The Oxidation Number Of Nitrogen In Hno3 Is
Ever wondered about the hidden talents of everyday molecules? Today, we're diving into something that might sound a little scientific but is actually quite fascinating and surprisingly useful: the oxidation number of nitrogen in nitric acid (HNO₃). Think of it like a secret code that helps chemists understand how atoms behave and interact. It's a little puzzle that unlocks big chemical secrets, and once you get the hang of it, you'll see it everywhere!
So, what's the big deal with this oxidation number? Essentially, it tells us how many electrons an atom has gained or lost in a chemical compound. For nitrogen in nitric acid, this number is +5. This might seem like just another number, but for aspiring chemists, students, or even curious families doing science experiments at home, it's a key to understanding reactions, predicting how substances will behave, and even ensuring safety. For hobbyists who enjoy, say, gardening (nitric acid is a precursor to fertilizers!), understanding this helps appreciate the science behind what they do.
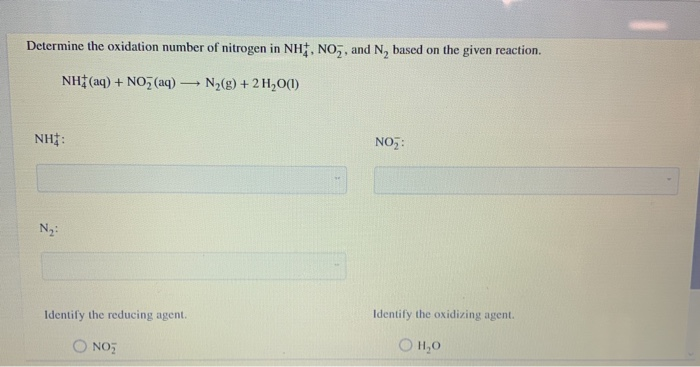
Let's break it down with an example. Think of a simple chemical reaction where nitric acid might be involved, like its reaction with a metal. Knowing nitrogen's oxidation state helps predict what new compounds will form and how much energy might be released. Variations come into play when nitrogen is in other compounds. For instance, in ammonia (NH₃), nitrogen has an oxidation number of -3. This difference in oxidation states shows how nitrogen can be quite versatile, acting as both an electron giver and receiver!
Must Read
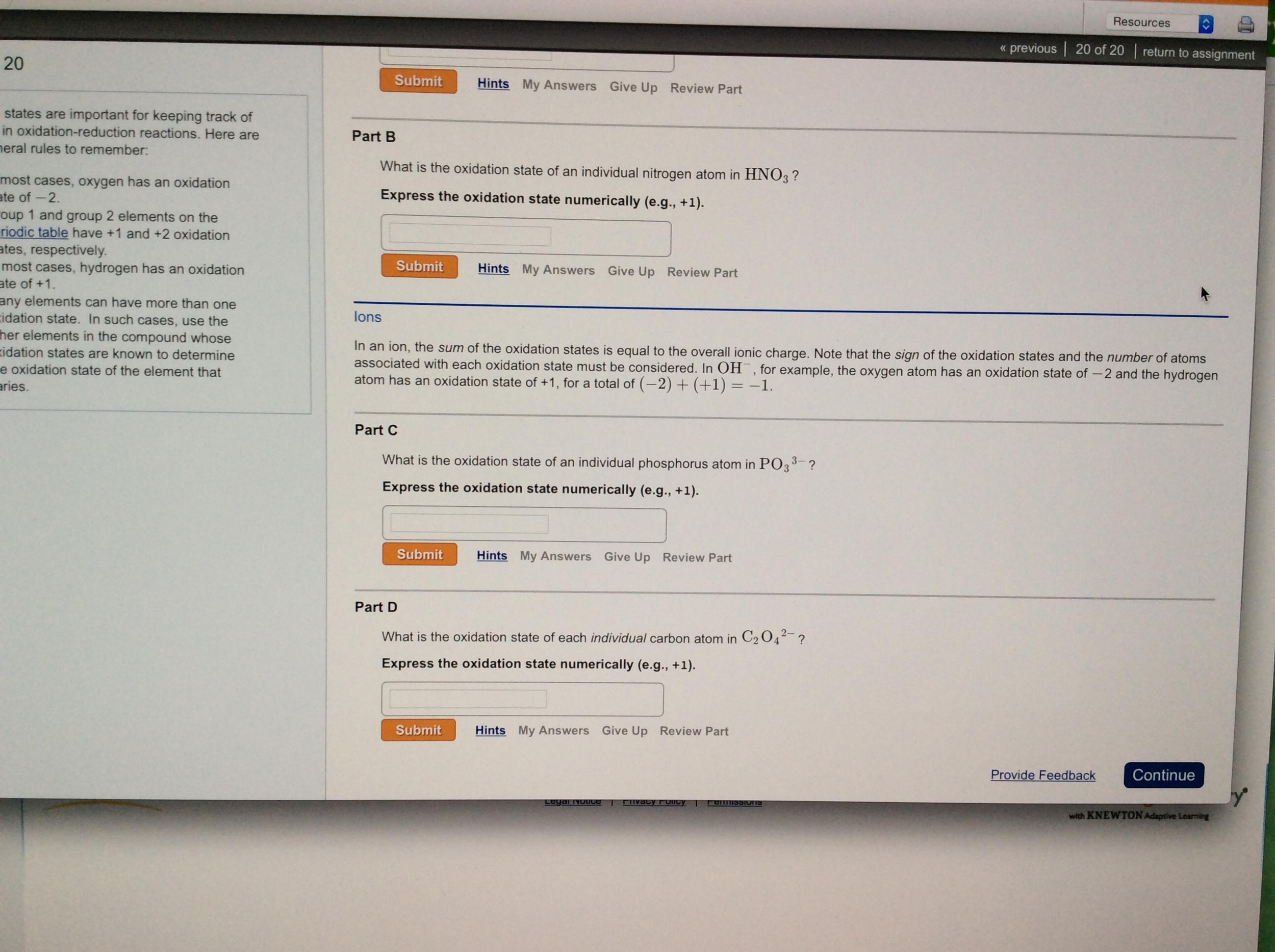
Getting started with understanding oxidation numbers is easier than you think. For beginners, focus on the basic rules: oxygen is usually -2 (except in peroxides), hydrogen is usually +1 (except in metal hydrides), and the sum of oxidation numbers in a neutral molecule is always zero. For HNO₃, you'd set it up like this: (+1 for H) + (N for nitrogen) + (3 * -2 for O) = 0. Solving for N, you get N = +5. Simple, right? You can even practice with common compounds you find around the house, like water (H₂O) or table salt (NaCl).
The beauty of learning about oxidation numbers is that it opens up a new way of looking at the world. It's like learning a secret language that chemicals speak. Whether you're a student tackling chemistry homework, a parent looking for engaging ways to teach kids about science, or a hobbyist wanting a deeper understanding of your craft, the oxidation number of nitrogen in HNO₃ and other compounds is a fundamental building block. It's a small piece of knowledge that leads to a much larger appreciation for the incredible world of chemistry. It’s not just a number; it’s a story waiting to be told!



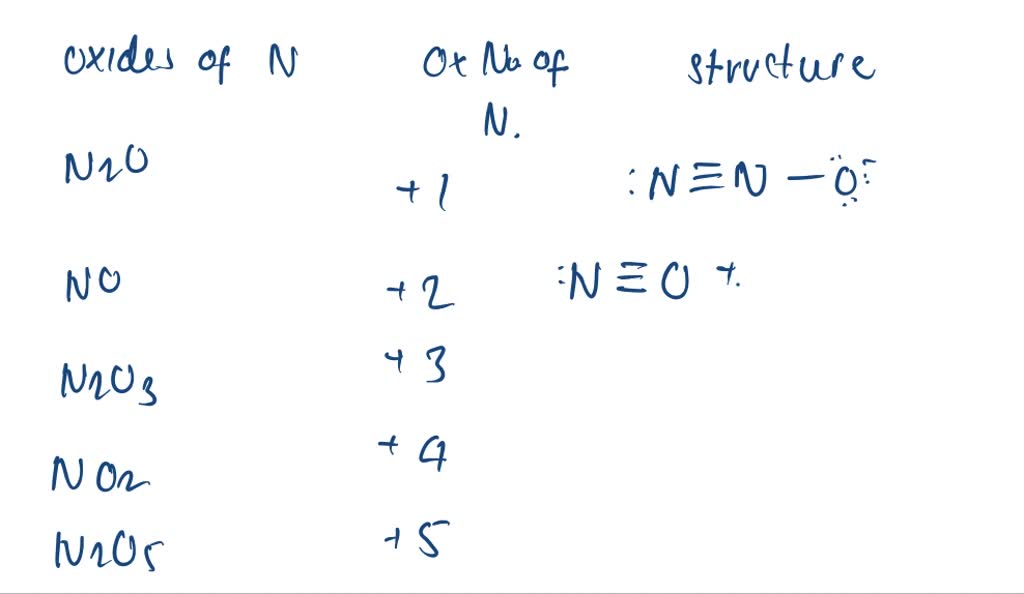



![[GET ANSWER] 4. The oxidation numbers of nitrogen in NH3, HNO3, and NO2](https://cdn.numerade.com/ask_images/70671db89b6240d9ad03fe1482362c30.jpg)