The Lowest Allowable Energy State Of An Atom

So, there I was, fiddling with an old chemistry textbook the other day, the kind with that delightful musty smell that only ancient paper can produce. I stumbled upon a diagram of an atom, you know, the classic Bohr model – little electrons whizzing around a nucleus like tiny planets. And it got me thinking, really thinking, about what keeps those little guys in their orbits. It’s not like they’re tethered by invisible strings, right? There must be a fundamental reason, a cosmic set of rules, dictating their every move. It’s this fundamental quest for stability, for the least amount of fuss, that leads us to a rather fascinating concept: the lowest allowable energy state of an atom.
Think about it. If you’re anything like me, you probably remember that feeling of absolute exhaustion after a particularly grueling day. You just want to curl up on the couch, put your feet up, and do… well, nothing. You’re seeking your own personal lowest energy state, aren’t you? You don’t want to be running a marathon or doing complex calculus; you want peace, quiet, and minimal exertion. Atoms, in their own tiny, quantum-mechanical way, are surprisingly similar. They too, have a preferred state of being, a kind of atomic chill-out zone.
The Atomic "Zen" Mode
This "lowest allowable energy state" is more formally known as the ground state. It’s the most stable configuration an atom can achieve. Imagine a ball at the bottom of a valley. It’s not going anywhere unless you give it a serious shove. That’s the ground state for an atom. It's where the electrons are arranged in the most energetically favorable way possible.
Must Read
Why is it so special? Well, everything in the universe, at its core, tends towards stability. Think about it: a perfectly balanced structure is less likely to topple than a precariously stacked pile of rocks. For atoms, this stability comes from their electrons finding the lowest possible energy levels. It’s like finding the comfiest chair in the house – you’re not going to voluntarily get up and stand, are you?
Now, these energy levels aren’t just arbitrary numbers. They’re dictated by the laws of quantum mechanics, which, let’s be honest, can feel like trying to understand a dream. Unlike the classical physics we’re used to – where things can have any speed or any position – quantum mechanics says that energy for electrons in atoms is quantized. This means it can only exist in discrete, specific amounts, like steps on a ladder, not a smooth ramp.
So, an electron can be on the first step, or the second, or the third, but it can’t be hovering halfway between steps. And the lowest step on that ladder? That’s the ground state. It’s the most energetically economical position for that electron to be in. It’s where it feels most at home, most… content.
The Electron Dance: Not Just Random Whizzing
The Bohr model, bless its simplicity, gets the idea across. Electrons orbit the nucleus. But the reality is a bit more nuanced, and a lot more mind-boggling. Electrons don’t really "orbit" in a predictable path like planets. Instead, they exist in what we call orbitals. These are regions of space around the nucleus where there’s a high probability of finding an electron.
Think of orbitals like fuzzy clouds. The denser the cloud, the more likely you are to find the electron there. And these orbitals have different shapes and sizes, each corresponding to a specific energy level. The lowest energy orbitals are generally closer to the nucleus and have simpler shapes, like spheres.

As you move to higher energy levels, the orbitals get bigger, more complex, and further away from the nucleus. It's like having different rooms in a house, each with a different "cost" to occupy. The ground floor is the cheapest, the most accessible, and therefore, where you’d want to be if you were an energy-conscious electron.
So, when we talk about the ground state of an atom, we’re essentially talking about how all its electrons are arranged in the lowest available energy orbitals. It’s the atom’s most fundamental, most stable configuration.
What Happens When Atoms Get Excited?
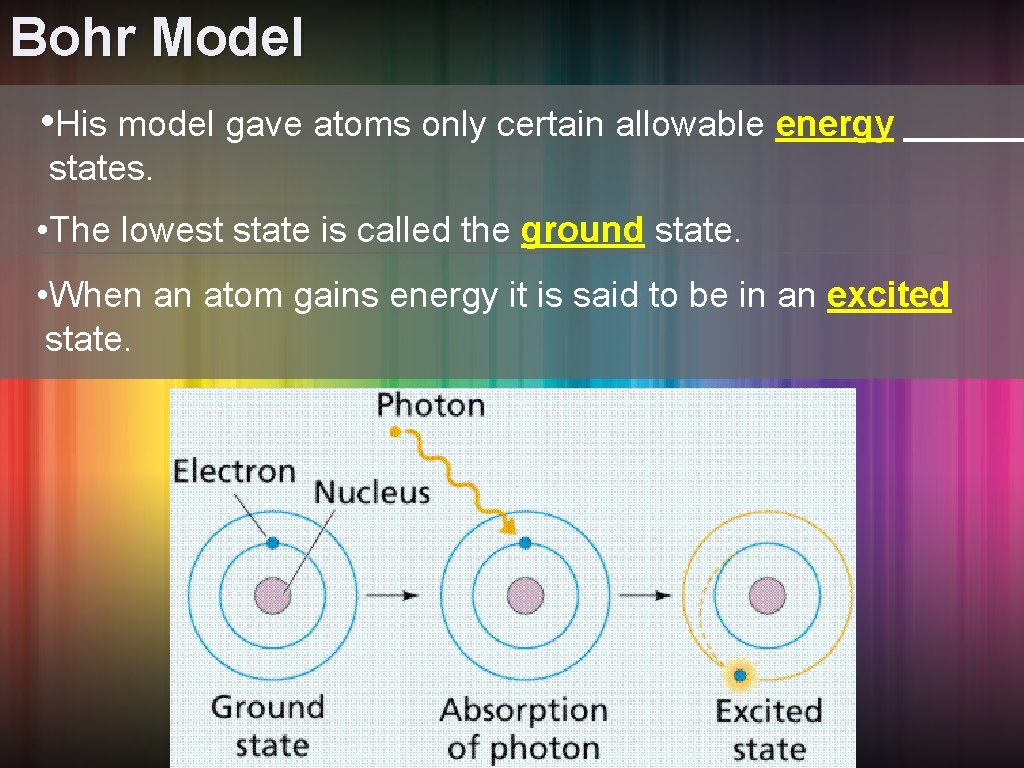
If the ground state is the atomic equivalent of a long, luxurious nap, then what happens when an atom isn't in its ground state? It’s in an excited state. This happens when an electron absorbs energy from an external source, like a photon of light or heat. Imagine giving that ball at the bottom of the valley a good kick. It's going to roll up the side, right?
When an electron absorbs energy, it jumps to a higher energy orbital – a step higher on that quantum ladder. This makes the atom unstable. Think of it as being all wired up, full of nervous energy. It doesn't want to stay in this excited state for long. It’s like holding a baby bird: it’s cute, but you know it’s going to want to fly away eventually.
And just like that baby bird wants to return to its nest, the electron wants to return to its ground state. To do this, it has to release the excess energy it absorbed. How does it do that? By emitting a photon of light!
.PNG)
This is where things get really cool and frankly, a little bit magical. The color of the light emitted depends on the exact amount of energy the electron releases. Different energy jumps correspond to different photon energies, and different photon energies mean different colors of light. This is the fundamental principle behind things like neon signs and fireworks.
When you see a vibrant red neon sign, you’re seeing the result of excited electrons in neon atoms returning to their ground state. The specific energy difference between the excited state and the ground state in neon atoms happens to correspond to the energy of red light photons. It’s like each element has its own unique "fingerprint" of light it can emit when it’s excited. Pretty neat, huh?
The Building Blocks of Light Shows
So, the next time you see a dazzling display of fireworks, remember that you're witnessing countless atoms being kicked into excited states and then, in their rush back to equilibrium, painting the sky with light. It’s a grand, energetic ballet of electrons, all governed by the simple desire to be in their lowest, most comfortable state.
This whole idea of ground states and excited states is absolutely crucial to understanding so much of chemistry and physics. It explains why elements behave the way they do, how they interact, and how they produce light. It’s the bedrock of spectroscopy, a powerful tool scientists use to identify elements and study their properties by analyzing the light they absorb or emit.
The "No Free Lunch" Rule of Atoms
There's a very important implication of the ground state: atoms can't have just any amount of energy. They're not like a car with an infinitely variable transmission. They have these specific energy levels, and the lowest one is the ground state. This means you can't just "chill out" an electron to an energy lower than its ground state. It's like trying to dig a hole deeper than the bottom of the Earth – it’s physically impossible within the rules of the system.

This principle is sometimes referred to as the "no free lunch" rule for atoms. They have to abide by the energy economy. To get to a higher energy state, they must absorb energy. To return to the ground state, they must release energy. They can't just magically lower their energy without consequence.
And this is why, for instance, when you heat up a piece of metal, it eventually starts to glow. The heat energy is absorbed by the atoms, exciting their electrons. As these electrons fall back down to their ground states, they emit photons of light. The hotter it gets, the more energy is absorbed, the higher the energy of the emitted photons, and the whiter the light appears (because you're getting a mix of all visible colors). It's a direct consequence of atoms seeking their lowest energy state.
The Ever-Present Quest for Minimum Energy
It’s fascinating to consider how this fundamental principle of seeking the lowest energy state applies not just to individual atoms, but to larger systems too. Molecules form because the bonded atoms are in a lower energy state than the individual atoms were. Chemical reactions often proceed in a direction that leads to more stable (lower energy) products.
It’s a universal drive, this seeking of minimum energy. From the smallest subatomic particles to the grandest cosmic structures, there’s an inherent tendency towards stability, towards the most energetically favorable arrangement. The ground state of an atom is just one of the most fundamental and beautifully elegant manifestations of this universal principle.
The "Forbidden" States
Because energy is quantized, there are also states that atoms cannot exist in. These are the energies between the allowed energy levels. You can think of it like trying to stand on a staircase where there are gaps between the steps. You can be on step 1, or step 2, but you can't be floating in mid-air between them. These "forbidden" energies are simply not permissible for the atom's electrons.

This is a key differentiator between classical and quantum mechanics. In the classical world, you could imagine a ball rolling at any speed, possessing any amount of kinetic energy. But in the quantum world of atoms, it's much more restrictive. The electrons are constrained to specific energy levels.
When an atom absorbs energy, it’s not like slowly filling up a tank. It’s more like clicking a button that instantly teleports an electron to a higher, allowed energy level. And when it emits energy, it's like clicking another button that teleports it back down to a lower, allowed level, releasing the difference as a photon.
It's All About The Rules, Man
These rules, these specific energy levels, are determined by the atom's structure – the number of protons in the nucleus and the number and arrangement of electrons. This is why different elements have different ground states and different sets of allowed energy levels. It's why a helium atom behaves differently from an oxygen atom. Their internal "rules" are unique.
Understanding the lowest allowable energy state of an atom isn’t just about memorizing textbook definitions. It’s about appreciating the fundamental nature of matter, the elegance of quantum mechanics, and the universal drive towards stability that shapes our entire universe. It’s a concept that, once you grasp it, opens up a whole new way of seeing the world around you, from the simplest glow of a light bulb to the complex chemistry of life itself.
So, the next time you feel that irresistible urge to just… be… and find your own lowest energy state, spare a thought for the atoms. They’re doing the same thing, just on a much, much smaller scale, and with a lot more quantum weirdness involved. And honestly? It’s kind of comforting to know we’re all just seeking a bit of atomic peace and quiet.