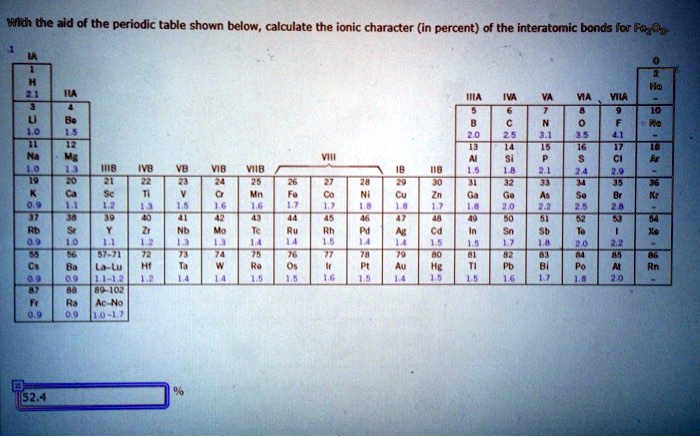
Percent Ionic Character Of Interatomic Bonds

Hey there, you! Grab a coffee, settle in. We're gonna chat about something that sounds super science-y but is actually… well, kinda cool. We're talking about percent ionic character. Yeah, I know, sounds like something you’d find in a dusty textbook. But stick with me, it’s like figuring out how “sticky” atoms are to each other. You know, those little guys that make up everything?
So, imagine two atoms are hanging out, right? They’re like, best buds, or maybe just acquaintances. They’ve got these things called electrons. Think of them as their social media followers. And sometimes, they decide to share these followers. Or, you know, one of them just straight-up steals them. Classic!
This whole sharing or stealing thing? That's what makes bonds between atoms. We’ve got two main types, the ones that are all about sharing is caring – those are covalent bonds. Super chill, right? Like, "Hey, let's both have this electron, cool?"
Must Read
Then there are the ones where one atom is like, "Mine! All mine!" and basically snatches the electron(s) from the other. That's where the ionic bond comes in. It’s a bit more dramatic, a bit more… charged. Get it? Charged? Because one atom ends up with a negative vibe (extra electrons) and the other a positive vibe (missing electrons). It's like a cosmic tug-of-war for electrons.
But here's the juicy part. It’s not always a clear-cut case of sharing or stealing. Most of the time, it's somewhere in the middle. Like, one atom really wants the electron, but the other one’s like, "Okay, okay, you can borrow it for a while. But don't scratch it!" This is where our friend, the percent ionic character, struts onto the stage.
Think of it as a sliding scale. On one end, you have a 100% covalent bond. Pure sharing. No drama. Like when you and your friend both contribute equally to a pizza. Everyone's happy. Or, theoretically, a bond between two identical atoms, like two hydrogen atoms. They're pretty much guaranteed to share nicely. No one has a major advantage.
On the other end, you’ve got a 0% ionic character bond. Wait, no, that’s not right. 0% ionic character means 100% covalent. Phew, glad we cleared that up. The other extreme is when one atom completely dominates the electron-sharing game. It’s so good at hogging the electrons that it’s practically an ionic bond. We’re talking about a bond that’s almost entirely ionic. Almost. Because true 100% ionic bonds are, like, super rare in the real world. Most things aren't that black and white, are they?

So, percent ionic character tells us how much of that electron-stealing, I-mean-sharing, vibe is happening in a bond. Is it mostly covalent with just a hint of ionic behavior? Or is it leaning heavily towards ionic, with just a smidgen of sharing left? It’s like judging a movie: is it a pure action flick, or does it have some romantic comedy elements? This number gives us the scoop.
How do we figure this number out, you ask? Well, it's not like we can just stick a tiny measuring tape on the electrons. Science has ways, though! It usually involves something called electronegativity. Ooh, fancy word! Don't let it scare you. Electronegativity is basically an atom's greed for electrons. How badly does it want them? Some atoms are like ravenous vacuum cleaners for electrons, while others are pretty laid-back. They're just chilling.
Imagine two atoms, A and B. Atom A has an electronegativity of, say, 3.0, and atom B has an electronegativity of 1.0. That’s a difference of 2.0. Big difference, right? This big difference means atom A is way more electronegative than atom B. It’s going to be a serious electron hog. So, the bond between them is going to have a high percent ionic character.
If atom C has an electronegativity of 2.5 and atom D has an electronegativity of 2.0, the difference is only 0.5. Much smaller difference. They're both pretty electron-greedy, or maybe not so greedy. They're closer in their electron-wanting desires. So, the bond between them will have a low percent ionic character. It'll be more on the covalent side.

There are charts for this stuff, you know. Tables of electronegativity values. They’re like the cheat sheets of the atomic world. Chemists love their charts. They’re like, "Oh, look, this element has a 3.9 electronegativity, and that one has a 0.9. That means… drama!"
Once you have the electronegativity difference, there are formulas. Don't worry, I'm not going to make you do calculus over coffee. But basically, they take that difference and spit out a percentage. That percentage is your percent ionic character. It’s like a report card for the bond. Is it an A+ ionic bond? Or a C- covalent bond with a slight ionic lean?
So, what does this actually mean in the real world? Why should you care if a bond is 70% ionic or 30% ionic? Well, it affects everything about the substance. Seriously, everything. Think about melting points, boiling points, how things dissolve, whether they conduct electricity. It's all tied to how these atoms are holding onto their electrons. It’s the unseen hand controlling the properties of matter!
Take salt, for example. Sodium chloride (NaCl). Sodium is, like, super eager to give away an electron. Chlorine is, like, "Oh yes, gimme!" Their electronegativity difference is pretty big. So, the bond in NaCl has a high percent ionic character. That’s why salt forms crystals, has a high melting point, and dissolves in water. It’s all about those charged ions hanging out together.
Now, think about water (H₂O). Oxygen is pretty electronegative, but hydrogen isn't super weak. The bonds between oxygen and hydrogen are polar covalent. That's a fancy way of saying they share electrons, but not equally. Oxygen pulls the electrons closer, making it slightly negative, and hydrogen slightly positive. It's not a full-on ionic bond, but it's definitely not a perfectly equal sharing either. It's got a significant, but not dominant, ionic character. This polarity is why water is such an amazing solvent and why it has those weird properties, like ice floating. Mind-blowing, right?
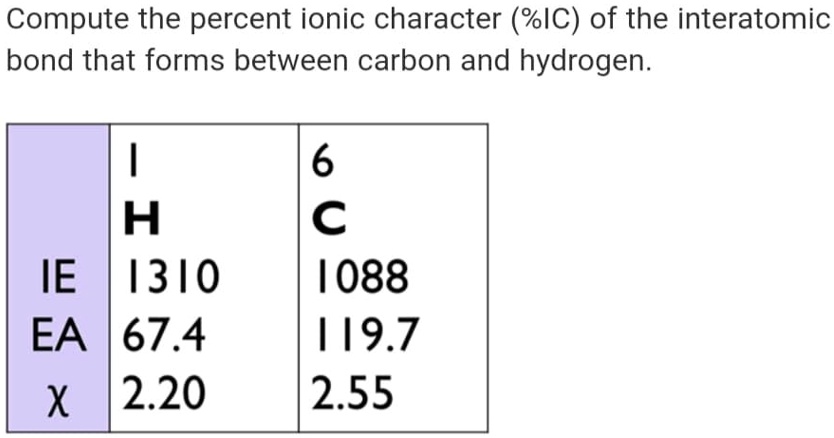
What about something like methane (CH₄)? Carbon and hydrogen have pretty similar electronegativities. The difference is small. So, the C-H bonds are very, very close to being purely covalent. Almost no ionic character. That’s why methane is a gas at room temperature, doesn't dissolve in water, and is generally pretty unreactive compared to, say, salt. It’s all down to how those electrons are behaving.
The percent ionic character essentially tells us about the polarity of the bond. A high percent ionic character means a highly polar bond. Think of it like a tiny little magnet. One end is positive, the other is negative. A low percent ionic character means a nonpolar or weakly polar bond. Much less of a magnetic pull.
It’s also a spectrum, remember? No bond is truly 100% ionic or 100% covalent. It’s always a mix. Even in what we call ionic compounds, there's often a tiny bit of electron sharing going on. And in covalent bonds, especially polar ones, there's a partial transfer of electrons. It's a continuum, and percent ionic character is our way of pinpointing where on that continuum a particular bond lies.

Chemists use this concept all the time. When they're designing new materials, predicting how a reaction will happen, or even just understanding why a substance behaves the way it does. It’s a fundamental building block for understanding chemical behavior. It's the secret sauce, the hidden architecture of molecules!
So, next time you hear someone talking about electronegativity or bond polarity, you can nod knowingly and think, "Ah, yes, they’re just trying to figure out how much of a diva that electron is!" It’s all about the tug-of-war, the sharing, the stealing, the electrifying dance of electrons between atoms. Pretty wild when you think about it, huh?
And the really cool thing? This isn't just some abstract theory. It has real-world consequences. It helps us create medicines, develop new fuels, build stronger materials. All because we figured out how to quantify the "stickiness" and "sharing-ness" of atomic bonds. It’s science making the world go ‘round, one electron at a time!
So, there you have it. Percent ionic character. It's not just a number; it's a story about how atoms interact, a tale of electron sharing (or not!), and the fundamental force that shapes the world around us. Who knew electrons could be so dramatic? Cheers to understanding the little things that make the big things happen!