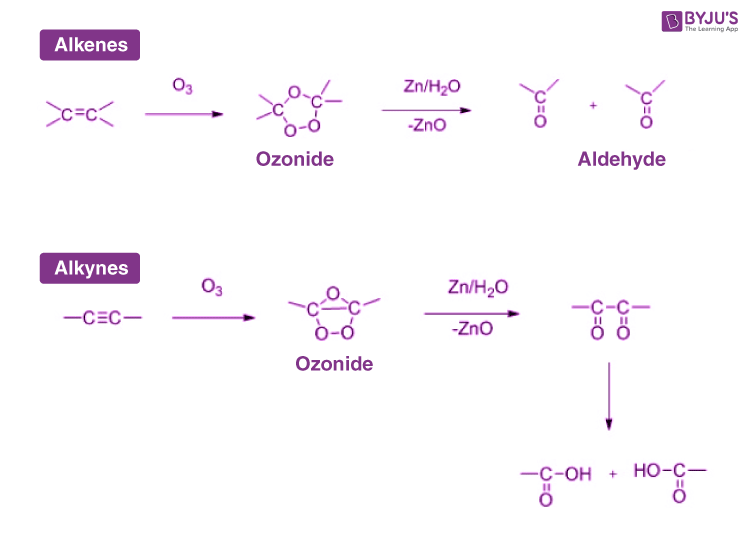
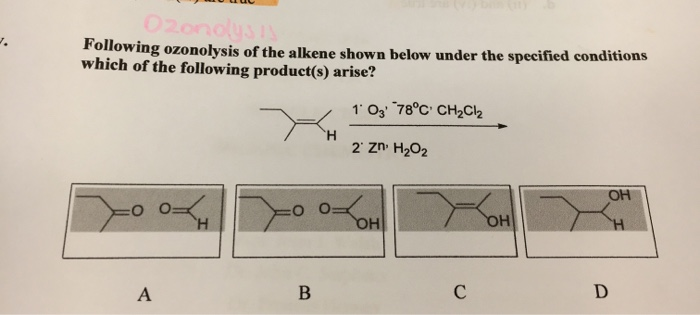
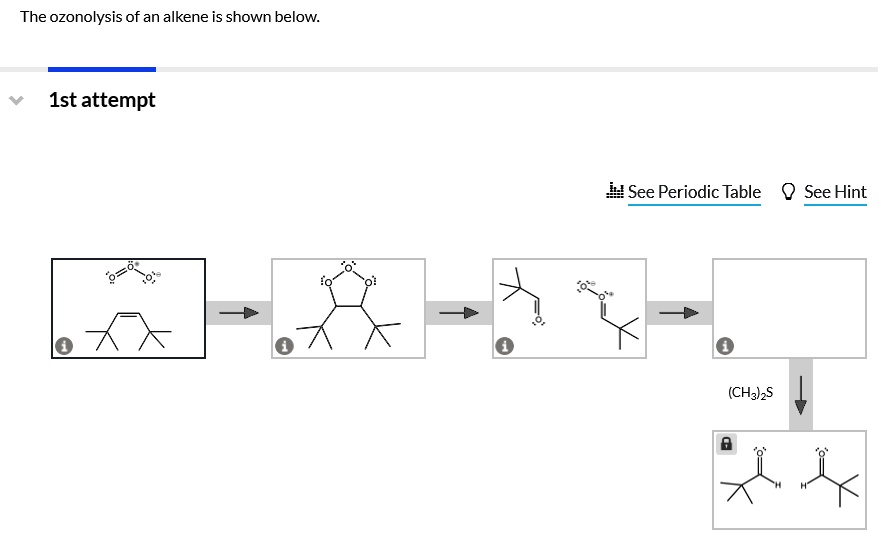
The Ozonolysis Of An Alkene Is Shown Below.
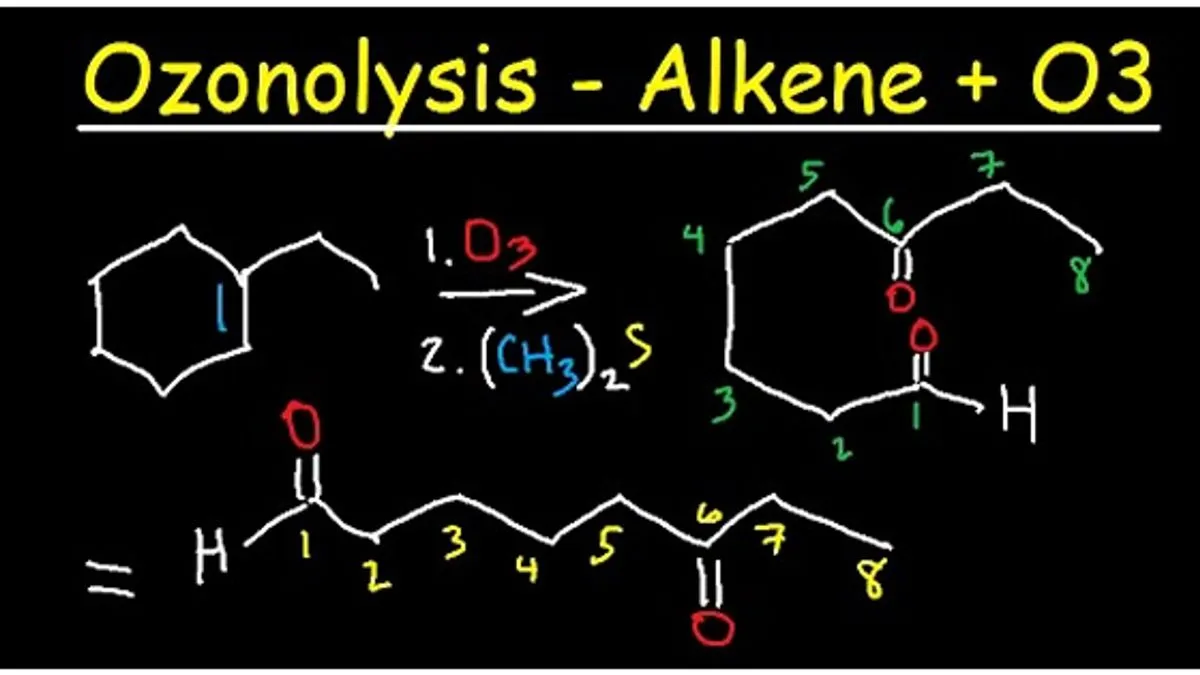
Hey there, science curious folks! Today, we’re going to dive into something that sounds super fancy – the ozonolysis of an alkene. Now, before your eyes glaze over and you start thinking about those tedious chemistry lectures you might have endured, let me assure you, this is actually pretty cool and, dare I say, even a little bit delicious. Think of it like this: it's a chemical party trick that helps us understand how things break down and what they turn into. And guess what? It happens all around us, influencing things from the air we breathe to the way our food gets made (or doesn't get made!).
So, what exactly is this "ozonolysis of an alkene"? Let's break it down. First, we have an alkene. In the simplest terms, an alkene is like a hydrocarbon molecule (think of the basic building blocks of oil and gas) that has a special double bond between two of its carbon atoms. Imagine two friends holding hands, but they’re holding hands really tightly, with two hands each. That double bond is like that super strong, extra-hand-holding connection. It’s a point of interest, a place where things can easily happen.
Now, enter ozone. You’ve probably heard of the ozone layer in our atmosphere, right? That’s our planet’s natural sunscreen, protecting us from harmful UV rays. But ozone (O3) is also a super-reactive molecule. It’s like that friend at the party who’s just bursting with energy, looking for something to do. When this energetic ozone molecule meets our handshake-holding alkene, sparks are about to fly!
Must Read
The term "ozonolysis" literally means "ozone splitting." And that’s exactly what our energetic ozone does to the alkene’s double bond. It’s like the ozone swoops in, sees that strong double bond, and says, "Hey, let's break this party up and see what’s inside!" It’s not a gentle separation; it’s a decisive cut, right through the middle of that double bond.
Imagine you have a really tightly tied ribbon – that’s your alkene's double bond. Ozonolysis is like taking a sharp pair of scissors and snipping that ribbon cleanly in two. And what do you get when you snip that ribbon? You get two shorter pieces! In chemistry terms, when the ozone breaks that double bond, it splits the alkene molecule into two smaller pieces. These smaller pieces are called carbonyl compounds. Think of them as the results of the ozonolysis party.
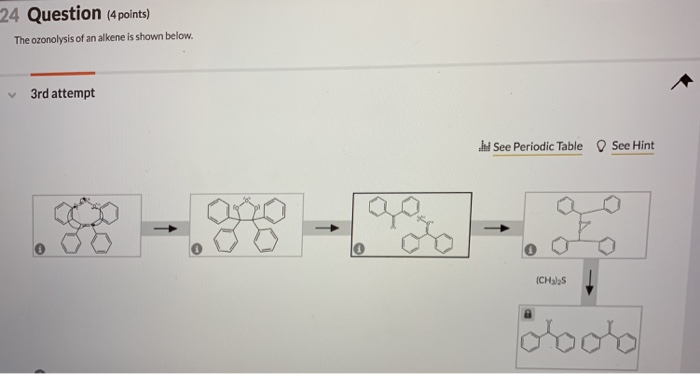
Now, why should you, dear everyday reader, care about this chemical maneuver? Well, it’s all about understanding how molecules behave and how we can manipulate them. This process is a powerful tool in chemistry, especially for organic chemists – the folks who study all the carbon-based molecules that make up life (and pretty much everything else around us!).
Think about it like this: Sometimes, you have a big molecule, and you need to break it down to understand its components, or maybe you need to create specific smaller molecules for a particular purpose. Ozonolysis is like a precision cutting tool for organic chemists. They can use it to take a large, complex alkene and chop it up into predictable, manageable pieces. It’s like having a really fancy recipe for dismantling a Lego castle to use the bricks for something else entirely.
One of the most important reasons we care about ozonolysis is its role in synthesizing new compounds. This means creating new molecules that can be used for all sorts of things. Pharmaceuticals are a huge area where this is vital. Many medicines are complex organic molecules, and chemists often build them piece by piece, or they might start with a larger molecule and cleverly break it down using reactions like ozonolysis to get the necessary building blocks.

Imagine you’re a chef trying to create a signature dish. You can't just throw everything into a pot and hope for the best. You need to chop your vegetables precisely, measure your spices, and understand how different ingredients interact. Ozonolysis is a bit like that precise chopping step for chemists. They can get exactly the aldehyde or ketone (types of carbonyl compounds) they need to build their “chemical dish” – whether it’s a new drug, a fragrance, or even a special kind of plastic.
Let’s get a little more specific. The type of carbonyl compound you get depends on the alkene you start with. If the double bond is at the end of the chain (like a molecule with a single hand holding on), you’ll get an aldehyde. Aldehydes are super common in nature. Think about the wonderful smells of vanilla (vanillin!) or cinnamon. Those lovely aromas often come from aldehydes!
If the double bond is in the middle of the chain, where both carbon atoms are holding onto two other carbon atoms, you’ll get a ketone. Ketones are also everywhere. Acetone, the stuff in nail polish remover, is a simple ketone. They’re also used as solvents and in the production of plastics.
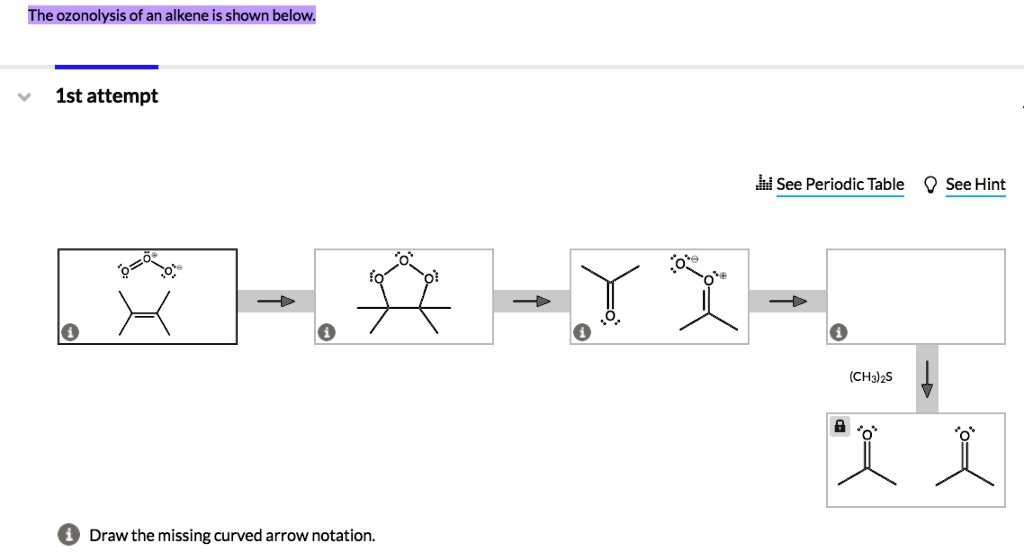
So, the next time you catch a whiff of a pleasant fragrance, or you’re using nail polish remover, or even just thinking about the complex molecules that make up your body, remember that a process like ozonolysis might have played a role in creating those things. It’s a fundamental reaction that helps us understand and build the chemical world around us.
It’s also important to note that ozonolysis isn’t just a lab experiment. Ozone itself is a natural component of the atmosphere, and while the stratospheric ozone layer is crucial for life, ground-level ozone can be a pollutant. When it interacts with organic molecules in the air – like those coming from car exhaust or industrial processes – it can cause them to break down. While this can sometimes be a good thing, breaking down harmful pollutants, it also highlights the reactive nature of ozone and how it can affect our environment.
Think of it like a tiny, energetic superhero molecule that can break bonds. In the upper atmosphere, it’s a hero protecting us. Near the ground, it’s a bit more of a wild card, capable of both good and bad depending on what it encounters. Ozonolysis is the story of that encounter – the moment the superhero meets its target and the transformation that follows.
Chemists have even developed ways to control ozonolysis to be very specific. They can choose which double bonds to break and get predictable results. This level of control is what makes it so valuable. It’s not just about random breaking; it’s about intentional, smart chemistry.
So, while the phrase "ozonolysis of an alkene" might sound intimidating, it's really just a way for us to understand how we can cleave (or cut) apart molecules, particularly those with double bonds, using ozone. It’s a fundamental reaction that underpins a lot of modern chemistry, from creating life-saving medicines to understanding environmental processes.
Next time you see a chemical equation or hear about a new material being developed, give a little nod to the humble alkene and its energetic friend, ozone. They might be quietly working behind the scenes, making the world a more interesting, and sometimes even a more fragrant, place. It’s a chemical story of transformation, and it’s happening all the time!