Red Blood Cell Placed In Pure Water Would

Alright, picture this. You've got a tiny little red blood cell. Think of it as a microscopic donut, but way more important. It’s cruising around your bloodstream, doing its job, carrying oxygen, all that jazz. Super important little guy, right?
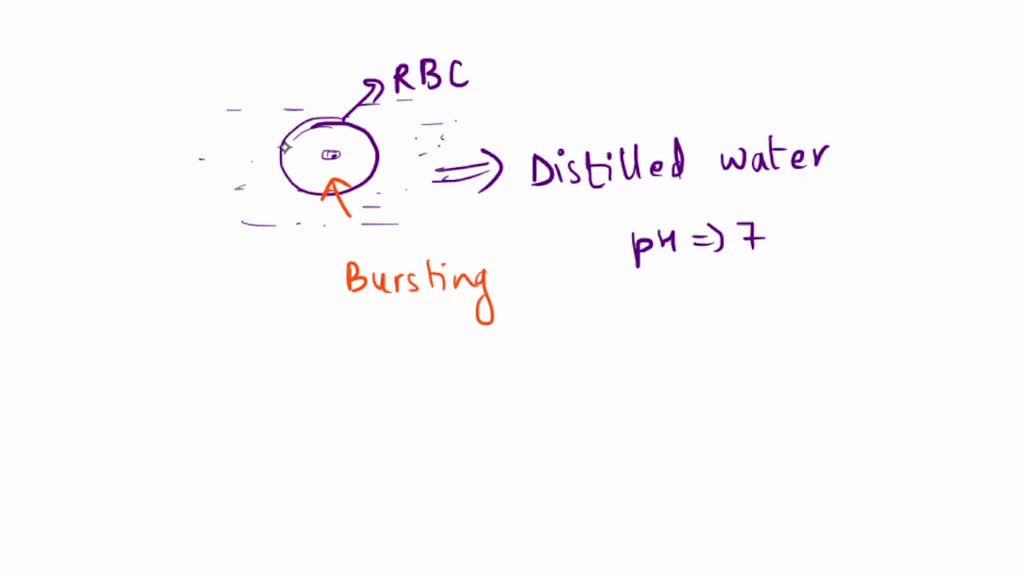
Now, imagine this little guy suddenly finds itself in a place where there’s absolutely no salt. No anything, really, except pure, unadulterated water. Like, the fanciest, most mineral-free water you can think of. Maybe it fell into a puddle after a super clean rain shower, or perhaps it's on a tiny adventure that went… sideways.
What do you think happens? Does it just chill out? Maybe take a refreshing dip? I've got a sneaking suspicion it’s not quite that relaxing. My completely unqualified, but totally entertaining, guess? It gets a little… overwhelmed. Like when you’ve had too much fizzy drink and your tummy feels all… puffy. Yeah, something like that.
Must Read
Think about it. Our red blood cells are used to a specific environment. It’s like being in a perfectly seasoned soup. Everything is just right. The saltiness, the other bits and bobs floating around – it keeps them happy and in shape. They’re like little water balloons, but very, very carefully inflated.
But pure water? That's like taking that perfectly seasoned soup and diluting it down to… well, nothing. It’s like someone turned the flavor dial all the way to zero. And our little blood cell, bless its tiny, oxygen-carrying heart, doesn’t know what to do.

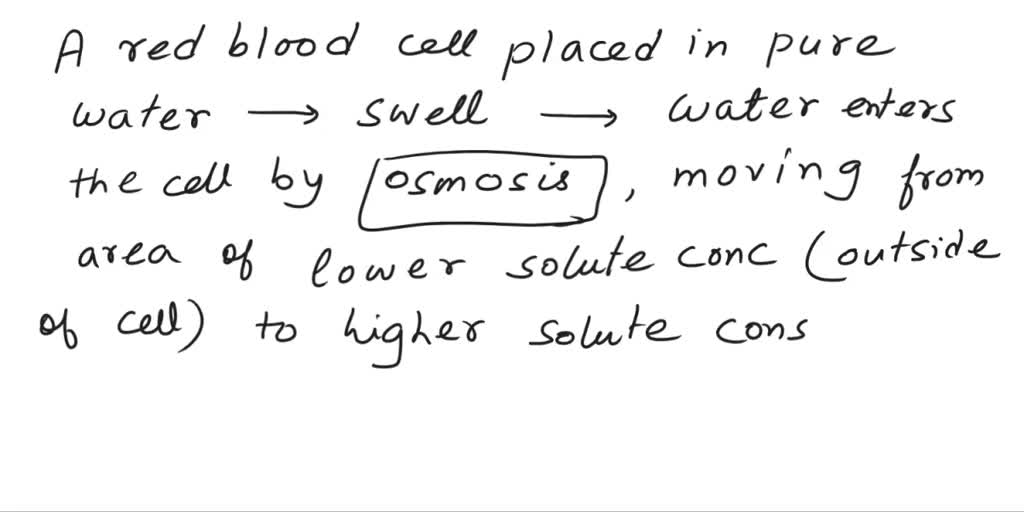
It’s probably thinking, “Whoa, what is this stuff? It’s so… thin!” It’s like stepping out of a cozy, slightly crowded room into a vast, empty desert. There’s just too much space for the water. And because water is naturally a bit of a nosy parker, it starts to sneak in. It wants to spread out, you see. It's trying to make things even, like a really enthusiastic tidier-upper.
So, this water starts to flood into the red blood cell. And the cell, not built for such an influx of pure hydration, starts to swell. It’s like it’s trying to hold onto its precious interior, but the outside is just so… welcoming to water. It’s a one-way street for H2O, and the cell is the unfortunate destination.
I imagine it getting puffier and puffier. Like a tiny balloon being overinflated by an eager child. It’s not a gentle puff, either. This is serious puffing. It’s probably getting quite uncomfortable for our little friend. Maybe it starts to feel a bit stretched. A bit… wobbly.

And then, at some point, the inevitable happens. You can only stretch a microscopic donut so far, right? It’s got to give. It’s like a party balloon that’s been blown up a bit too much. You know that moment? That little pop? Yeah, I’m thinking that’s what happens.
My highly scientific, completely fabricated theory is that the red blood cell just… bursts. It’s like its personal bubble just can’t take it anymore. The pure water rushes in, swells it up like a soufflé gone wrong, and then… splat. It’s the end of its oxygen-carrying career. A dramatic exit, for sure.

It's like it goes from a perfectly formed, slightly squishy disc to a sad, watery puddle of its former self.
So, while some might tell you about osmosis and hypotonic solutions (booooring!), I prefer to think of it as a dramatic, if slightly messy, end for our little red blood cell. It tried its best to hold on, but the allure of pure water was just too much. It got a bit too enthusiastic about drinking, and well, you know how that can go.
It’s a harsh reality, perhaps, but a funny one to imagine. Our brave little red blood cells, so vital and strong in their natural habitat, meeting their maker in a puddle of pure, unadulterated nothingness. They’re not built for that kind of extreme minimalist lifestyle. They need their salty soup!

So, next time you think about your blood, spare a thought for the humble red blood cell. It’s a tough job, and it’s even tougher if it gets accidentally dumped into the spa treatment room of pure water. They’re basically tiny, living water balloons that don’t do well with too much splashing.
And that, my friends, is my entirely unscientific, but hopefully entertaining, explanation of what happens when a red blood cell meets pure water. It’s not a happy reunion. It’s more of a… watery explosion of destiny. A tiny tragedy, but one that makes you smile, at least a little, when you picture it.
It's a tough gig, being a red blood cell. You gotta stay in your lane, and your lane is definitely not pure water. Keep it seasoned, people! Keep it seasoned!