Rank The Following Elements By Atomic Radius
So, I was at my nephew Leo's birthday party last weekend, right? You know how it is. Face painting, a bouncy castle that looked like it was about to achieve orbit, and a cake that was suspiciously shaped like a superhero's cape. Amidst the chaos of tiny humans fueled by sugar, Leo, in his newfound superhero persona, pointed at the balloons and asked, "Uncle Alex, why are some balloons bigger than others?"
Honestly, my first instinct was to say, "Because they have more air, silly!" But then I saw the wheels turning in his little head, the genuine curiosity. And it got me thinking. You see, Leo's question, in its own adorable way, is a bit like asking about the atomic radius of elements. Why are some atoms bigger than others? It's not about air, of course. It's about something much, much smaller, but equally fascinating: the fundamental building blocks of everything we know.
And wouldn't you know it, the universe, in its infinite wisdom (and our handy periodic table), has given us a whole cast of characters to play with when it comes to atomic size. Today, we're going to dive into that! We're going to take a few elements and try to rank them by their atomic radius. Think of it as a miniature science party, but with fewer sticky fingers and a lot more protons. Sounds fun, right?
Must Read
The Sneaky Sneaky Nature of Atomic Size
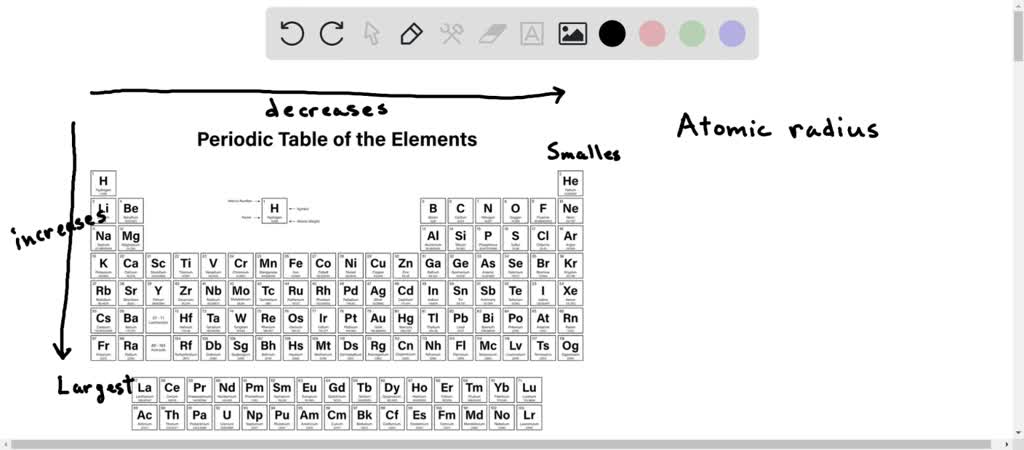
Now, before we start throwing elements around like confetti, let's get a handle on this whole "atomic radius" thing. You'd think it would be straightforward, wouldn't you? Just measure from the center of the nucleus to the outermost electron. Boom. Done. But oh no, science loves to keep us on our toes. It's not quite that simple. Atoms don't have neat, defined edges like, well, a balloon!
Electrons are a bit like enthusiastic, but slightly erratic, party guests. They don't just hang out in neat little circles. They exist in orbitals, which are more like probability clouds. So, when we talk about atomic radius, we're often talking about an average distance, or sometimes the distance between the nuclei of two identical atoms bonded together. It’s a bit of a fuzzy concept, which, frankly, is kind of cool. Like trying to catch a perfectly formed soap bubble – it's there, but not really.
And the magic behind these size differences? It all boils down to two main players: the number of protons in the nucleus (which determines the element, obviously) and the number of electron shells. Think of the nucleus as the sun, and the electrons as planets orbiting it. More protons mean a stronger "gravitational pull" (that's the positive charge of the nucleus attracting the negative electrons). And more electron shells mean the outer electrons are further away from the pull of the nucleus. It's a constant tug-of-war, and that tug-of-war dictates the size.
Our Lucky (or Unlucky?) Elements
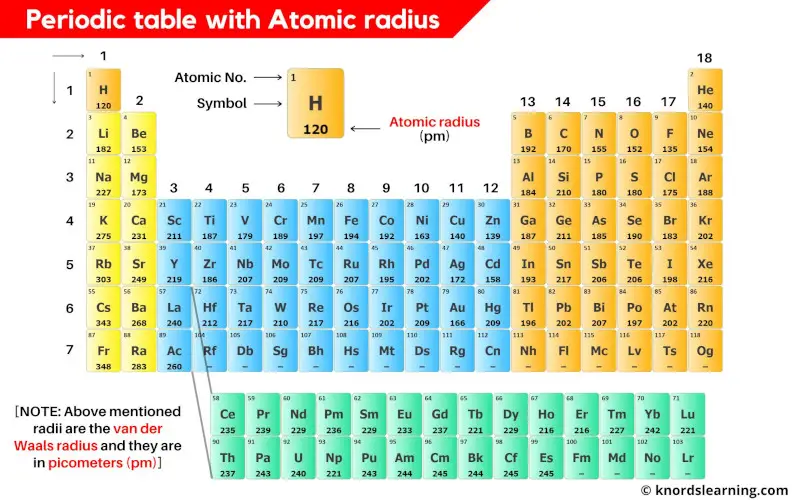
Alright, drumroll please! For our little size comparison today, we're going to pick a few elements from across the periodic table. This isn't an exhaustive list, of course – that would take forever – but it will give us a good feel for the trends. We've got:
- Sodium (Na) – A classic alkali metal.
- Chlorine (Cl) – Our electronegative friend from the halogens.
- Potassium (K) – Sodium's bigger cousin.
- Argon (Ar) – The noble gas, chillin' in its own lane.
- Sulfur (S) – The element that gives eggs their distinctive aroma.
So, what do we have here? We've got a mix. Sodium and Potassium are in the same group (column) but different periods (rows). Chlorine and Sulfur are in the same period, and Argon is right next to Chlorine. This is going to be interesting!

Let the Size Ranking Begin!
Okay, let’s put on our hypothetical scientist lab coats (mine is probably stained with coffee, yours is probably pristine). We need to figure out who's the biggest, who's the smallest, and why.
The Reigning Champion (of Size): Potassium (K)
Let's start with the big guy. Potassium (K). Why is Potassium likely to be the largest? If you look at the periodic table, Potassium is in Period 4, Group 1. This means it has 19 protons pulling on its electrons, but crucially, it has electrons in four main energy shells. The outermost electrons are pretty far from the nucleus, and the inner shells shield them somewhat from the nucleus's full pull. It's like having a large house with many rooms – the furthest rooms are quite distant from the central heating.
Think about its neighbor in the same group, Sodium. Sodium (Na) is in Period 3, Group 1. It has fewer electron shells (three). Even though it has fewer protons (11), the extra shell in Potassium makes a significant difference. More shells almost always mean a bigger atom, all else being equal.
So, Potassium is our current reigning monarch of atomic radius among this selection. It's got the real estate!
Next in Line: Sodium (Na)
As we just touched upon, Sodium (Na) comes in next. It's in the same group as Potassium, so it has that single valence electron in its outermost shell, making it eager to react (like most alkali metals). But, it’s in Period 3, meaning it has three electron shells instead of Potassium’s four. Fewer shells mean the electrons are held a bit closer to the nucleus.
It's like comparing two houses again. Potassium is the sprawling mansion, and Sodium is the cozy, but still substantial, bungalow. Both have a front yard, but the mansion's front yard is much further from the house itself. So, while Sodium is definitely on the larger side, it can’t quite compete with Potassium’s shell count.

The Middle Ground: Sulfur (S)
Now we’re moving into Period 3, where things get a bit more complex because we’re moving across the row. We have Sulfur (S), which is in Period 3, Group 16. It has 16 protons and electrons in three main shells, just like Sodium. However, it has a lot more protons in its nucleus (16 vs. Sodium's 11).
This is where the increasing nuclear charge comes into play. With more protons, the nucleus has a stronger positive pull on the electrons. This stronger pull draws the electron shells closer to the nucleus, making the atom smaller. So, even though Sulfur is in the same period as Sodium, its higher number of protons is going to shrink it down a bit compared to Sodium.
It’s like having two people of the same height, but one has a much stronger grip. That stronger grip pulls everything in tighter. Sulfur is still a reasonably sized atom, but it’s definitely getting squeezed compared to our alkali metal friends.
Getting Tighter: Chlorine (Cl)
Following the trend across a period, Chlorine (Cl) is next. It’s in Period 3, Group 17. Chlorine has 17 protons, one more than Sulfur, and also occupies three electron shells. That extra proton provides an even stronger nuclear pull.
So, like Sulfur, Chlorine is experiencing that inward tug. The increased positive charge in the nucleus is pulling those three electron shells even more tightly. Imagine that grip getting even stronger. Chlorine is going to be smaller than Sulfur, which was already smaller than Sodium.

This is the classic trend of atomic radius decreasing as you move from left to right across a period. The number of electron shells stays the same, but the nuclear charge increases, shrinking the atom.
The Smallest Sprout: Argon (Ar)
And finally, we have Argon (Ar). It’s in Period 3, Group 18, right next to Chlorine. Argon has 18 protons and electrons filling three main shells. The key here is that its outermost shell is full. This makes Argon a noble gas, meaning it’s very stable and unreactive. It’s not desperate to gain or lose electrons.
Because it has the most protons (18) in this period and a full outer shell, the nuclear pull is at its strongest, and the electron cloud is as compact as it can be for this electron configuration. It's the ultimate "pulling its own weight" scenario. The electrons are held very tightly to the nucleus.
So, amongst our chosen contenders, Argon is the smallest. It’s the compact, self-contained unit.
Putting It All Together: The Grand Ranking!
So, let's recap our findings, from largest to smallest atomic radius:
- Potassium (K): The champion! Four electron shells, relatively few protons for that number of shells.
- Sodium (Na): Still a contender. Three electron shells, fewer protons than Sulfur and Chlorine.
- Sulfur (S): Getting smaller. Three shells, but a higher nuclear charge than Sodium.
- Chlorine (Cl): Smaller still. Three shells, one more proton than Sulfur, increasing the pull.
- Argon (Ar): The smallest. Three shells, the highest nuclear charge in this group, and a full outer shell means tight control.
So, our ranking is: K > Na > S > Cl > Ar. Pretty neat, huh? It's a lovely demonstration of how the interplay between the number of electron shells and the nuclear charge dictates the size of an atom.

Why Does This Even Matter? (Besides Your Nephew's Balloon Question)
You might be thinking, "Okay, Alex, that's cool and all, but why should I care about atomic radius?" Great question! It's not just abstract knowledge. The atomic radius of an element influences its chemical properties. How it bonds, how reactive it is, what kinds of compounds it forms – all of it is tied back to its size and how its electrons are arranged.
For example, alkali metals like Sodium and Potassium have large atomic radii, and their single, loosely held valence electron makes them very reactive. They readily give up that electron to form ionic bonds. Noble gases, on the other hand, like Argon, have full outer shells and small atomic radii (relative to their position in the period), which makes them incredibly unreactive. They’re the introverts of the periodic table, perfectly happy in their own little electron shells.
Understanding atomic radius is like understanding the fundamental dimensions of the Lego bricks that build our universe. It helps us predict how they’ll fit together, how strong those connections will be, and what amazing structures we can create. It’s the secret language of chemistry!
A Tiny Bit of Ironic Fun
Isn't it ironic, though? We're talking about things unimaginably small – atoms, protons, electrons – and yet their relative sizes have such massive implications for the world around us. The way water behaves, the strength of steel, the colors of a sunset – it all comes back to these fundamental atomic structures and their sizes.
And thinking back to Leo and his balloons, it’s a perfect analogy. Some balloons are bigger because they have more air, stretching the material further. Atoms are bigger (or smaller) based on the fundamental forces and configurations of their subatomic particles, dictating how far those electron clouds extend. Both are about the extent of something, just on wildly different scales. Science is everywhere, even in a chaotic birthday party!
So, the next time you look at the periodic table, don’t just see a grid of letters and numbers. See a collection of characters with their own unique sizes, personalities, and a whole lot of stories to tell about how they interact. And remember, even the smallest questions can lead to some of the biggest understandings. Now, if you'll excuse me, I think I hear the ice cream truck. That’s another kind of radius I can get behind!