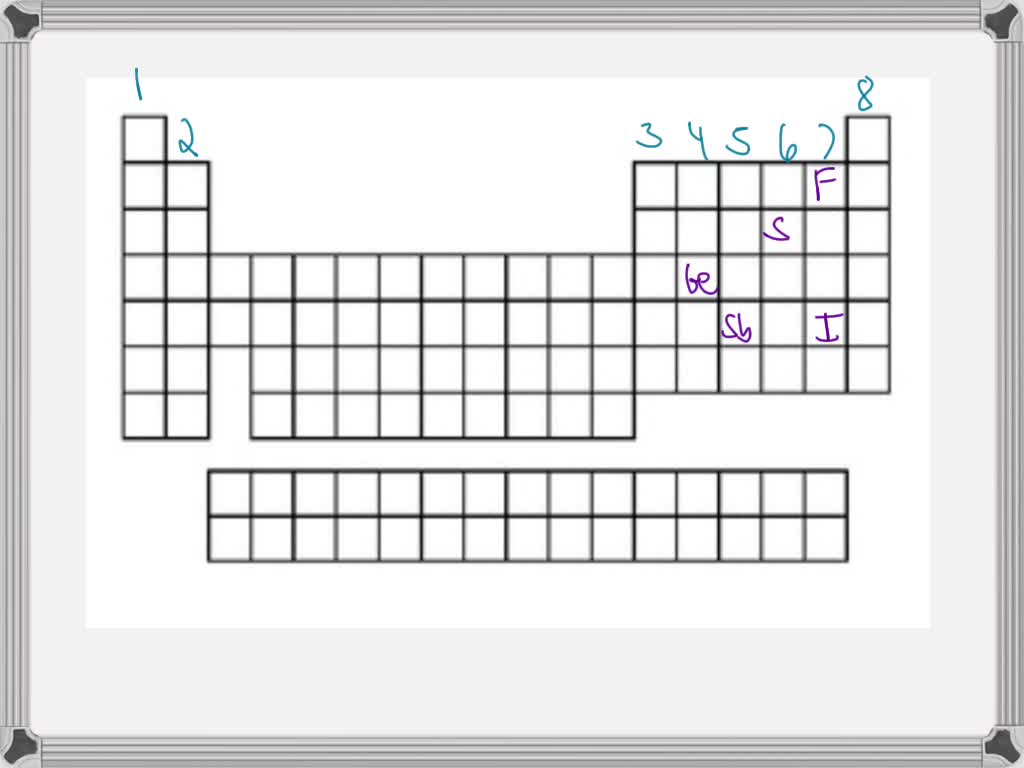
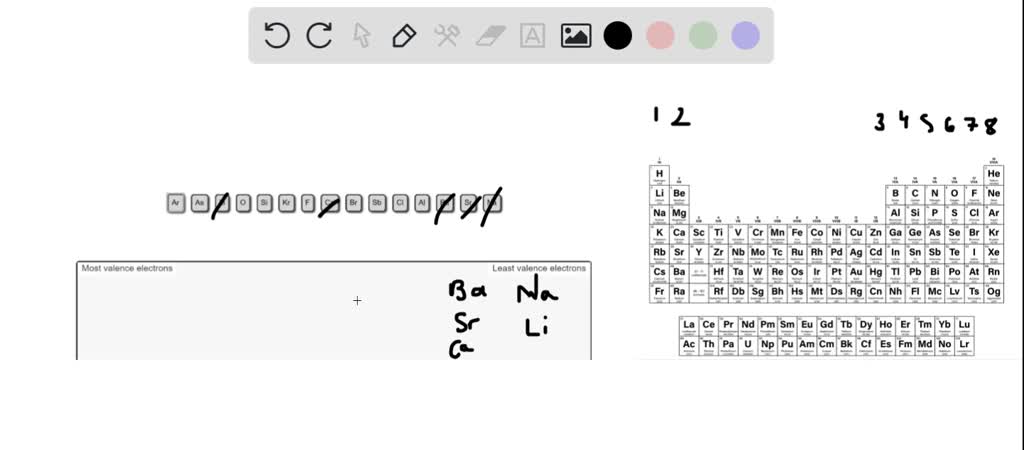
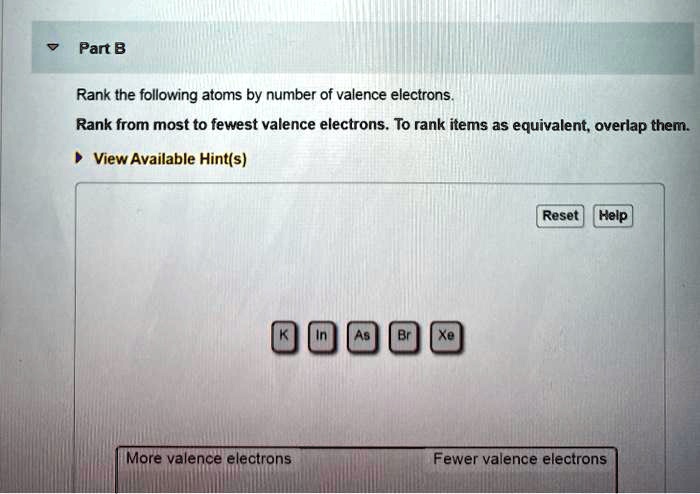
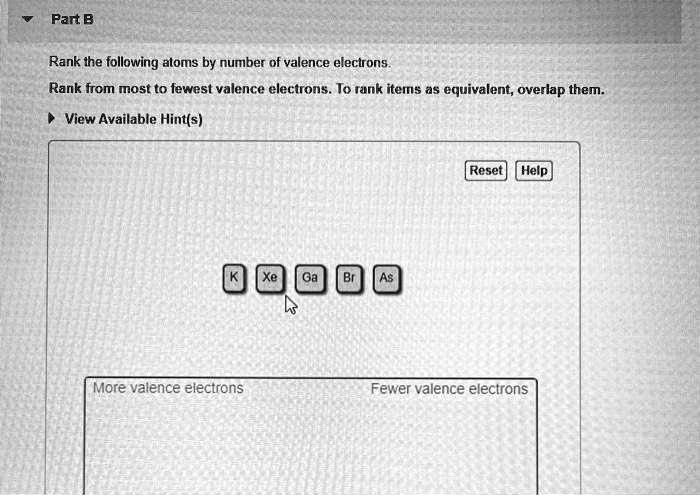
Rank From Most To Fewest Valence Electrons

Hey there, science explorers and curious minds! Today, we're diving headfirst into a super cool, yet totally not scary, topic: the "Valence Electron Olympics!" Think of it like a popularity contest for atoms, where the prize is… well, getting to mingle and make awesome stuff happen. We're going to rank some of the most famous players on the periodic table, from those who are practically drowning in their outer electrons to the ones who are a bit more… selective.
Imagine atoms are like little parties. The valence electrons are the guests hanging out on the dance floor, the outermost layer. These are the VIPs, the ones who get to interact with other atoms and create all the magic of chemistry. More valence electrons? More potential for fabulous interactions!
So, who's bringing the biggest crowd to the party? Let's get this grand ranking underway, starting with our undisputed champions of electron abundance!
Must Read
The Champions: Drowning in "Dance Floor Guests"!
At the very top of our list, practically bursting with guests, we have the Noble Gases. These guys are the ultimate introverts, but in the coolest way possible. They've got a full house of 8 valence electrons (except for Helium, which is perfectly content with 2). It's like they've already thrown the most epic party and are now just chilling, fully satisfied.
Think of them as the celebrities who've already signed every autograph and taken every selfie. They're so complete, they rarely feel the need to mingle. They're the "contented connoisseurs" of the atomic world, perfectly happy in their own electron-rich bliss.
Leading the pack, or rather, exuding their electrons are elements like Neon and Argon. They're like the friend who shows up to every party with a perfect 8 guests already in tow. No need to invite anyone else, thank you very much!
The Close Seconds: The "Ready-to-Party" Crowd!
Just a hair behind our noble gas champions are the Halogens. These guys are the life of the party, always looking for just one more dance partner to complete their set. They typically have 7 valence electrons, so close to that magical 8!
Imagine them as the enthusiastic host who's got almost all their decorations up, just missing that one final sparkle. They are incredibly eager to bond and form new connections. They're the go-getters, the ones who will absolutely make chemistry happen!
Leading this energetic group is Fluorine, the most electronegative element of them all. It's like the ultimate social butterfly, desperately seeking that eighth electron to achieve ultimate party perfection. Then comes Chlorine, also with 7 valence electrons, ready to jump into action and make some chemical magic.
"These elements are so close to being 'full,' they're practically vibrating with anticipation!"
The Powerhouses: The "Building Blocks" of Everything!
Next up, we have elements with 6 valence electrons. These are the master builders, the ones who can form chains and complex structures. They're like the party planners who have a solid foundation and are ready to add all sorts of fun activities.
Take Oxygen, for example. It's got 6 valence electrons and is absolutely crucial for life as we know it. It's like the generous host who can share their resources to create amazing things. It's always ready to form bonds, making it essential for everything from breathing to burning.
Then there's Sulfur, also with 6 valence electrons. It might not get as much press as Oxygen, but it's a vital component in proteins and has that distinctive, albeit sometimes pungent, aroma. These elements are the workhorses, making sure the party (and life) keeps going!
The Versatile Vanguards: The "Everything Makers"!
We’re moving along to elements with 5 valence electrons. These guys are also super important, capable of forming diverse and complex molecules. They’re like the organizers who can create intricate decorations and set up diverse entertainment options.

Think about Nitrogen, the most abundant gas in our atmosphere! It's got 5 valence electrons and is a fundamental building block for DNA and proteins. It’s the quiet organizer, always there, forming the backbone of so much!
Then there’s Phosphorus, another element with 5 valence electrons, crucial for our bones and energy transfer. These elements are the backbone of many biological processes, showing incredible versatility in their chemical relationships.
The Reliable Collaborators: The "Steady Joes"!
Now we're getting into the realm of elements with 4 valence electrons. These are the reliable friends, the ones who are always ready to share and form stable structures. They’re like the dependable guests who bring great snacks and are happy to help set up.
The absolute star here is Carbon! With its 4 valence electrons, it's the undisputed king of organic chemistry. It can link up with itself and other atoms in an almost infinite number of ways, forming the basis of all life on Earth. It’s the ultimate team player!
Then there's Silicon, also with 4 valence electrons. While Carbon rules the biological world, Silicon is the backbone of the technological one, found in sand and computer chips. These elements are incredibly foundational, building the very world around us.
The Eager Explorers: The "One-Bond Wonders"!
As we continue our electron count, we arrive at elements with 3 valence electrons. These guys are ready to get things started, forming foundational bonds. They’re like the friends who are always the first to suggest a game and get everyone involved.
Leading this group are the Boron group elements, like Boron itself. They are often found in compounds that have unique bonding properties. They are eager to connect and form the initial structure of many chemical reactions.
These elements are essential for creating networks and starting the complex assembly process. They’re the initial spark that gets the chemical party truly rolling.
The Humble Helpers: The "Two-Timer Team"!
Next on our list are elements with 2 valence electrons. These are the steady partners, often found in pairs or forming simple, strong connections. They’re like the best friends who always have each other's back, forming a solid duo.
The most famous of these are the Alkaline Earth Metals, like Beryllium and Magnesium. They are known for forming stable compounds and are found in everything from bones to alloys. They are the reliable backbone of many structures.
These elements are fantastic at forming ionic bonds, readily giving up their two electrons to achieve a more stable configuration. They are the dependable, straightforward members of the atomic community.
The Solitary Stars: The "One and Only"!
Finally, at the other end of our spectrum, we have elements with just 1 valence electron. These are the lone rangers, the highly reactive ones who are desperate to find a partner. They’re like the friend who’s always looking for someone to hang out with, eager for any interaction!
At the very forefront of this category are the Alkali Metals, like Lithium, Sodium, and Potassium. They are so eager to lose that single electron and achieve stability that they react quite vigorously with other elements. They are the ultimate charmers, always looking to make a connection!
These elements are the most reactive because they have that one electron they’re just dying to give away. They form the basis of many important chemical reactions and are essential for understanding atomic behavior.
And there you have it! Our fun-filled, playful ranking of atoms based on their "dance floor guests," their valence electrons. From the overflowing parties of the Noble Gases to the eager lone rangers of the Alkali Metals, every atom plays a vital role in the incredible, ongoing chemistry show that makes up our universe. Isn't science just the coolest?