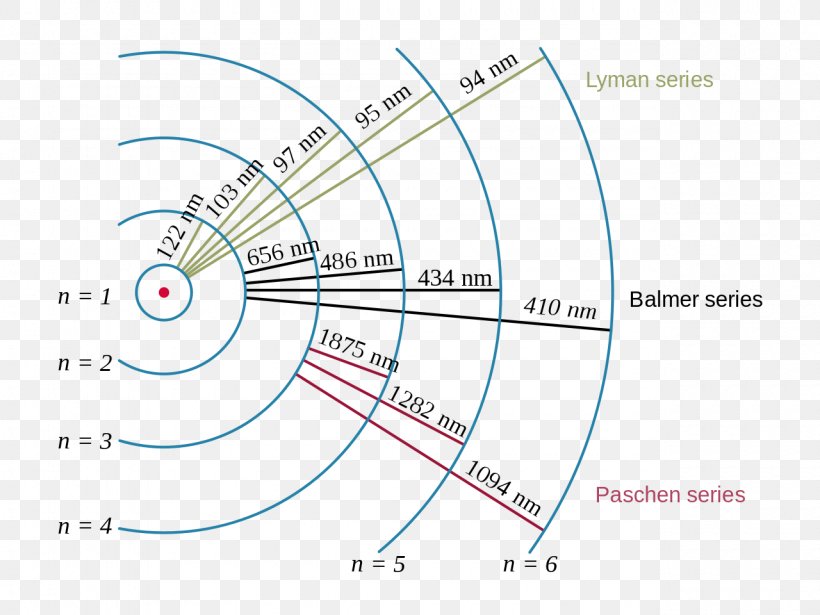
Partial Energy Level Diagram For Hydrogen

Have you ever wondered about the secret life of an atom? Specifically, the humble hydrogen atom, the simplest one out there? Well, scientists have found a super cool way to peek into its energy world. It's called a Partial Energy Level Diagram for Hydrogen.
Think of it like a super-exclusive club for electrons. These electrons in a hydrogen atom don't just hang out anywhere they please. They have to follow very strict rules about where they can be. These "allowed spots" are what we call energy levels.
So, what's so entertaining about a diagram of energy levels? It’s like a secret blueprint of how an atom dances! It shows us the special places an electron can be, like little balconies in a theater. Each balcony is at a different height, representing a different amount of energy.
Must Read
The lowest balcony, the one closest to the ground floor, is the most important. This is where the electron loves to be most of the time. It's the ground state, where the atom is feeling most relaxed and happy.
But what happens if we give this electron a little nudge? Like a tiny spark of energy? It can get excited and jump up to a higher balcony! This is like a concert ticket being upgraded. The electron is now in an excited state.
These excited states are pretty fun to think about. They are not stable for long. The electron wants to get back to its comfy ground state. So, it will eventually fall back down.
And when it falls back down, something really neat happens. It releases the extra energy it had. This energy usually comes out as a little packet of light. Imagine a tiny firework going off inside the atom!
The Partial Energy Level Diagram for Hydrogen shows us exactly which "balconies" the electron can jump between. It’s not just any jump; it’s specific jumps. Think of it like a pre-approved set of stairs.
The diagram usually starts with the ground state at the very bottom. Then, as you go up, you see the higher energy levels. Each level is labeled with a number. The lowest is n=1, the next is n=2, then n=3, and so on.

The spacing between these levels is also super important. They aren't evenly spaced, like steps on a regular staircase. The gaps get smaller and smaller as you go higher. It’s like the higher balconies are getting closer together.
This decreasing spacing is a key part of the diagram's charm. It hints at a hidden order in the atom's world. It’s a visual cue that this isn't just random. There's a beautiful logic at play.
When an electron jumps from a higher level (say, n=3) to a lower level (like n=1), it releases a specific amount of energy. This amount of energy corresponds to a specific color of light. It's like the atom is sending out a coded message in light!
The Partial Energy Level Diagram for Hydrogen helps us predict which colors of light will be emitted. If the electron jumps from n=2 to n=1, it emits one specific color. If it jumps from n=3 to n=1, it emits a different color.
This is where the "partial" in the name comes in. We're not looking at all the possible energy levels. We're often focusing on the lowest few, because those are the most common and easiest to observe. It's like looking at the first few rows of seats in a stadium.
The diagram often uses horizontal lines to represent these energy levels. The ground state (n=1) is the lowest line. Then comes n=2 above it, and n=3 above that.
Sometimes, you'll see arrows on the diagram. These arrows show the possible transitions, the jumps electrons can make. An upward arrow means the electron absorbed energy. A downward arrow means it emitted energy, usually as light.

Imagine an electron at n=1. If it absorbs the right amount of energy, it can jump to n=2. That's an upward arrow. Then, from n=2, it can fall back to n=1. That's a downward arrow, and a photon (light particle) is released!
What makes this diagram so entertaining is that it’s a window into the quantum world. It’s where things don't behave like they do in our everyday lives. Electrons can't just be anywhere. They have to pick from these special energy levels.
It’s like a cosmic rulebook for atoms. The Partial Energy Level Diagram for Hydrogen is a simplified version of this rulebook. It shows us the most important rules for hydrogen.
This simple diagram has a massive impact on science. It was a huge clue in understanding how light interacts with matter. It helped unlock the secrets of spectroscopy, which is used to analyze everything from distant stars to biological molecules.
Think about the colors of fireworks. Those vibrant hues are a direct result of electrons in atoms jumping between energy levels and emitting light. Hydrogen’s energy levels are the most basic building blocks of this phenomenon.
The diagram is also aesthetically pleasing in its own way. The clean lines, the clear labels, the visual representation of energy steps – it's like a minimalist piece of art. It’s elegant and informative.
It’s also quite addictive. Once you start looking at these diagrams, you realize how much information they pack. They are like secret codes waiting to be deciphered.

The fact that it’s for hydrogen, the simplest element, makes it even more special. It’s the foundational concept. If you understand hydrogen's energy levels, you’re well on your way to understanding more complex atoms.
It’s a great starting point for anyone curious about atoms. You don't need to be a rocket scientist to appreciate the beauty and logic of it. It’s accessible.
So, next time you hear about the Partial Energy Level Diagram for Hydrogen, don't just think of boring science. Think of electrons having a grand time jumping up and down energy balconies, releasing colorful light. It’s a tiny, energetic ballet happening at the heart of everything.
It’s a reminder that even the simplest things, like a single hydrogen atom, are incredibly complex and fascinating. The diagram is a key to unlocking that fascination. It’s a little peek into the amazing universe of quantum mechanics.
Give it a look! You might find yourself captivated by this atomic dance. It’s more entertaining than you might think.
The Partial Energy Level Diagram for Hydrogen is a visual symphony of atomic energy.
It’s where the magic of light and matter truly begins to reveal itself. A simple diagram, a profound story.
The elegance of the diagram lies in its ability to explain complex phenomena with a straightforward representation. It's a testament to the power of scientific visualization.

Think of it as the atom's to-do list for energy. What it can do, what it can't. What happens when it gets a little too much energy.
It’s a fundamental concept that underpins so much of modern physics and chemistry. And it all starts with hydrogen.
The diagram makes the abstract concept of energy levels tangible. You can see the steps, imagine the jumps.
It’s a bit like a musical score for an atom. Each line is a note, each jump is a melody. And the result is the spectrum of light we observe.
The "partial" aspect means we're getting a glimpse of the most important performances. The core energy transitions that define hydrogen's behavior.
It’s a gateway to understanding atomic spectra. The unique fingerprints of elements. All thanks to hydrogen's simple energy levels.
So, if you're looking for something intriguing and fundamental, dive into the world of the Partial Energy Level Diagram for Hydrogen. You might be surprised by how much fun you have exploring the energy levels of the universe's simplest atom.