Packing Efficiency Of Simple Cubic Unit Cell
Imagine you're at a music festival, and you've got to fit your entire life into a single backpack. Not just clothes, but snacks, a sleeping bag, maybe even a tiny disco ball. That's kind of what scientists are dealing with, but instead of disco balls, they're packing atoms!
Today, we're talking about a super simple way to pack these tiny building blocks of everything around us. Think of it as the most basic, no-frills packing strategy imaginable. We're diving into the world of the Simple Cubic Unit Cell.
The Humble Beginnings of Packing
When scientists first started thinking about how atoms arrange themselves, they looked for the easiest patterns. The simplest possible arrangement is like putting marbles in a perfectly square box, with one marble perfectly centered in each corner. Nothing fancy, just pure, unadulterated corner-stuffing.
Must Read
This basic arrangement is called the Simple Cubic Unit Cell. It's the absolute starter pack for understanding how solids are built at a microscopic level. Think of it as the elementary school of crystal structures.
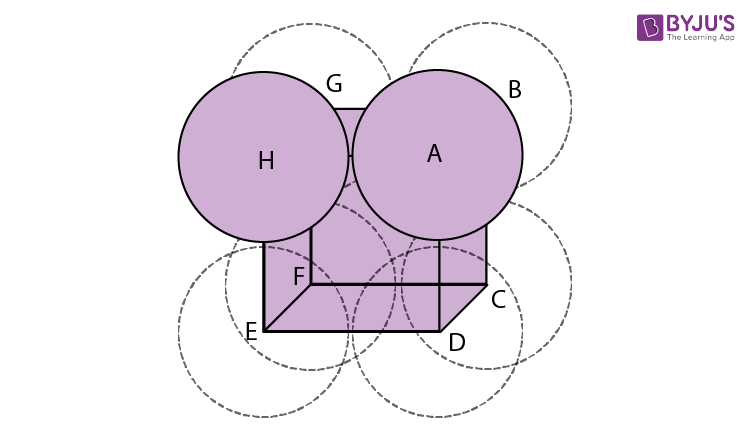
Meet the Corner Dwellers
In a Simple Cubic Unit Cell, the "things" being packed – let's call them our little atom-friends – are pretty shy. They only hang out at the very edges, right in the corners of our imaginary box. They don't really want to get too close to each other.
Each of these corner atom-friends is technically part of eight different boxes. It’s like having a single cookie that’s shared among eight friends. They each get a tiny crumb, but the whole cookie is technically "theirs."

This sharing is a big deal! It means that even though we see an atom in each corner, there's only a fraction of a whole atom actually inside any one box. It’s a community effort, really.
So, if you have eight corners, and each corner has 1/8th of an atom contributing to our box, how many full atoms do we have in total? Do the math, and you'll find we've got exactly one whole atom per unit cell. It's like a magic trick where eight tiny pieces add up to one whole.
The Big Question: How Much Space Do They Take Up?
Now for the million-dollar question: how efficiently are these atom-friends packed? Are they squished in like sardines in a can, or are they lounging around with plenty of personal space?
With the Simple Cubic Unit Cell, it's more of the latter. Our atom-friends are sitting at the corners, and there's quite a bit of empty space between them. Think of it like a sparsely decorated room where the furniture is only in the corners.
.jpg)
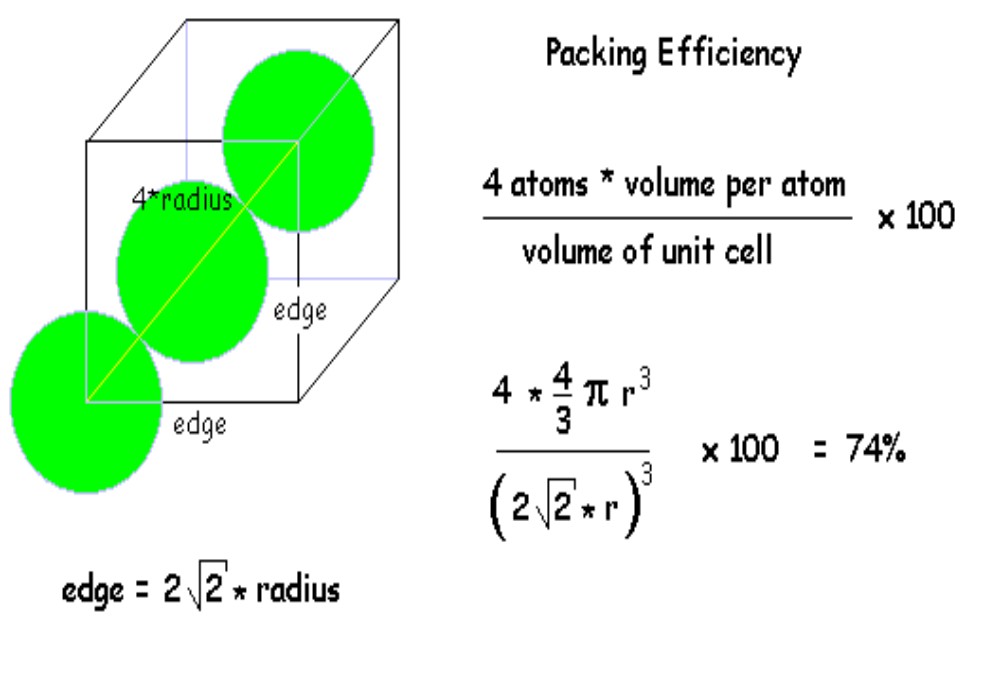
To figure out the packing efficiency, we compare the volume taken up by the atoms to the total volume of the box. It's like asking, "What percentage of your backpack is actually filled with your stuff, and what percentage is just air?"
The Not-So-Packed Result
When we do the calculations for the Simple Cubic Unit Cell, we find that only about 52.4% of the space is actually occupied by atoms. The rest, a whopping 47.6%, is just empty air!
That means more than half of the space in these little boxes is just… well, nothing. It’s like buying a fancy, oversized gift box for a tiny ring. It looks big, but a lot of it is just air.

This isn't the most efficient packing you can imagine. It’s like trying to fit an elephant into a Smart car – you're going to have a lot of leftover space. But hey, it’s a start!
Why This Matters (Even Though It's a Bit Loosey-Goosey)
You might be thinking, "So what? Why should I care if atoms are packed efficiently or not?" Well, it turns out this packing business is super important for understanding all sorts of things.
The way atoms pack together affects everything from the strength of a metal to how a crystal behaves. Even this loosely packed Simple Cubic Unit Cell gives us a baseline. It's the simplest scenario, and by understanding the simplest, we can then move on to more complex and efficient ways atoms can arrange themselves.
Think of it like learning to walk before you can run. The Simple Cubic Unit Cell is our first wobbly steps in the world of atomic packing. It shows us the most basic way things can be put together.
A Foundation for More Exciting Arrangements
While the Simple Cubic Unit Cell might seem a bit… spacious… for its inhabitants, it’s a crucial stepping stone. It’s the foundation upon which more complex and efficient crystal structures are built. Without this basic understanding, we wouldn’t be able to appreciate the cleverness of other packing arrangements.
It's like appreciating a simple melody before you can enjoy a symphony. This basic cube teaches us the ABCs of how atoms can organize themselves. And from these humble beginnings, we get to see some truly amazing and tightly packed structures.
So, next time you look at a crystal or a piece of metal, remember the little atom-friends in their simple cubic boxes. They might not be the most efficient packers, but they're the pioneers, the brave souls who showed us how it all begins. And in their own way, that’s pretty heartwarming.