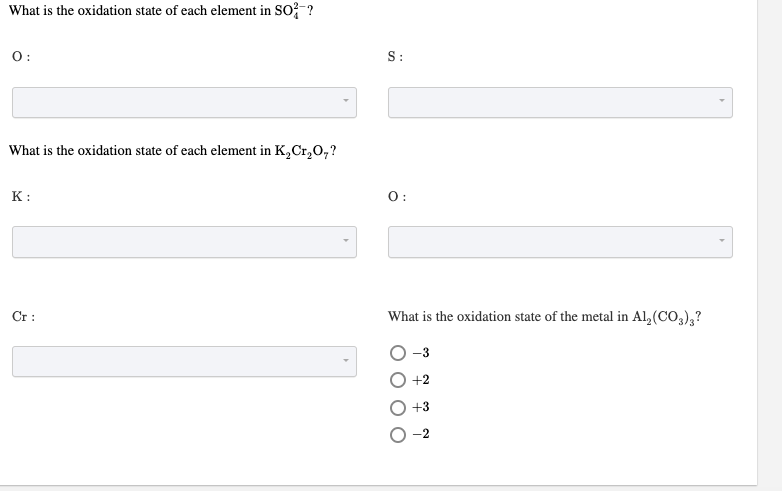
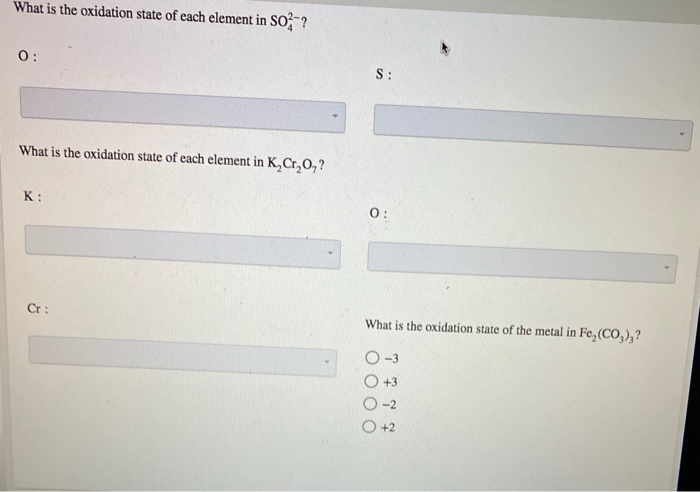
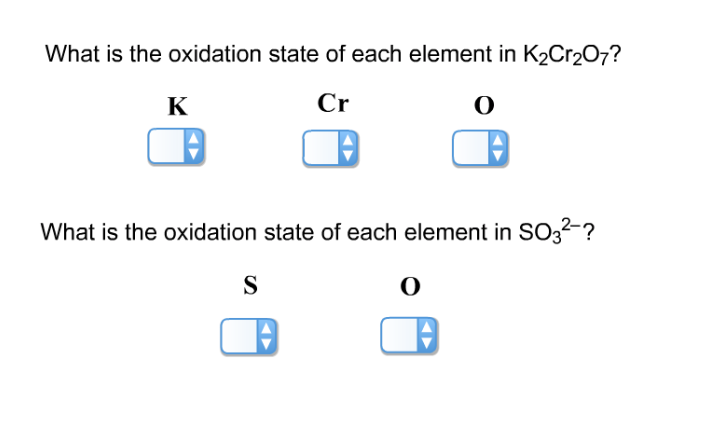
Oxidation State Of Each Element In K2cr2o7

Hey there, science curious folks! Ever looked at a label on a cleaning product, or maybe even a weird glowing rock at a museum, and wondered what all those little letters and numbers actually mean? Well, today we're going to dive into something that sounds a bit science-y, but trust me, it's as relatable as trying to figure out who ate the last cookie. We're talking about the oxidation state of elements in a compound called K₂Cr₂O₇. Don't let the fancy name scare you; think of it as figuring out who's the 'leader,' who's the 'follower,' and who's just 'going along for the ride' in a chemical family gathering.
Imagine your kitchen pantry. You've got your flour, your sugar, your baking soda. They all have their own jobs, right? Flour is the base, sugar is the sweet one, and baking soda… well, it’s the feisty one that makes things bubble up. In the same way, elements in a compound have different roles, and their oxidation state is like their 'role-playing score' for that particular party.
So, what's this K₂Cr₂O₇ thing? It’s Potassium Dichromate. Sounds like something you'd find in a mad scientist's lab, and honestly, it has some pretty cool (and sometimes dangerous) properties. Think of it as a super-duper strong stain remover, or something that makes fireworks extra… well, fiery. But for now, let's just focus on the individual characters in this chemical drama.
Must Read
The Case of the Missing Electrons: What is Oxidation State, Anyway?
Before we assign roles, let’s get a handle on what oxidation state actually is. In chemistry, we talk a lot about electrons – those tiny particles zipping around the nucleus of an atom. Sometimes, atoms are feeling generous and give away electrons. Other times, they're feeling needy and take electrons. It's a bit like a cosmic game of 'hot potato' with these little electron marbles.
Oxidation state is basically a number that tells us how many electrons an atom has gained or lost in a compound compared to its natural, neutral state. A positive number means it lost electrons (it's feeling a bit 'oxidized,' hence the name – like it got a tan from losing its shiny electrons!), and a negative number means it gained electrons (it's feeling 'reduced,' like it’s shrunk a bit by taking on more electron baggage).
Think of it like this: imagine you’re at a party, and you have a certain number of your favorite snacks. If you share some snacks with friends, your snack count goes down (you're ‘oxidized’). If your friends generously give you some of their snacks, your snack count goes up (you're ‘reduced’). The oxidation state is that final snack count compared to when you arrived.
Breaking Down K₂Cr₂O₇: Our Chemical Family Portrait
Now, let's get to our star compound: K₂Cr₂O₇. It’s made up of three different types of atoms: Potassium (K), Chromium (Cr), and Oxygen (O). We’ve got two Potassium atoms, two Chromium atoms, and seven Oxygen atoms. It’s like a mixed sports team with different player positions.
To figure out the oxidation state of each element, we rely on some tried-and-true rules. These are like the "common sense" rules of chemistry, the ones everyone agrees on so we don't end up with a chemical civil war. Think of them as the unwritten rules of a potluck: someone always brings the potato salad, and someone always brings the drinks.
Potassium (K): The Reliable 'Always Brings the Potato Salad' Guy
Let's start with Potassium (K). Potassium is in a special group of elements called alkali metals. These guys are like the super-friendly, always-ready-to-help neighbors. In almost every compound they form, alkali metals are super predictable. They always want to get rid of one electron to become stable, like someone who always brings the same dish to every party because they know it's a crowd-pleaser.
So, for Potassium in K₂Cr₂O₇, its oxidation state is always a nice, solid +1. It’s like the trusty friend who shows up on time, with the same reliable contribution. Two Potassium atoms? That’s two +1s, easy peasy. This guy is definitely not playing hard to get with his electrons; he’s practically handing them over with a smile.
Oxygen (O): The 'Takes What It Needs' Friend
Next up, we have Oxygen (O). Oxygen is one of the most common elements out there, and it’s a bit of a diva when it comes to electrons. It really likes to hoard them. In most compounds, Oxygen has an oxidation state of -2. It’s like that friend who’s always a bit peckish and tends to grab more than their fair share of the snacks. They’re not malicious, they just really like snacks.
Now, there are a few exceptions to Oxygen’s rule, like when it’s hanging out with fluorine (which is an even bigger electron hog, but that’s a story for another day!). But in our K₂Cr₂O₇ scenario, Oxygen is behaving its usual self. We have seven Oxygen atoms, so that’s seven instances of -2. That's a total of 7 * (-2) = -14. This Oxygen crew is collecting electrons like they’re going out of style!
Chromium (Cr): The 'Mystery Guest Who Might Bring Dessert'
And now, for the star of our little chemical show, Chromium (Cr)! This is where things get a bit more interesting. Unlike Potassium and Oxygen, Chromium can be a bit of a chameleon. Its oxidation state can change depending on who it’s hanging out with. It’s like that friend who sometimes brings a fancy homemade cake, and other times just grabs a bag of chips. It depends on the vibe of the party!
So, how do we figure out Chromium’s role in K₂Cr₂O₇? We use a neat trick. The total oxidation state of a neutral compound has to be zero. Think of it as the total bill at a restaurant – it has to balance out. We know the contributions from Potassium and Oxygen, so we can work backward to find Chromium’s share.
Let's do the math, but keep it chill:
We have: 2 Potassium atoms * (+1 each) = +2
We have: 7 Oxygen atoms * (-2 each) = -14
So, the total contribution from Potassium and Oxygen is +2 + (-14) = -12.
Since the whole compound (K₂Cr₂O₇) has to have a total charge of 0, the Chromium atoms have to make up the difference. That means the two Chromium atoms together must have a total charge of +12 to cancel out the -12 from the Oxygen and balance the scales. It’s like they're saying, "Okay, you guys took all the electrons, we'll put in the positive energy to make this whole thing work!"
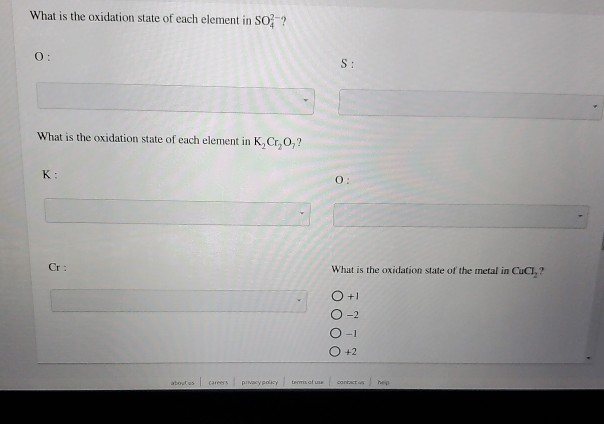
So, if two Chromium atoms add up to +12, then each individual Chromium atom must have an oxidation state of +6. 🎉
Chromium's Dual Personality: More Than One Trick Up Its Sleeve
And there you have it! In K₂Cr₂O₇, Chromium is rocking a +6 oxidation state. This is a pretty high oxidation state for Chromium, and it's what gives this compound its powerful oxidizing (electron-stealing) properties. It's like when a friend is super motivated and takes charge of organizing the entire event – they’re really working hard!
It’s fascinating to think that Chromium can have different oxidation states. Sometimes, you’ll find it as Chromium(II) (+2), Chromium(III) (+3), or even other numbers. It’s like that same friend showing up at different parties with different levels of enthusiasm and responsibility. Each oxidation state gives it a slightly different chemical personality, affecting how it interacts with other elements.
For instance, Chromium(III) is often found in more stable, less reactive compounds, like in some pigments that give things a greenish-brown color. Think of it as our friend being in a relaxed, chill mood. But Chromium(VI) (that’s our +6 guy!) is the high-energy, super-reactive version. It’s the one that’s like, "Let’s get this done, and let’s do it with force!" This is why K₂Cr₂O₇ is used in some industrial cleaning processes – it has the electron-stealing power to get the job done.
Why Should We Care About This Chemical Family Drama?
You might be thinking, "Okay, that's neat, but why should I care about the oxidation state of Chromium in some obscure chemical?" Well, understanding these oxidation states is super important for all sorts of things! It helps chemists predict how substances will react, how to make new materials, and even how to clean up environmental messes.
Think of it like knowing your friends' strengths and weaknesses. If you know your friend Bob is amazing at baking, you’ll ask him to bring the cake for the party. If you know Sarah is a whiz at fixing things, you’ll call her if your car breaks down. In chemistry, knowing the oxidation states tells us which elements are likely to give away electrons (become oxidized) and which are likely to take them (become reduced).
Potassium dichromate itself, K₂Cr₂O₇, is a classic example of a strong oxidizing agent. This means it loves to take electrons from other things. It’s like the ultimate electron vacuum cleaner! This property makes it useful in some analytical chemistry techniques and as a strong oxidant in organic synthesis. However, because it’s so reactive and its chromium is in a high oxidation state, it can also be quite toxic and is a known carcinogen. So, it's powerful, but you have to treat it with respect, much like you’d handle a very strong cleaning product – follow the instructions and wear your safety goggles!
The fact that Chromium can exist in multiple oxidation states is also a big deal. It’s crucial in biological processes (though typically in the less dangerous +3 state) and in the development of new technologies. It’s like learning that your friend can not only bake but also play a mean guitar – they have more hidden talents than you initially thought!
The Takeaway: It’s All About Balance and Roles
So, to wrap it all up, the oxidation states in K₂Cr₂O₇ are: Potassium (K) is +1, Oxygen (O) is -2, and Chromium (Cr) is a whopping +6.
It’s a beautiful dance of electron sharing and taking, all aimed at achieving a stable, neutral compound. Each element plays its part, from the ever-reliable Potassium to the electron-hoarding Oxygen, and the versatile Chromium. It’s a reminder that even in the seemingly complex world of chemistry, there are underlying rules and predictable behaviors, just like in our own everyday lives.
Next time you see a chemical formula, don't just see letters and numbers. Think of it as a little chemical family, each member with their own personality and role, all working together (or sometimes against each other!) to create something new. And remember, understanding these 'role-playing scores' can unlock a whole universe of chemical understanding. Happy calculating, and may your electron counts always be balanced!