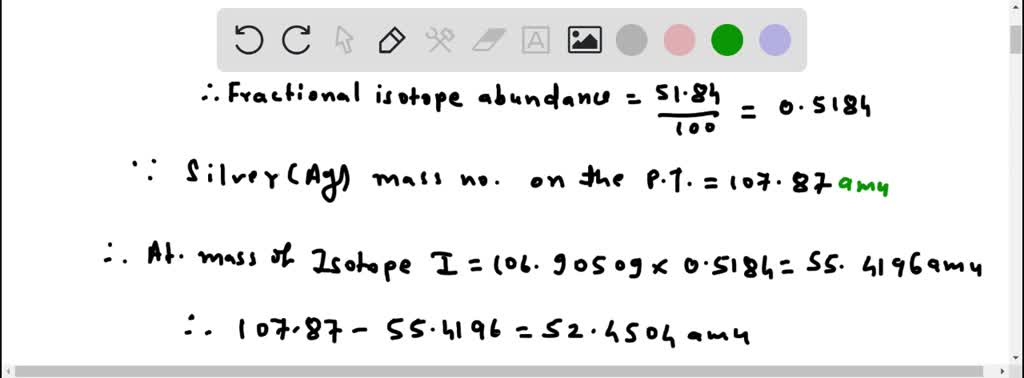
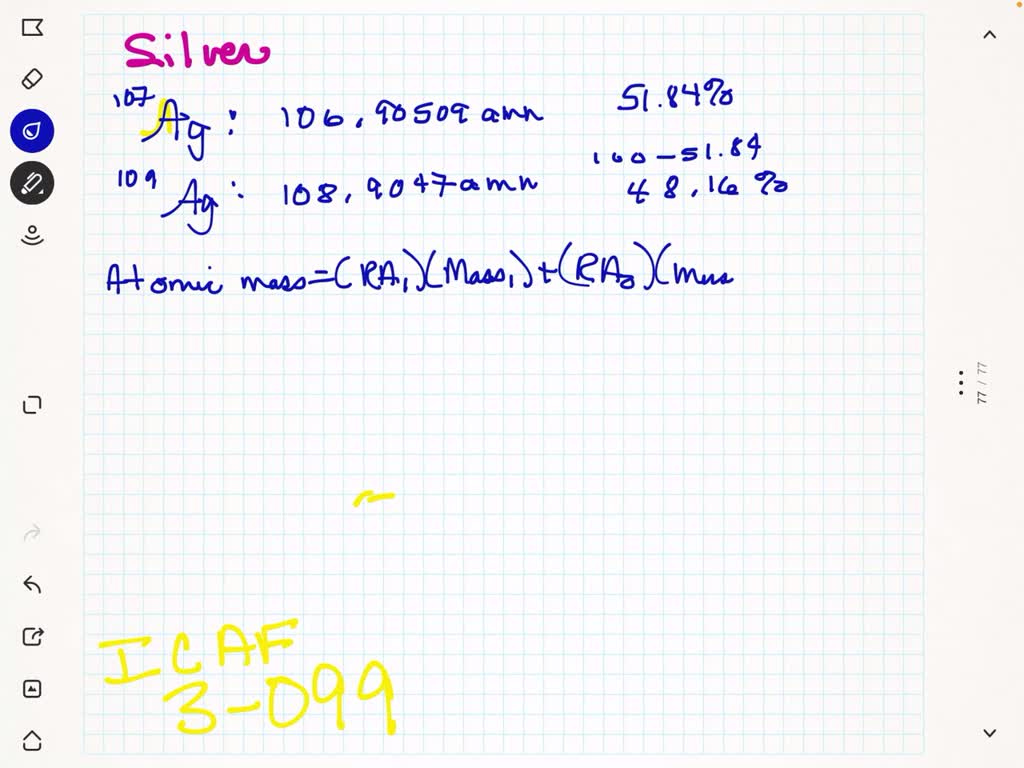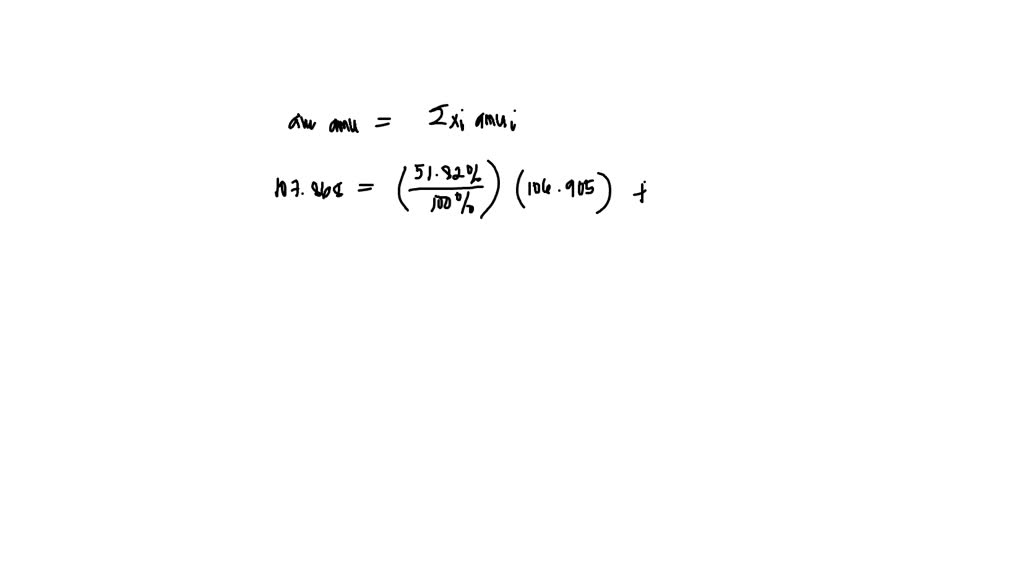Silver Has Two Naturally Occurring Isotopes
Hey there, curious cats and science enthusiasts! Ever look at that shiny silver in your jewelry or your fancy cutlery and think, "Wow, that's pretty!"? Of course you do! Silver is undeniably cool, isn't it? But what if I told you that the silver you're admiring isn't just one, single thing? It's actually a bit of a duo, a dynamic pair working together behind the scenes. Mind. Blown. Let's dive into the wonderfully weird world of silver's naturally occurring isotopes and discover why this little scientific fact can actually make life a whole lot more interesting (and maybe even a little sparkly!).
So, what exactly are these "isotopes" we're talking about? Think of them like siblings. They're essentially the same element, sharing the same core identity (in silver's case, that's 47 protons – a super important number!). But, like brothers and sisters, they have a little something extra that makes them unique. In the case of isotopes, that "something extra" is usually a different number of neutrons in their atomic nucleus. Neutrons are like the silent, heavy lifters in the atomic world, hanging out with the protons. Adding or subtracting them changes the atom's mass, but not its fundamental chemical personality. Pretty neat, right?
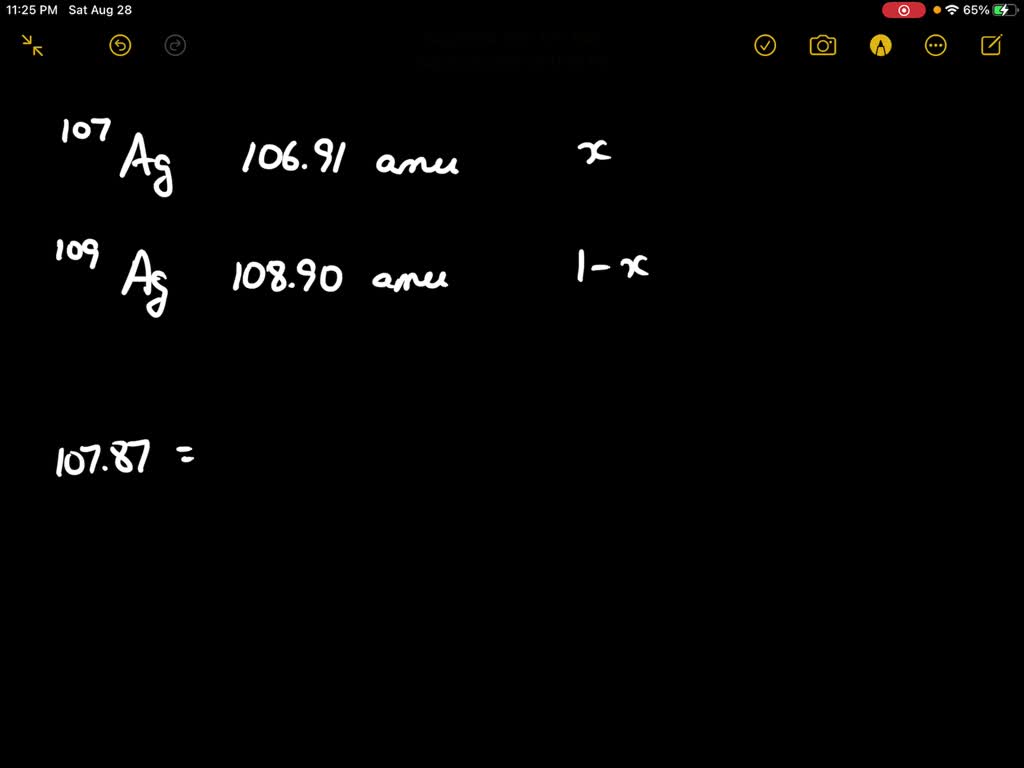
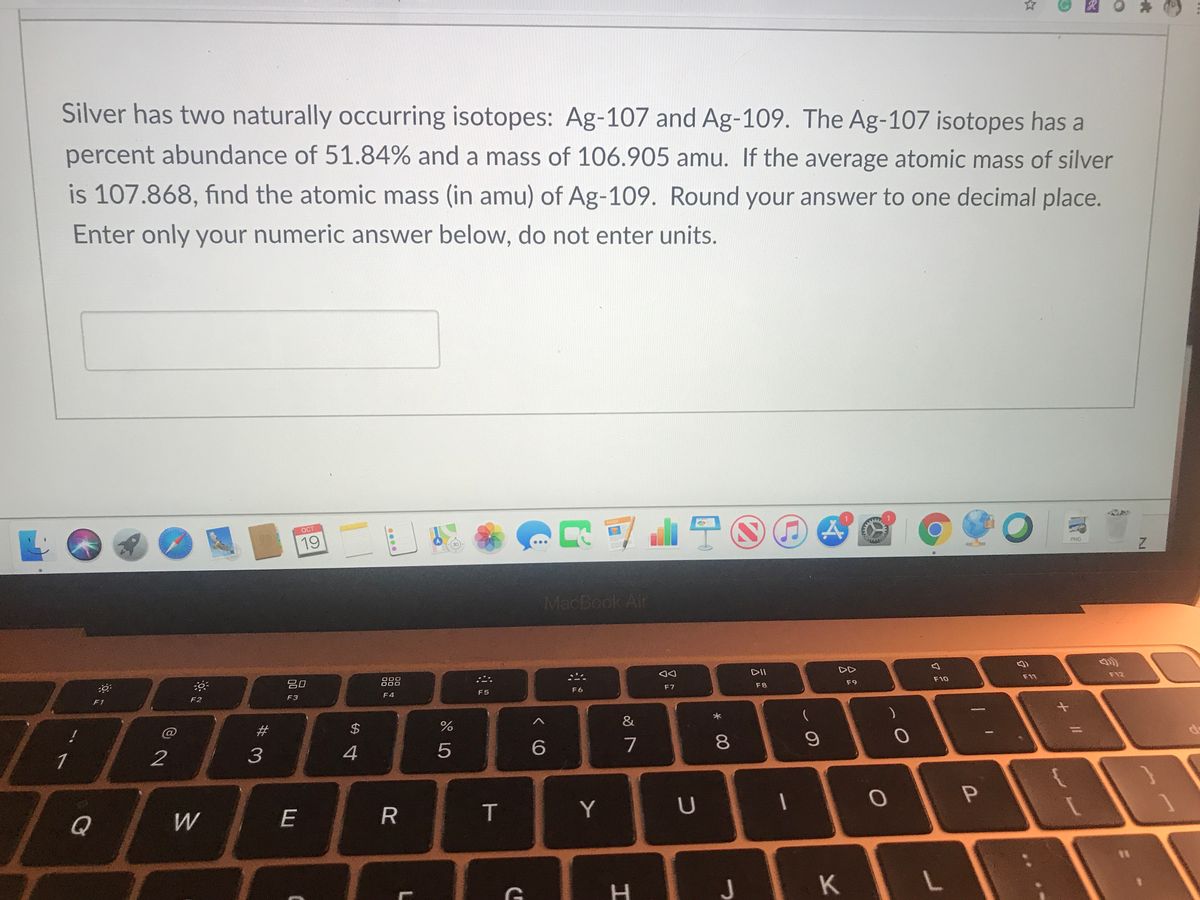
Now, back to our star: silver! Turns out, nature, in its infinite wisdom and playful experimentation, decided to give us two main types of silver atoms that exist in the wild. These are called Silver-107 and Silver-109. See the numbers? That's the total count of protons and neutrons in their respective nuclei. So, Silver-107 has 47 protons and a total of 107 particles in its core, meaning it has 60 neutrons (107 - 47 = 60). And Silver-109? You guessed it! It has 47 protons and 109 particles in its core, meaning it has 62 neutrons (109 - 47 = 62). They're like the silver twins, both unmistakably silver, but with a slight difference in their atomic weight.
Must Read
This might sound like a tiny detail, the kind of thing you'd only find in a dusty textbook, but trust me, it's actually a pretty big deal! The fact that silver comes in these two natural flavors is a testament to the beautiful complexity of the universe. It means that every piece of silver you encounter, whether it's a gleaming pendant or a vintage coin, is a subtle blend of these two isotopes. It's like a secret recipe, crafted by the cosmos itself!
Think about it this way: imagine you're baking cookies. You can use different types of flour, right? All of them are flour, but some might give you a chewier cookie, while others make it crisper. Similarly, the mix of Silver-107 and Silver-109 in a particular sample of silver can subtly influence its properties. While the difference isn't drastic enough to make your jewelry suddenly turn into a magnet or anything, it's a fundamental characteristic that scientists use all the time.
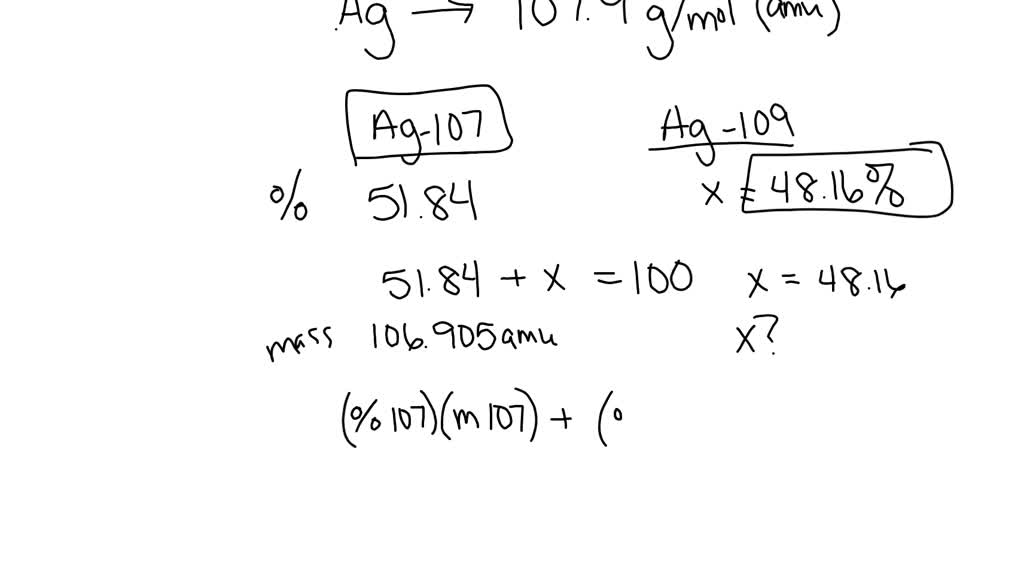
Why is this so cool, you ask? Well, for starters, it’s a fantastic conversation starter! Next time you’re at a dinner party and someone’s admiring your silver earrings, you can casually drop, "Oh, these? You know, the silver in them is actually a blend of two naturally occurring isotopes, Silver-107 and Silver-109. Isn't that wild?" Prepare for some raised eyebrows and intrigued smiles. You'll instantly be the most fascinating person in the room, armed with a little nugget of atomic trivia.
But the fun doesn't stop there! These isotopes have real-world implications that are, dare I say, dazzling.
The Art of the Trace
One of the most exciting applications of understanding isotopes is in radiometric dating. While silver itself isn't typically used for dating things like ancient fossils (that's more the realm of carbon or potassium!), the principles are the same. Scientists can use the specific ratios of isotopes in different materials to figure out how old they are. It's like a cosmic clock! And who knows, maybe future scientists will find clever ways to use the Silver-107/Silver-109 ratio in some super-advanced dating techniques. The possibilities are truly endless!
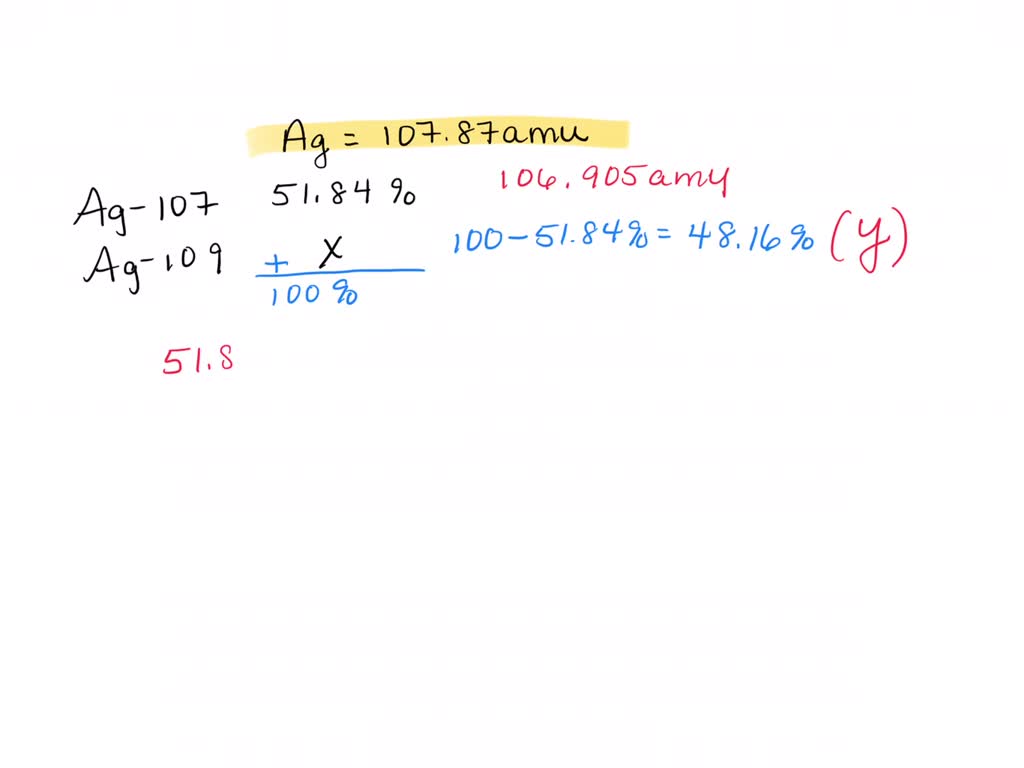
Detective Work at the Atomic Level
Isotopes are also incredibly useful in forensics. Imagine a crime scene. If a tiny, almost invisible speck of silver is found, scientists can analyze its isotopic composition. Because different silver sources (like from different mines or manufacturing processes) might have slightly different ratios of Silver-107 and Silver-109, this analysis can help trace the origin of that silver. It's like a tiny, unique fingerprint for each batch of metal! So, the next time you watch a crime drama and they talk about "trace evidence," remember that isotopes are often the silent heroes working behind the scenes.
From Jewelry to Your Health
Did you know that silver has antibacterial properties? This is a well-known fact, and understanding its isotopic makeup can sometimes help researchers refine how we utilize silver in medicine and hygiene products. While the bulk of silver's antibacterial power comes from the element itself, the subtle differences in its isotopes might play a role in how efficiently it interacts with microbes or how it’s absorbed in certain biological processes. It's a little bit of atomic nuance that could lead to big advancements!
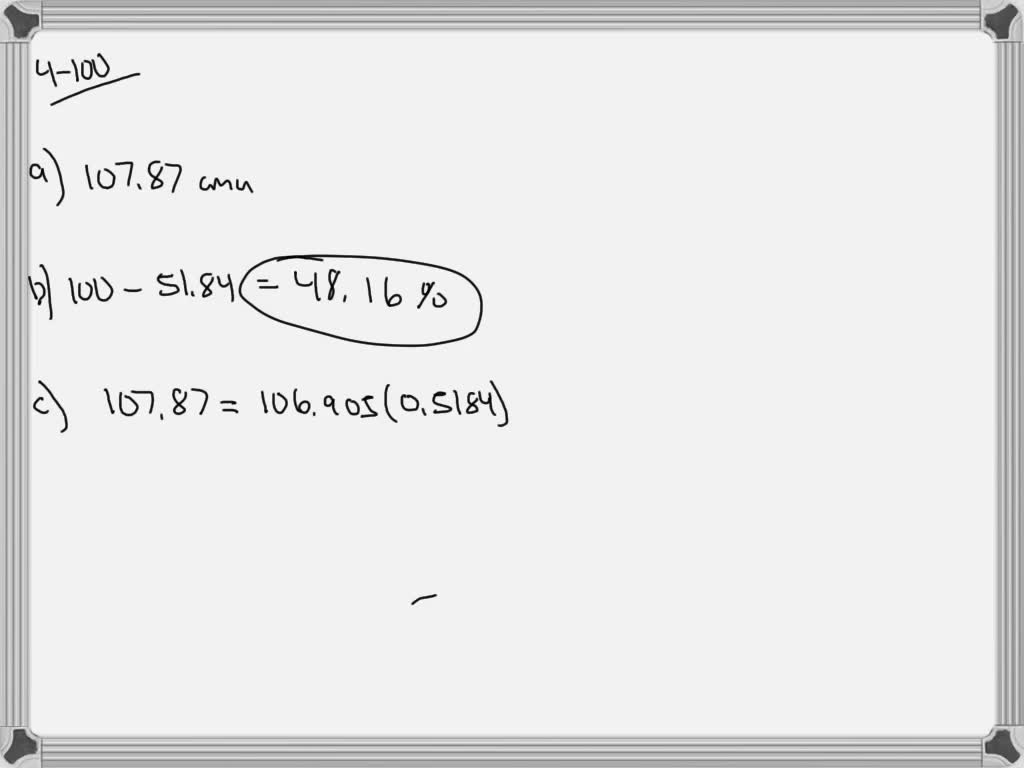
The Beauty of Natural Variation
Honestly, just knowing that silver isn't just a simple, uniform substance is kind of inspiring, don't you think? It reminds us that even the most familiar things in our world have hidden depths and fascinating complexities. It's a beautiful illustration of how nature loves to play with its building blocks, creating variety and uniqueness even within the same fundamental structure.
So, the next time you admire a piece of silver, take a moment to appreciate its two-part nature. Think about Silver-107 and Silver-109, the unsung heroes contributing to its luster and its story. This little bit of atomic knowledge isn't just for scientists in lab coats; it's a reminder that the world is full of wondrous details waiting to be discovered. It encourages us to look a little closer, ask a few more questions, and find the extraordinary in the ordinary.
Isn't that just a fantastic thought? It makes you want to grab a magnifying glass and see what other everyday objects hold such fascinating secrets, doesn't it? Go forth, my friends, and let your curiosity sparkle! Who knows what other amazing atomic tales you'll uncover!