Select The Correct Statement About Isotopes

So, picture this: I’m in my late teens, absolutely convinced I know everything about the universe. You know the feeling, right? When you’ve just mastered a new concept and suddenly the whole world feels like it’s explained? Yeah, well, I was deep in my chemistry phase, and I’d just learned about elements. Easy peasy. Hydrogen is H, oxygen is O, carbon is C. Got it. And then, my teacher, bless his patient soul, dropped the word “isotopes” on us. Isotopes. It sounded vaguely alien. I remember thinking, “Wait, so not all atoms of the same element are… identical? What kind of sorcery is this?” It was a tiny crack in my perfectly ordered, simplistic understanding of reality, and honestly, it kind of blew my mind.
It’s like thinking all apples are exactly the same, and then someone tells you, “Actually, there are Fuji apples, Gala apples, Granny Smith apples… they’re all apples, but they have their own little quirks.” And you’re standing there with a confused half-eaten apple, wondering if your whole fruit-based worldview is about to crumble. It wasn’t crumbling, of course, just… expanding. And that, my friends, is exactly where we’re headed today. We’re going to talk about isotopes, and I promise, it’s way cooler than it sounds. We’re going to pick out the correct statement about them, and hopefully, by the end, you’ll feel that satisfying “aha!” moment, just like I did (albeit with a bit more maturity and less dramatic internal monologue).
Let’s start with the absolute basics, because, you know, we all have to start somewhere. Elements are defined by their number of protons. This is the fundamental identity card of an atom. Think of it as its social security number, its DNA. If you change the number of protons, you change the element. Period. End of story. So, all atoms of carbon, no matter what else is going on, will have 6 protons. All atoms of oxygen have 8 protons. Simple enough, right?
Must Read
But here’s where the plot thickens. Atoms also have neutrons, and these little guys hang out in the nucleus right alongside the protons. And here’s the kicker: the number of neutrons can vary within the atoms of the same element. This is the secret sauce, the little twist that makes isotopes possible. They are like cousins in the same family, sharing the same basic DNA (protons) but having slight physical differences (neutrons).
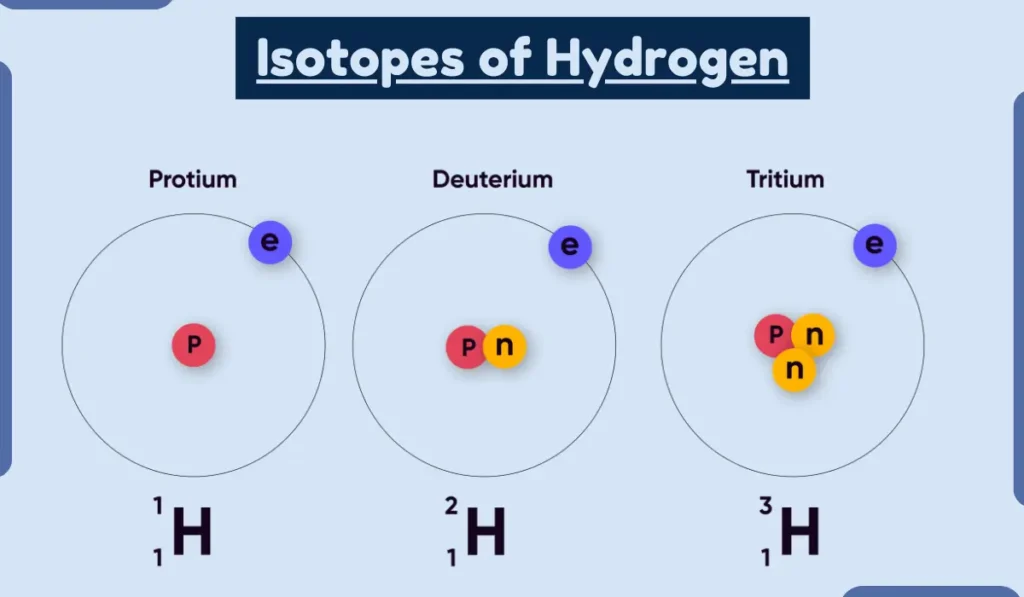
So, an isotope of an element is basically an atom of that element that has a different number of neutrons than the most common form of that element. It’s still the same element, still the same number of protons, but it’s got a slightly different weight, a different atomic mass. Imagine you have a set of identical twins (protons). Now, give one twin a small backpack to carry (neutrons), and the other twin a slightly heavier backpack. They’re still the same person, fundamentally, but one is carrying a bit more. That’s kind of what’s happening with isotopes.
The All-Important Distinction: Protons vs. Neutrons
This is the absolutely, unequivocally most crucial point to grasp when talking about isotopes. It’s the bedrock upon which all understanding of isotopes is built. If you remember nothing else from this whole rambling discourse, remember this: The number of protons defines the element. The number of neutrons can vary within an element, creating isotopes.
Let’s dive into an example, because examples are, dare I say it, deliciously illustrative. Carbon, that ubiquitous element found in everything from diamonds to your own body, is a fantastic case study. Most carbon atoms you encounter in the world, the everyday, run-of-the-mill carbon, have 6 protons and 6 neutrons. We call this Carbon-12 (the 12 comes from 6 protons + 6 neutrons, you see? Easy math!).
But then, there’s Carbon-13. It still has 6 protons (because if it didn’t, it wouldn’t be carbon, would it?). But this version of carbon has 7 neutrons. So, it’s still carbon, but it’s a bit heavier. And then, there’s the famous Carbon-14. Again, 6 protons, but this one has 8 neutrons. It’s even heavier. These are all isotopes of carbon.
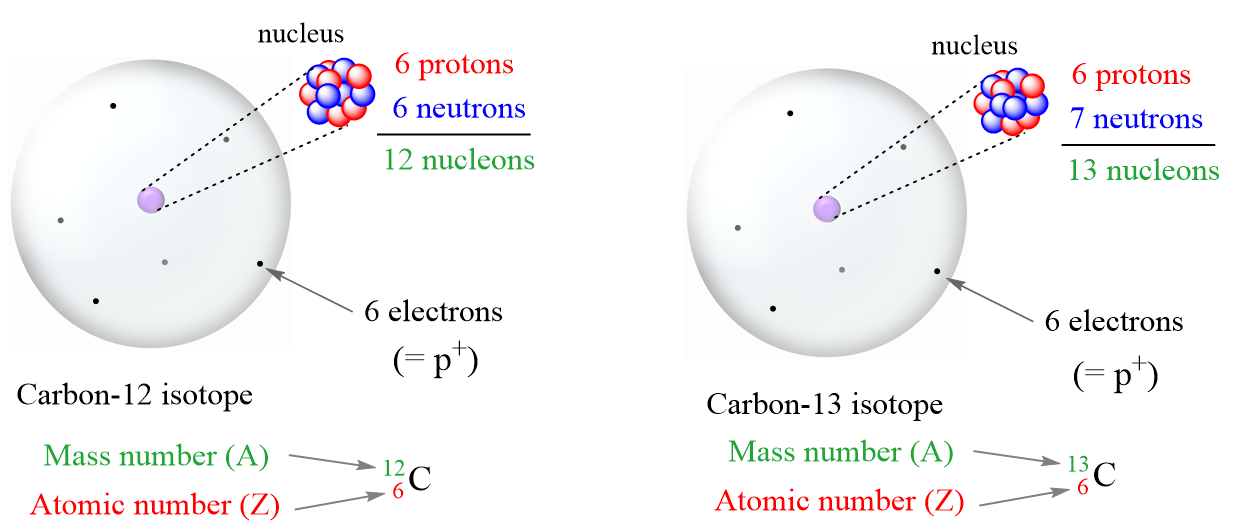
The key takeaway here is that while Carbon-12, Carbon-13, and Carbon-14 are different in their neutron count and therefore their mass, they all behave chemically in pretty much the exact same way. Why? Because chemical reactions are primarily driven by the electrons, and the number of electrons in a neutral atom is equal to the number of protons. Since all these carbon isotopes have the same number of protons, they have the same number of electrons and thus, the same chemical properties. Isn’t that neat?
Think of it like this: Imagine you have three identical cars, same make, model, and color. But one has a slightly bigger engine, and another has a really beefy engine. They’ll all drive, they’ll all turn corners, they’ll all brake. They might have slightly different acceleration or top speeds, but their fundamental function as a car remains the same. The engine size is like the neutron count – it affects some performance aspects (like mass or stability), but the core identity and operation (chemical behavior) are dictated by the chassis and the driver’s inputs (protons and electrons).
Common Misconceptions (and How to Dodge Them Like a Pro)
Okay, now that we’ve laid the groundwork, let’s tackle some of the common pitfalls that can trip you up when thinking about isotopes. These are the things that might make you pick the wrong statement. So, let’s arm you with the knowledge to be right!
Misconception 1: Isotopes have different numbers of electrons. Nope! As we just discussed, in a neutral atom, the number of electrons equals the number of protons. Since isotopes of an element have the same number of protons, they also have the same number of electrons. This is why their chemical behavior is so similar. If you see a statement that says isotopes have different electron counts, that’s a big red flag. Unless we’re talking about ions, but that’s a whole other kettle of fish, and not the defining characteristic of isotopes themselves.
Misconception 2: Isotopes have different numbers of protons. This is the classic switcheroo. If an atom has a different number of protons, it’s not an isotope of the original element; it’s a completely different element. Remember our carbon example? Change the 6 protons to 7, and you’ve got nitrogen. Change it to 5, and it’s boron. Always, always, always stick to the proton count for elemental identity.
Misconception 3: Isotopes are always radioactive. Ah, this is a common one, especially with the exciting world of radioactive dating using Carbon-14. It’s true that many isotopes are radioactive (meaning their nucleus is unstable and decays over time), but not all of them are. For instance, Carbon-12 and Carbon-13, the most common forms of carbon, are perfectly stable and not radioactive at all. So, while radioactivity is a property some isotopes possess, it’s not a universal defining feature of all isotopes. A statement claiming all isotopes are radioactive is, unfortunately, incorrect.

Misconception 4: Isotopes have different chemical properties. This one is tricky because while their fundamental chemical behavior is the same, there can be subtle differences, known as kinetic isotope effects. These are minor variations in reaction rates due to the different masses of the isotopes. For example, a reaction involving Carbon-14 might proceed slightly faster or slower than the same reaction involving Carbon-12. However, when we’re talking about the general defining characteristics of isotopes in introductory chemistry, the statement that their chemical properties are the same is overwhelmingly considered correct because the differences are usually negligible for most purposes. So, if a statement emphasizes identical chemical properties, it’s generally on the right track, but if it claims absolutely no difference whatsoever in any context, that might be a bit too absolute and therefore technically debatable in very specific scientific scenarios. But for our purposes, focusing on the dominant, shared chemical behavior is key.
So, What’s the Correct Statement?
Alright, let’s distill all this down. Imagine you’re presented with a few statements. Which one will be the winner? It’s going to be the one that accurately reflects our understanding. Here are a few possibilities, and we’ll pick the truly correct one:
Statement A: Isotopes of an element have different numbers of protons.
Statement B: Isotopes of an element have different numbers of neutrons and therefore different atomic masses.
Statement C: Isotopes of an element have different numbers of electrons.
Statement D: Isotopes of an element have significantly different chemical properties.
Let’s break them down, using what we’ve learned:
Statement A is incorrect. Different numbers of protons mean a different element, not an isotope.
Statement C is incorrect. In neutral atoms, electron count matches proton count, and isotopes have the same proton count.
Statement D is generally incorrect. While minor kinetic isotope effects exist, their fundamental chemical behavior is largely the same.
This leaves us with Statement B. Let’s examine it closely. “Isotopes of an element have different numbers of neutrons…” Yes, that’s our core definition! “…and therefore different atomic masses.” Absolutely! Since the atomic mass is roughly the sum of protons and neutrons, and the neutron number changes, the atomic mass will change. This statement perfectly captures the essence of what isotopes are and why they are distinct from each other, while still being part of the same element family. It’s the gold standard!
Why Does This Even Matter? (Besides acing a test, of course!)
You might be thinking, “Okay, interesting chemistry lesson, but why should I care about these slightly different versions of atoms?” Well, these subtle differences are actually HUGE in many fields.

For starters, radioactive isotopes are the backbone of medical imaging and treatment. Think about PET scans; they use isotopes to see what’s happening inside your body. They’re also used in cancer therapy to target and destroy diseased cells.
Then there’s radiometric dating. Carbon-14 dating, as I mentioned, is how we figure out how old ancient artifacts are. It’s like a tiny, built-in clock within the material itself, ticking away based on the decay of a specific isotope.
In geology, scientists use the ratios of different stable isotopes in rocks and water to understand past climates, track the movement of water through the environment, and even figure out the origins of different geological formations. It’s like a fingerprint left by nature.
And in research labs all over the world, isotopes are used as tracers. They can be incorporated into molecules to track their pathways in biological systems or chemical reactions, giving scientists incredibly detailed insights into complex processes. It’s like attaching a tiny GPS to molecules.
So, the next time you hear the word “isotope,” don’t picture something obscure and theoretical. Picture those apple varieties, or those cars with different engines, or those tiny clocks and trackers that unlock so many secrets of our universe. It’s a concept that’s both simple and profoundly impactful, and now you’re armed with the knowledge to correctly identify the defining characteristics of these fascinating atomic variations.
Remember that feeling of wanting to know everything when you were younger? Well, science is a constant unfolding of those “aha!” moments. Isotopes are just one more piece of the incredible puzzle. Keep that curiosity alive!