The Molecular Mass Of Sodium Oxide Na2o Is

Okay, so let’s talk about something that sounds way more intimidating than it actually is: the molecular mass of sodium oxide, or as us science nerds affectionately call it, Na₂O. Now, before your eyes glaze over like a freshly baked donut, imagine this. You’re at a potluck, right? Everyone brings their signature dish. There’s Aunt Carol’s legendary casserole, your cousin’s suspiciously neon-colored Jell-O mold, and then there’s… well, sodium oxide. It’s not exactly the life of the party, but it’s got its own special role, and understanding its "weight" is kind of like knowing how much of Aunt Carol’s casserole you can realistically fit on your plate without regretting it later.
Think of atoms as tiny, invisible LEGO bricks. Each element, like sodium (Na) and oxygen (O), has its own specific type of LEGO brick with a particular weight. When these bricks stick together to form a compound – like Na₂O – their combined weight is what we’re talking about. It's like figuring out the total weight of a LEGO spaceship you’ve just built. You don't just guess; you’d probably weigh each brick type and then do some quick math. Easy peasy, right?
So, what are these LEGO bricks made of? Sodium (Na) is a pretty reactive fellow. It’s that element that, if you leave it out in the open air, it’ll do a little dance with the oxygen and moisture and get all… well, gunky. Not exactly the kind of thing you’d want to hug. And then there’s oxygen (O), which is a bit of a drama queen. It’s everywhere, it’s essential for life (thanks, O!), and it loves to bond with other elements. In sodium oxide, they've decided to get hitched.
Must Read
Now, the "molecular mass" part. It’s basically the grand total of all the atomic masses of the atoms in a molecule. For Na₂O, we’ve got two sodium atoms and one oxygen atom. Each of these has its own atomic mass, which is usually measured in something called atomic mass units (amu). Don't get bogged down in the acronym; just think of it as the "weight" of that specific LEGO brick. It’s like knowing that a standard LEGO brick weighs 2 grams and a smaller one weighs 1 gram. You add them up for the total.
Let’s get to the nitty-gritty, but in a chill way. The average atomic mass of sodium (Na) is about 22.99 amu. So, for our two sodium atoms in Na₂O, we have 2 * 22.99 = 45.98 amu. See? We’re already halfway there, and it wasn't even painful. It's like realizing you only have two more chores to do before you can kick back with a cup of tea.
Now, for our friend oxygen (O). Its average atomic mass is about 16.00 amu. Easy, right? Just one of those oxygen LEGO bricks. So, the total for our oxygen is 1 * 16.00 = 16.00 amu. Nothing to lose sleep over here, folks.
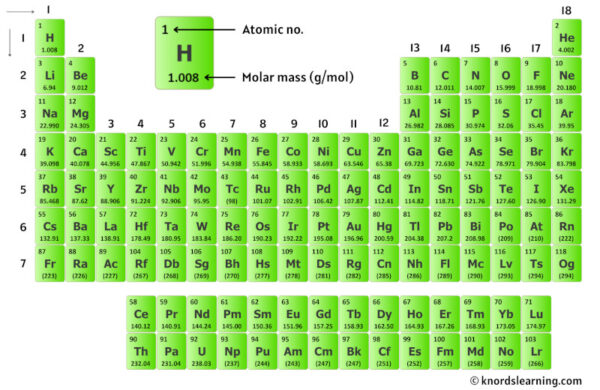
To find the molecular mass of Na₂O, we simply add the weights of all the atoms together. Drumroll, please! 45.98 amu (from the two sodiums) + 16.00 amu (from the one oxygen) = 61.98 amu. Ta-da! The molecular mass of sodium oxide is approximately 61.98 amu. You did it! You’ve conquered a bit of chemistry without breaking a sweat. It’s like finding that last missing sock that’s been hiding in the dryer for weeks – a small victory, but a victory nonetheless!
Now, why would anyone care about the molecular mass of sodium oxide? Well, in the grand scheme of things, it's super useful. Imagine you're a baker, and you need to make a specific recipe for, say, really fluffy pancakes. The recipe calls for a certain amount of baking powder, and you need to know how much of each ingredient is actually in that baking powder. That's where understanding molecular mass comes in. It helps chemists and scientists figure out how much of a substance they're dealing with, how it might react, and what its properties will be.
Think of it like this: if you’re trying to build a magnificent sandcastle, you need to know how much sand you’re hauling. If you’ve got 10 buckets of sand, and each bucket is pretty hefty, you know you’re in for some serious sculpting. Similarly, knowing the molecular mass of Na₂O tells us its "heft" in the chemical world. This is crucial when you're trying to mix chemicals in just the right proportions, like trying to get the perfect ratio of flour to sugar for those amazing cookies your grandma makes.

Sodium oxide itself isn't something you're likely to encounter casually on your grocery run, unless you’re a very niche collector of chemical compounds. It’s a white solid that’s highly reactive and usually found in industrial settings or in the lab. It’s kind of like that exotic spice you see in a specialty store – you know it exists, you know it has a purpose, but you’re not going to sprinkle it on your scrambled eggs.
But the principles behind calculating its molecular mass are fundamental. It’s the same way we’d calculate the weight of water (H₂O), or sugar (C₁₂H₂₂O₁₁), or even the stuff that makes your toothpaste fizz. The process is always the same: find the atomic masses of each element, multiply by how many of that atom are in the molecule, and then add them all up. It’s a universal formula for figuring out the weight of molecules.
It’s also a bit like understanding how much each ingredient contributes to the final flavor of a dish. If you add too much of a strong spice, the whole thing can go south. In chemistry, if you don’t have the right amount of a reactant (like sodium oxide), your reaction might not happen, or it might produce something unexpected. The molecular mass is the first step in understanding those quantities.
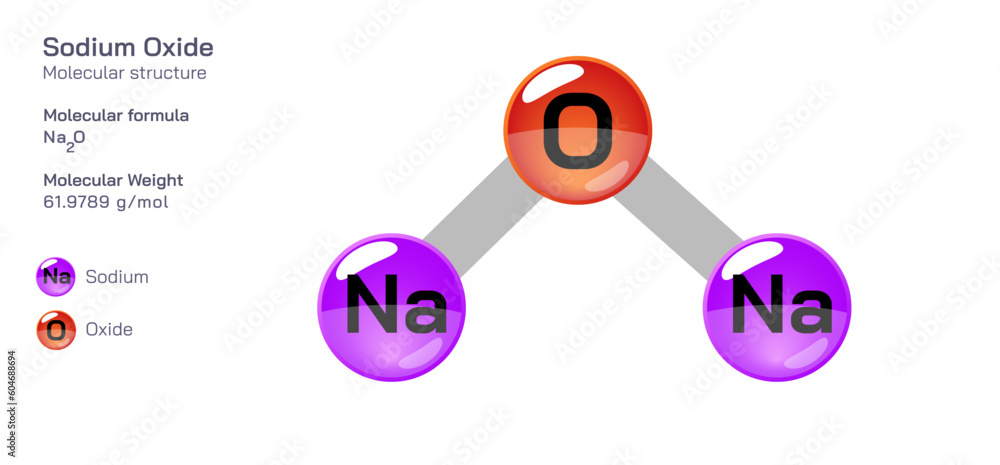
Let’s put it another way. Imagine you’re a detective trying to solve a case. You’ve got clues, right? The atomic mass of each element is like a clue about its identity and how much it contributes to the bigger picture. When you piece all the clues together – the atomic masses of sodium and oxygen, and how many of each are present – you get the full story of the molecular mass of sodium oxide. It’s a solved mystery!
Sometimes, you’ll see the molecular mass expressed in grams per mole (g/mol). This sounds a bit fancier, but it’s essentially the same idea. If you had a mole of sodium oxide molecules (a mole is just a huge number of particles, like a baker's dozen for atoms), it would weigh about 61.98 grams. It's like saying a dozen eggs weighs about 1.5 pounds. You’re relating the count of items to their weight. For chemists, a mole is a convenient way to talk about large numbers of atoms and molecules that we can actually measure in a lab.
So, the next time you hear someone mention the molecular mass of sodium oxide, you can nod knowingly. You understand it’s not some arcane secret, but a straightforward calculation based on the building blocks of matter. It’s the chemical equivalent of knowing that a bag of flour weighs 5 pounds and a bag of sugar weighs 4 pounds, and if you need a recipe that calls for two bags of flour and one bag of sugar, you know you’re looking at a total of 14 pounds of ingredients. It's practical, it's fundamental, and it’s a little piece of the amazing puzzle that is the universe.

And honestly, who doesn’t love a good puzzle? Even if it involves things as tiny as atoms and as seemingly dry as molecular masses. It’s the foundation for so much of the technology and products we use every day, from the medicines that keep us healthy to the materials that build our homes. So, a little bit of chemistry knowledge goes a long way, even if it’s just understanding the weight of a compound like Na₂O. It’s like learning a few words of a new language – it opens up a whole new world of understanding. And who knows, maybe one day you’ll be mixing up your own chemical concoctions, all thanks to knowing that sodium oxide weighs about 61.98 amu!
It's also kind of funny to think about how we break things down. We take something like sodium oxide, and we don't just accept it as a thing. We dissect it into its parts, figure out the weight of those parts, and then put it all back together to understand the whole. It’s like taking apart a watch to see how the gears work, but instead of gears, we have atoms. And the result of that dissection for Na₂O is that satisfying number: 61.98 amu. It's the chemical equivalent of saying, "Yep, that’s about right," after you've weighed something on a scale. A tangible, albeit microscopic, weight.
So, there you have it. The molecular mass of sodium oxide, Na₂O. It’s not a dragon to be slain, but a simple calculation that helps us understand the world around us. It’s the weight of two sodium atoms and one oxygen atom, coming together to form this particular compound. And just like knowing how much your groceries weigh helps you plan your meals, knowing the molecular mass of Na₂O helps chemists plan their reactions. It’s all about understanding the ingredients, one atom at a time.
Remember, science isn't always about complicated equations and intimidating jargon. Sometimes, it's just about understanding the basic building blocks and how they fit together. And in the case of sodium oxide, those building blocks give us a molecular mass of approximately 61.98 amu. So next time you see “Na₂O” written down, you can give a little wink, knowing that you understand its fundamental weight. It's a small victory, but in the vast universe of chemistry, every understanding is a win!





