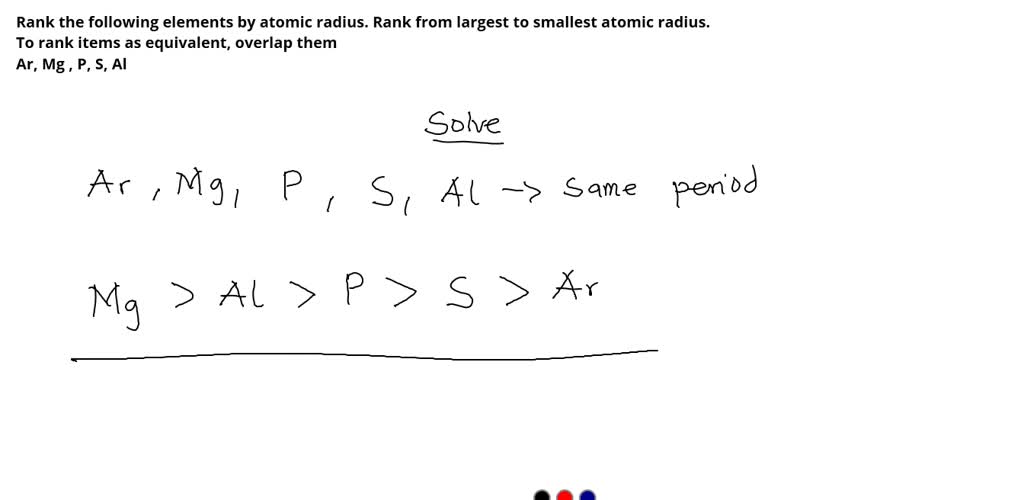
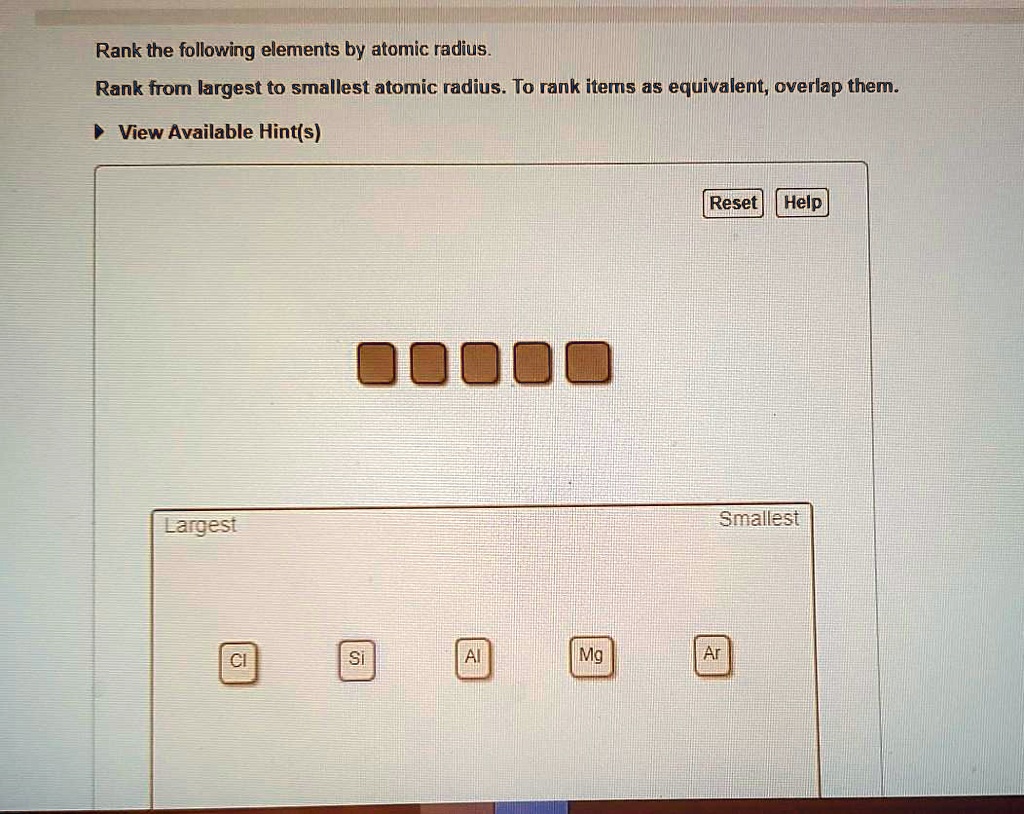
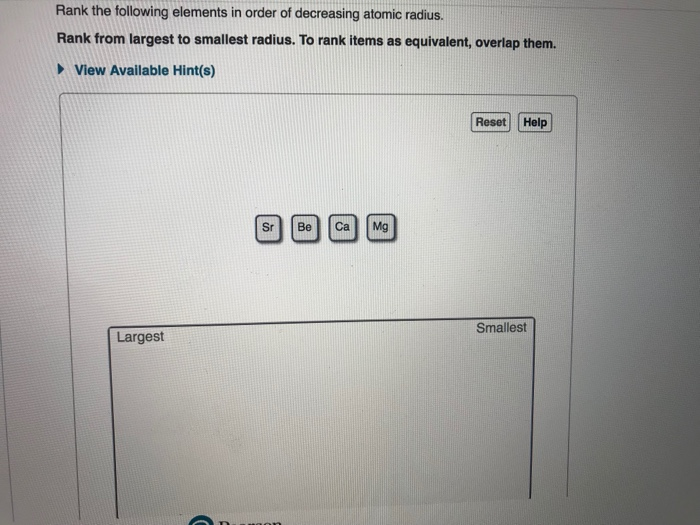
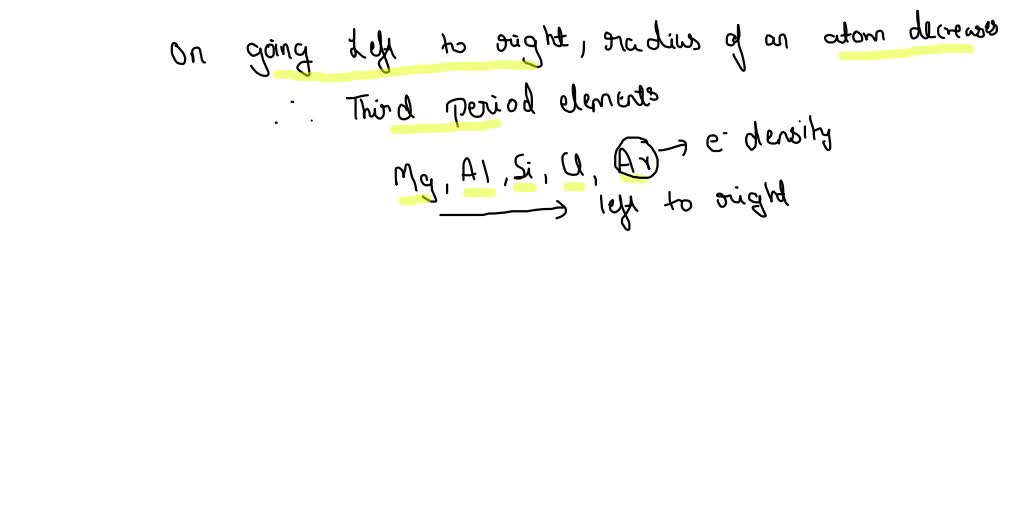Rank From Largest To Smallest Atomic Radius

Ever wondered if bigger is always better? Well, when it comes to the teeny-tiny building blocks of everything around us – atoms – size really does matter! Understanding how atomic size changes across the periodic table might sound like a lecture from your old chemistry teacher, but trust us, it's actually pretty fascinating. It’s like unlocking a secret code to how elements behave, why some are great at forming bonds and others are happy to go it alone. Plus, knowing this little tidbit can totally impress your friends at your next trivia night. So, let’s dive into the wonderfully weird world of atomic radius and explore how these minuscule marvels rank from largest to smallest!
So, what exactly is this “atomic radius” we’re talking about? Think of an atom as a miniature solar system. You've got the nucleus, a dense little sun in the center, and electrons, those speedy little planets, whizzing around. The atomic radius is essentially the distance from the center of the nucleus to the outermost edge of the electron cloud. It’s a way to measure the “size” of an atom. Now, you can’t exactly grab a ruler and measure an atom, so scientists have clever ways to figure this out, usually by looking at how atoms bond together. Imagine two atoms holding hands (forming a chemical bond); the atomic radius is like half the distance between their centers when they’re linked up.
Why is this concept so useful? Well, atomic radius is a fundamental property that helps us predict a whole bunch of other things about an element. For instance, it plays a big role in an element's electronegativity (how strongly an atom pulls on electrons in a bond) and its ionization energy (how much energy it takes to yank an electron away). Elements with larger atomic radii tend to have electrons further from the nucleus, making them easier to remove, and they also exert less pull on electrons in a bond. Conversely, smaller atoms have their electrons held much tighter. This understanding is crucial for chemists when they're designing new materials, synthesizing medicines, or figuring out how reactions will occur. It's the invisible hand guiding countless chemical processes.
Must Read
Now, let's get to the fun part: ranking these atomic sizes! The periodic table, that iconic chart of all known elements, isn't just a random arrangement. It's a masterpiece of organization, and the trends in atomic radius are beautifully displayed within it. If you were to look at the periodic table, you'd notice a distinct pattern.
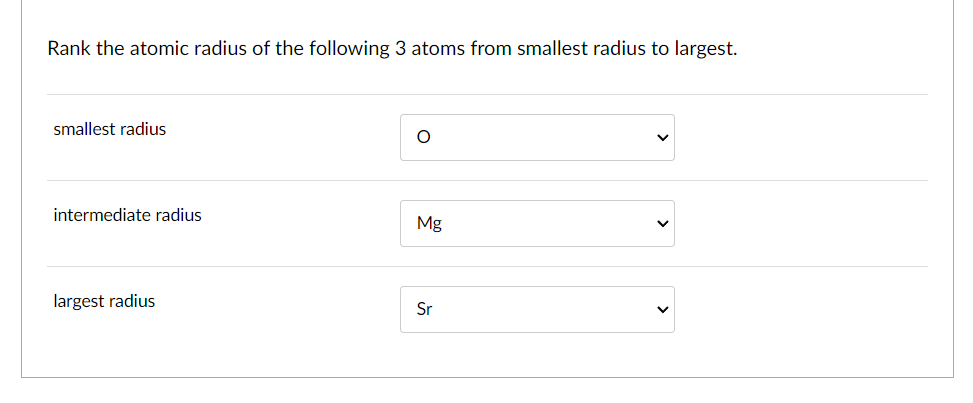
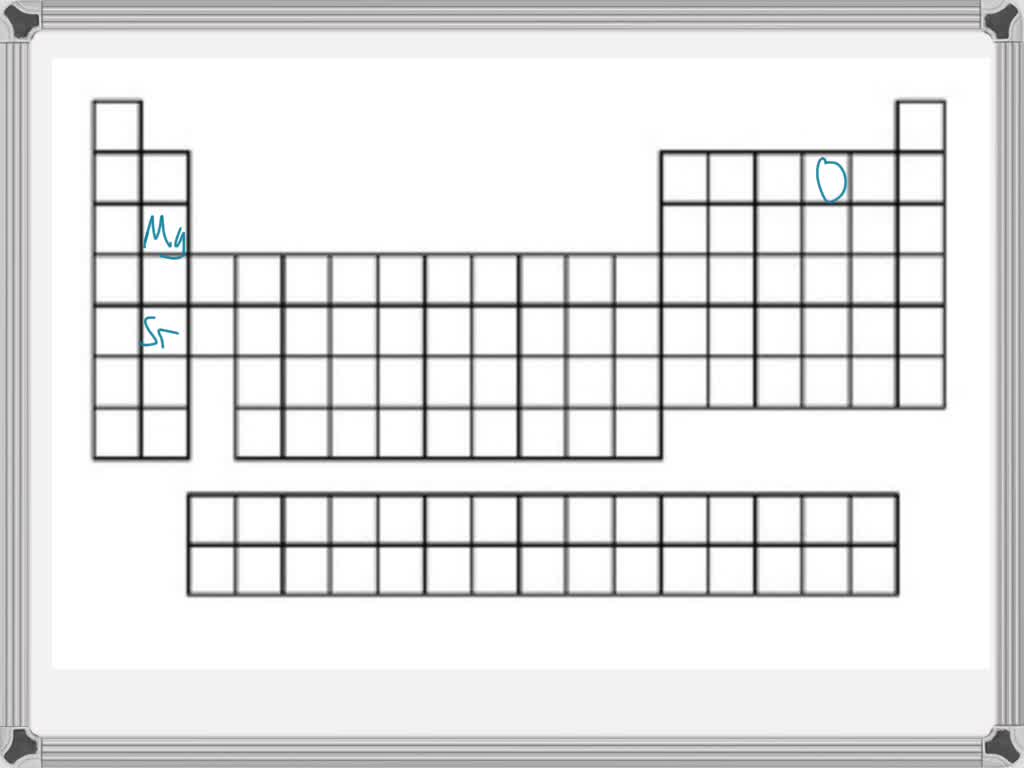
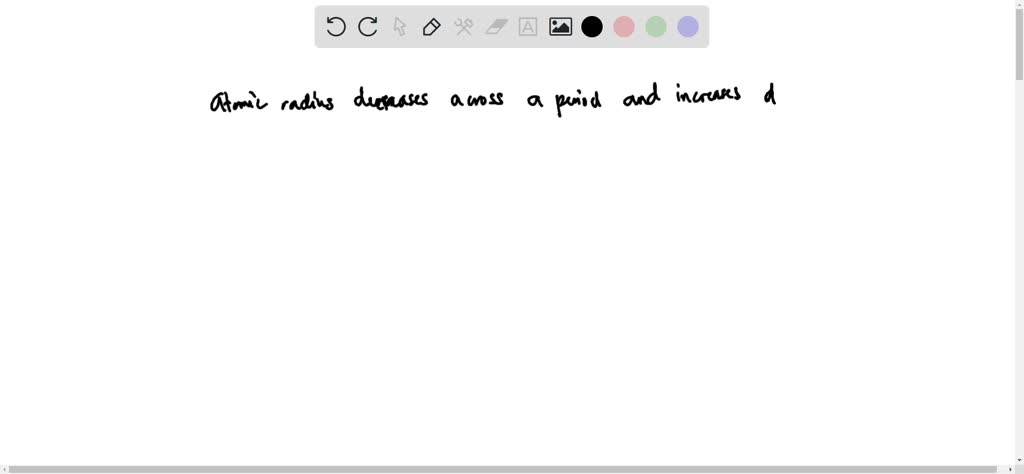
Generally speaking, as you move down a group (a vertical column) on the periodic table, the atomic radius increases. Why? Each new period (horizontal row) adds another electron shell, another layer of electron "orbit." Think of it like adding another ring to a tree trunk. The more rings, the wider the trunk. So, elements at the bottom of a group, like Francium or Cesium, are absolute giants in the atomic world. They have so many electron shells that their outermost electrons are quite far from the nucleus, making them the largest atoms you’ll find.

On the flip side, as you move across a period (a horizontal row) from left to right, the atomic radius generally decreases. This might seem counterintuitive at first. You’d think adding more protons to the nucleus and more electrons would make the atom bigger, right? Well, sort of, but the key is that all these new electrons are being added to the same outermost electron shell. The increasing number of positively charged protons in the nucleus exerts a stronger pull on all the electrons, including those in the outer shell. This stronger "nuclear charge" effectively squeezes the electron cloud, making the atom smaller. So, elements on the far left of a period, like Sodium or Potassium, are larger than elements on the far right, like Chlorine or Fluorine.
Putting it all together, if we were to rank elements from largest to smallest atomic radius, you’d find the true behemoths in the bottom-left corner of the periodic table. Elements like Francium (Fr) and Cesium (Cs) are the undisputed champions of atomic size. These alkali metals have very low ionization energies and are highly reactive precisely because their outermost electrons are so distant and weakly held.
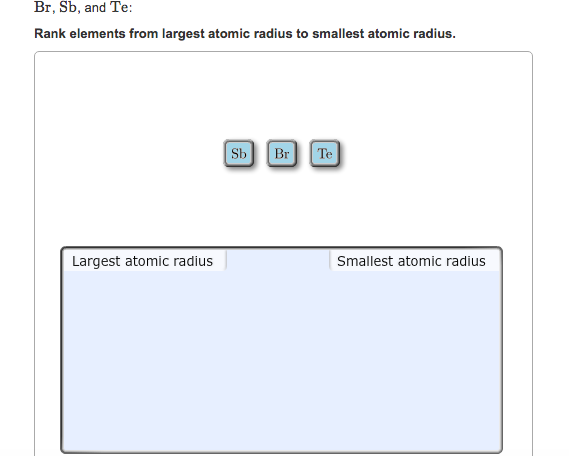
As you move up and to the right, the atomic sizes shrink dramatically. The noble gases, sitting in the far-right column, are generally quite small for their respective periods. And at the very top-right, you’ll find the tiniest atoms. Helium (He) is a prime example of a very small atom. Its two electrons are held very tightly by the nucleus in the very first electron shell. Other elements like Neon (Ne) and Hydrogen (H) also claim spots among the smallest atoms.
It’s important to note that these are general trends, and there are some exceptions and nuances, especially with the transition metals in the middle of the periodic table. However, for the most part, this left-to-right decrease and top-to-bottom increase is your golden ticket to understanding atomic radius.
So, next time you see the periodic table, don’t just see a jumble of letters and numbers. See it as a map of atomic sizes! You can visualize the giants at the bottom left and the tiny tots at the top right. This simple understanding of atomic radius is a powerful tool, unlocking a deeper appreciation for the structure and behavior of the elements that make up our universe. It’s a testament to the elegant order hidden within what might seem like chaos, all thanks to the subtle dance of protons and electrons!