The Number Of Unpaired Electrons In Fluorine

Ever wondered what makes some atoms click and bond while others are a bit more… solitary? It all boils down to a fascinating aspect of atomic personality: the number of unpaired electrons. While it might sound like a niche topic for hardcore science buffs, understanding this concept, especially as it applies to a famously feisty element like fluorine, unlocks a whole new appreciation for the invisible dance happening all around us. Think of it as the atomic equivalent of a dating scene – who’s looking for a partner and who’s perfectly happy flying solo? And when it comes to fluorine, its unpaired electron isn't just a detail; it's the key to its incredible reactivity and its widespread influence on everything from toothpaste to advanced materials.
The Magnetic Personality of Atoms
Atoms, the building blocks of everything, are governed by a set of rules that dictate their behavior. A huge part of this behavior is determined by their electrons, specifically the ones in their outermost shell, also known as valence electrons. These are the electrons that get to mingle and interact with other atoms. Now, electrons don't just float around randomly; they love to pair up. They generally come in pairs, spinning in opposite directions within specific energy levels called orbitals. Think of orbitals as little rooms in an atom's house, and electrons as tiny dancers. Most of the time, these dancers are happily paired up, each spinning a different way. However, sometimes, an orbital can end up with just one dancer. This is where the magic – and the potential for some serious action – happens!
An unpaired electron is like a dancer who’s still looking for a partner. It’s a bit unsettled, a bit eager, and incredibly ready to interact. This unpaired electron creates an imbalance, and atoms with unpaired electrons are often highly reactive. They actively seek out other atoms that can provide them with a partner, completing their outer shell and achieving a more stable, contented state. This drive to pair up is the fundamental force behind chemical bonding, the process that creates molecules and, ultimately, all the diverse substances we encounter in our world.
Must Read
Fluorine: The Superstar of Reactivity
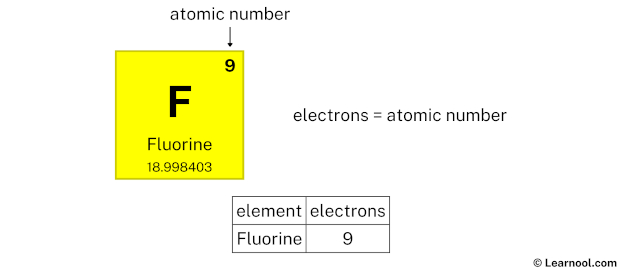
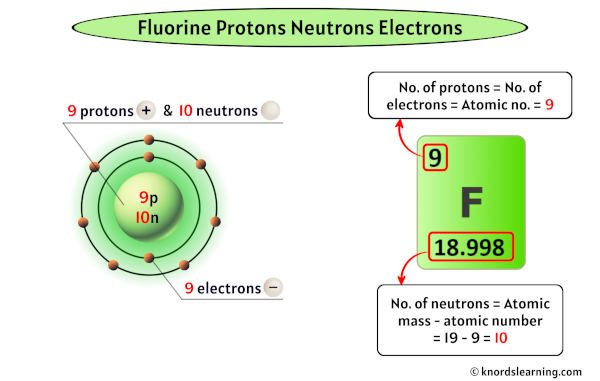
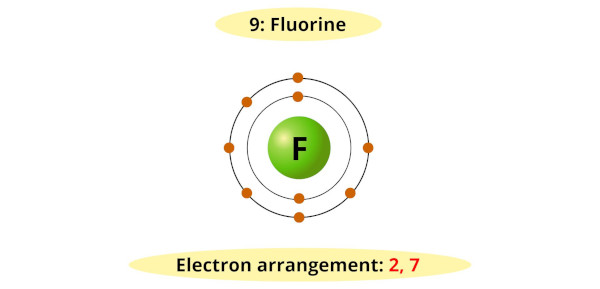
Now, let’s zoom in on fluorine (F). If atoms had personalities, fluorine would be the energetic, sometimes intimidating, but ultimately incredibly useful one. It’s the most electronegative element on the periodic table, meaning it has an almost insatiable appetite for electrons. This intense desire stems directly from its electronic configuration. A neutral fluorine atom has 9 electrons in total. These electrons are arranged in specific energy levels or shells. The innermost shell has 2 electrons, and the next shell, the outermost or valence shell, has 7 electrons.
Here’s where the crucial detail comes in: the outermost shell of an atom can hold a maximum of 8 electrons. Fluorine is just one electron away from achieving this stable, full configuration. This means that out of the 7 valence electrons in fluorine, 6 of them are paired up, happily spinning together. But there’s one electron left over, all by itself, in an orbital. This is the one unpaired electron in fluorine. This single, unpaired electron is the driving force behind fluorine’s extreme reactivity. It’s constantly on the lookout for an atom that can donate an electron, or one with which it can share an electron to complete its outer shell.

Why Does This Matter?
So, why should we care about fluorine’s single unpaired electron? Because it makes fluorine incredibly powerful and versatile. When fluorine encounters almost any other element, its unpaired electron immediately goes to work, forming a strong chemical bond. This is why fluorine is used in so many important applications:
- Toothpaste: Fluoride compounds, which contain fluorine, are added to toothpaste to strengthen tooth enamel and prevent cavities. The fluorine effectively bonds with the calcium in our teeth, making them more resistant to acid attacks.
- Non-stick Cookware: The famous non-stick coatings on our frying pans, like Teflon, are made of fluorocarbons. These are molecules where fluorine atoms are bonded to carbon atoms. The strong bonds created by fluorine make these materials incredibly stable and resistant to heat and chemicals.
- Refrigerants and Propellants: Certain fluorine compounds have been used as refrigerants and in aerosol cans, although their environmental impact is now carefully managed.
- Pharmaceuticals: The unique properties that fluorine imparts to molecules are incredibly useful in designing drugs. Many modern medicines contain fluorine atoms to improve their effectiveness, stability, and how they are absorbed by the body.
- Advanced Materials: From high-performance plastics to components in batteries and semiconductors, fluorine plays a vital role in creating materials with specialized properties.
Essentially, that single unpaired electron in fluorine is the key that unlocks a vast array of chemical reactions and leads to the creation of incredibly useful materials and technologies. It’s a perfect example of how a seemingly small detail at the atomic level can have enormous implications for our daily lives. So, the next time you brush your teeth or use a non-stick pan, remember the powerful role of fluorine and its solitary, electron, working tirelessly to make our world a little bit better, one bond at a time!