Show The Orbital Filling Diagram For Bromine
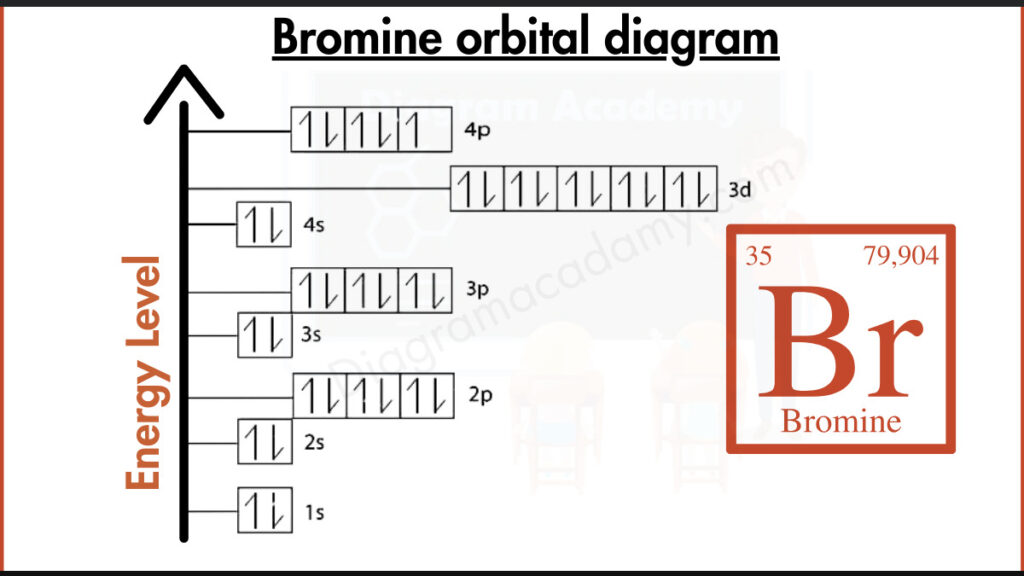
So, you want to talk about bromine? Specifically, its orbital filling diagram? Yeah, I get it. It’s not exactly the kind of thing you bring up at a dinner party, is it? Unless your dinner party guests are fellow chemists, in which case, more power to you! But for the rest of us, the idea of an orbital filling diagram can sound a bit… much. Like trying to fold a fitted sheet perfectly. It’s theoretically possible, but the outcome is often chaotic.
And when we talk about bromine, it’s like the universe decided to give us a bit of a curveball. It's not the simplest element. It’s got a certain… presence. A bit of a dramatic flair. You know how some people are always the center of attention? Bromine kind of feels like that in the elemental world. It’s got this whole entourage of electrons, and they’re all trying to find their rightful spots. It’s a whole electron party, and we’re here to check the guest list and see where everyone’s chilling.
Now, the orbital filling diagram. Think of it like a meticulously organized closet. But instead of sweaters and socks, we’ve got electrons. And instead of shelves, we’ve got orbitals. And these aren’t just any old orbitals; they’ve got specific shapes and energy levels. It’s like having different sized drawers for your different kinds of socks – the athletic ones, the fuzzy ones, the ones that mysteriously disappear in the wash.
Must Read
Let’s be honest, looking at an orbital filling diagram for something like bromine can feel a little overwhelming. It’s a series of boxes, little arrows, and a whole lot of letters and numbers. It’s like a secret code, and if you’re not fluent in electron-speak, you might just want to nod and smile. But that’s okay! We’re not here to judge. We’re here to… well, look at it. And maybe find it a little bit funny in its complexity.
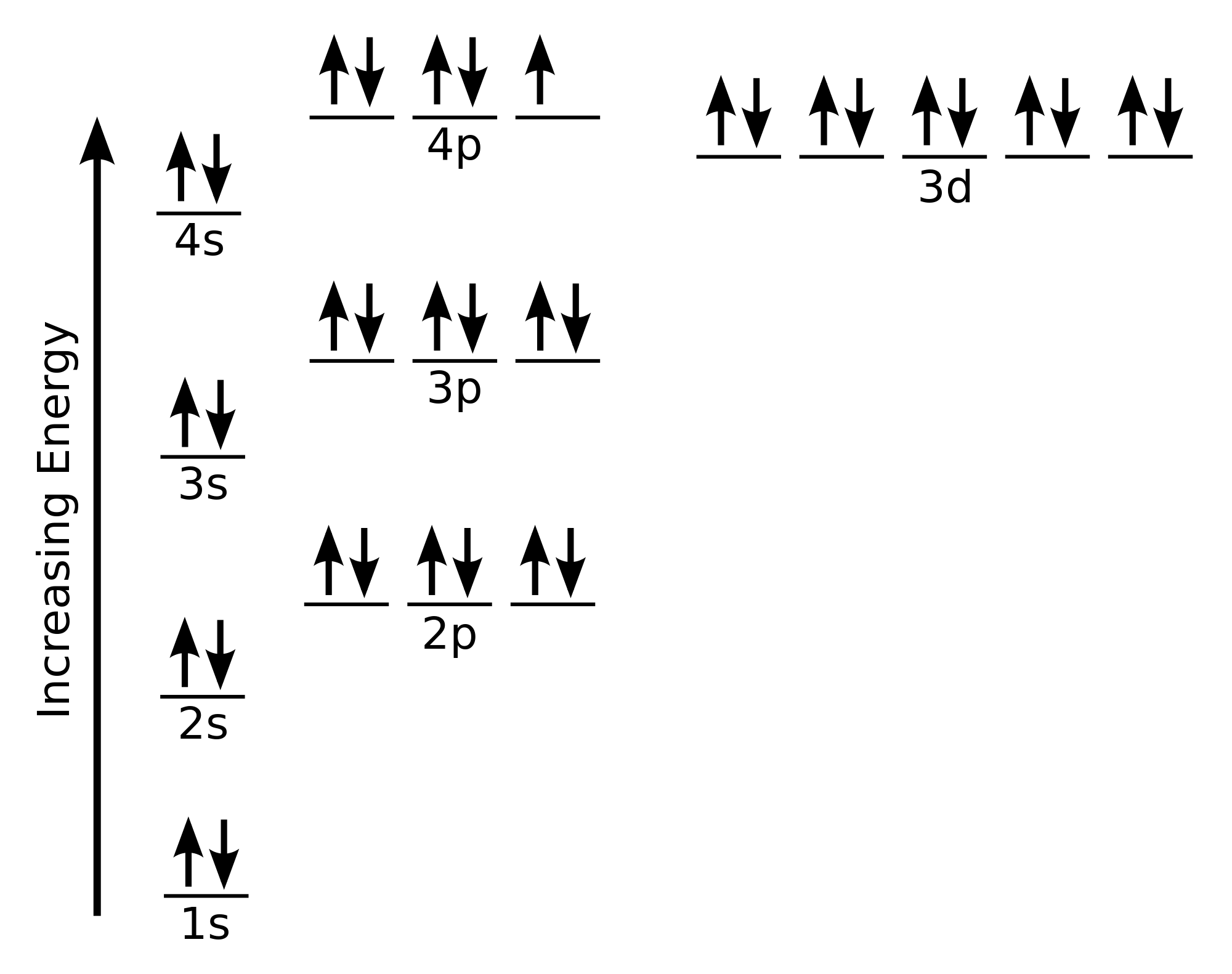
So, picture this: we’re building up the electron structure for bromine. It’s like filling up those sock drawers, but in a very specific order. There are rules, of course. Because the universe loves rules, especially when it comes to tiny, invisible particles. The Aufbau principle, the Pauli exclusion principle, and Hund's rule are the bouncers at this electron party, making sure everyone gets in and sits in the right seat. No double-bookings allowed, and everyone gets their own little space if there’s one available.
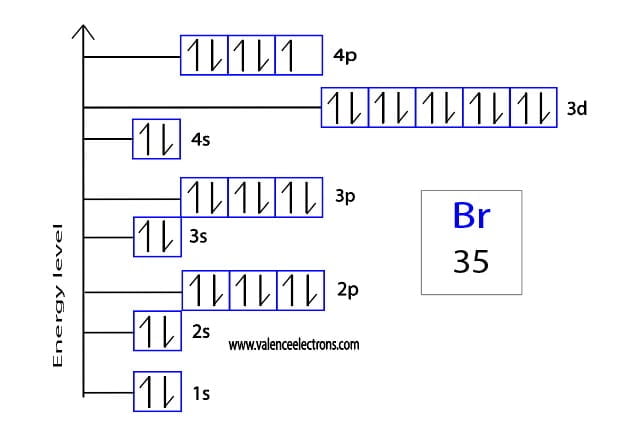
First, we’ve got the lowest energy levels. Think of these as the prime real estate. The 1s orbital. It’s small, it’s cozy, and it can only hold two electrons. Like a tiny studio apartment. Then we move up. The 2s orbital, another little apartment. And then the 2p orbitals. These are like a set of three small studios, all at the same level, but they can each hold a couple of electrons. It’s starting to feel like a bustling apartment complex, isn’t it?
We keep filling these orbitals, climbing up the energy ladder, always following the rules. The 3s orbital, then the 3p orbitals. It’s a systematic process. Like making a perfectly layered cake. You don’t just dump all the frosting on at once. You build it up, layer by layer. And each layer has to be just right.
Now, bromine. It’s element number 35. So, we have 35 electrons to place. That’s a lot of little guys trying to find their spots. We've got the inner shells, all filled up and happy. Then we get to the outer shells, the ones that are really involved in all the exciting chemistry. And this is where it gets a little more interesting.
We fill the 4s orbital. Then come the 3d orbitals. These are like a whole new set of apartments, a bit more spacious, and there are five of them. Five more places for electrons to settle down. It’s like a condo complex opening up.
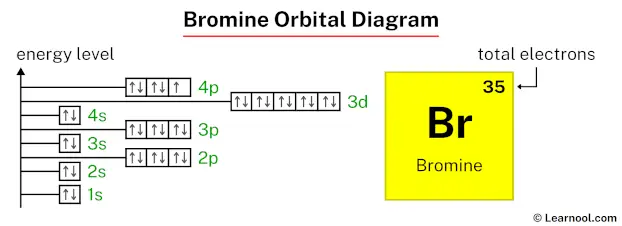
And finally, we reach the 4p orbitals. These are the outermost ones for bromine, the ones that are really going to interact with other atoms. And they’re not all full. This is where bromine gets its personality. It’s got some space left in those 4p orbitals. It’s like there are a few empty seats at the table, and bromine is just waiting to see who might join it. It's kind of relatable, right? That feeling of being almost complete, but with room for a little more interaction.
So, when you look at the orbital filling diagram for bromine, it’s not just a bunch of boxes and arrows. It’s a story of electrons finding their places, following strict but fair rules, and setting the stage for how bromine behaves. It’s a little bit organized, a little bit crowded, and a whole lot of precise electron placement. And while it might not be the most thrilling topic, there’s a certain elegance to it. The universe, in its own peculiar way, has a system, even for something as seemingly chaotic as an atom’s electron cloud. And that, my friends, is something to… well, to at least acknowledge with a slight smirk and a nod of understanding. It’s just electrons doing their thing, after all. No biggie. (Except it kind of is, when you think about it.)






