Oxidation Number Of Carbon In Oxalic Acid

So, there I was, elbow-deep in a seemingly innocent bowl of spinach. You know, the kind that’s supposed to make you strong like Popeye, though mostly it just makes me feel like I’ve eaten a bunch of slightly bitter leaves. Anyway, I was reading the nutrition label (because who doesn’t do that when they’re really, really bored with their salad?) and I spotted something that caught my eye: “Oxalic acid.” My brain immediately did that thing where it conjures up weird chemistry trivia from the depths of my high school days. Oxalic acid. Sounds a bit… aggressive, doesn’t it? Like something you’d use to scrub a stubborn stain, not something that lives in my leafy greens. And then, a much more pressing question popped into my head: what’s the deal with the carbon in that stuff? Is it a good carbon? A bad carbon? Does it have a number? Oh, the drama!
And that, my friends, is how I found myself embarking on a casual, internet-fueled quest to understand the oxidation number of carbon in oxalic acid. Because, let’s be honest, who needs actual sleep when there are molecules to decipher, right? Especially when those molecules are hiding in your everyday food.
The Curious Case of Carbon's "Charge"
Okay, so before we dive headfirst into the oxalic acid pool, let’s have a little chat about these "oxidation numbers." Think of them like little temporary charges that atoms get assigned when they’re part of a molecule or an ion. They’re not real charges, mind you, not like the ones on electrons or protons. It’s more like a bookkeeping system to track how many electrons an atom has effectively gained or lost compared to its pure elemental state. Super useful for figuring out how chemical reactions are going to play out. It’s like knowing if your friend is owing you money (negative oxidation number) or if you owe them (positive oxidation number).
Must Read
Why do we even bother with this? Well, it helps us predict how things will react. Will this acid be a mild annoyance, or will it be a full-on chemical Hulk smash? Oxidation numbers are like the crystal ball for chemists. And for us mere mortals who occasionally stumble upon chemistry facts, it’s a fun little puzzle to solve.
Now, there are some pretty solid rules for assigning these numbers. Like, for instance, oxygen usually gets a -2, and hydrogen usually gets a +1 (unless it’s bonded to a metal, then it’s a bit of a rebel and goes to -1, but let’s not complicate things too much just yet!). The grand finale, the element that ties it all together, is the one we’re interested in: carbon. And carbon, bless its versatile heart, can be a bit of a chameleon when it comes to oxidation numbers. It can be positive, it can be negative, it can even be zero!
Oxalic Acid: The Star of Our Show
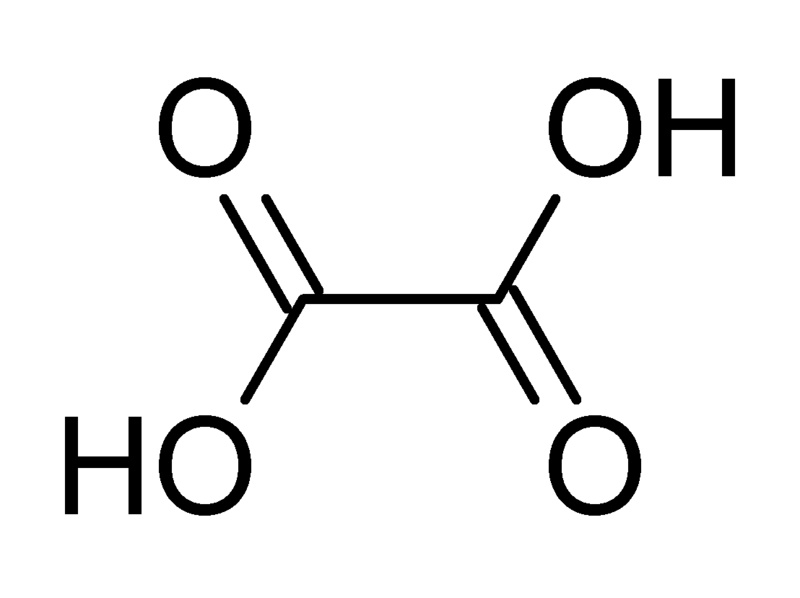
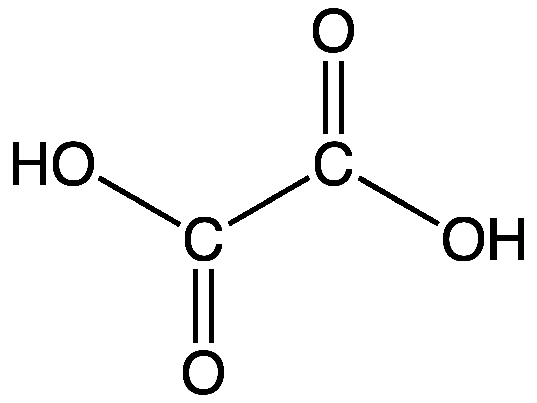
So, oxalic acid. Its chemical formula is C₂H₂O₄. Pretty straightforward, right? Two carbons, two hydrogens, four oxygens. Easy peasy. But the magic, or the chemical mystery, lies in how these atoms are arranged and what that means for the carbon. It’s not just a random jumble; it has a specific structure. The two carbon atoms are actually bonded to each other, and each carbon is also bonded to one oxygen atom via a double bond and to one oxygen atom via a single bond, which is then bonded to a hydrogen. Phew! Say that five times fast.
Here’s a little visual for you (imagine this with me, since I can’t actually draw it here): Think of it like a little molecule sandwich. The two carbons are the bread slices, and the oxygens and hydrogens are the fillings. But it’s a very organized sandwich, with specific connections.

Let’s break down how we figure out the oxidation number of the carbon atoms. We’re going to use our trusty rules. Remember the oxygen rule? Oxygen is almost always -2, except in rare cases like peroxides. In oxalic acid, we’ve got four oxygens, so that’s 4 * (-2) = -8 in total for the oxygens. Easy enough.
And the hydrogen? Hydrogen is usually +1. We have two hydrogens, so that’s 2 * (+1) = +2 for the hydrogens. Still with me? This is where it gets interesting.
Now, oxalic acid as a whole molecule has no overall charge. It’s neutral. That means the sum of all the oxidation numbers of the atoms within the molecule must equal zero. This is like the universe’s way of saying, “Okay, you’ve accounted for everything, nothing’s missing, nothing’s extra.”
So, we have: (Total oxidation number of carbons) + (Total oxidation number of hydrogens) + (Total oxidation number of oxygens) = 0.
Let’s plug in what we know: (Total oxidation number of carbons) + (+2) + (-8) = 0.
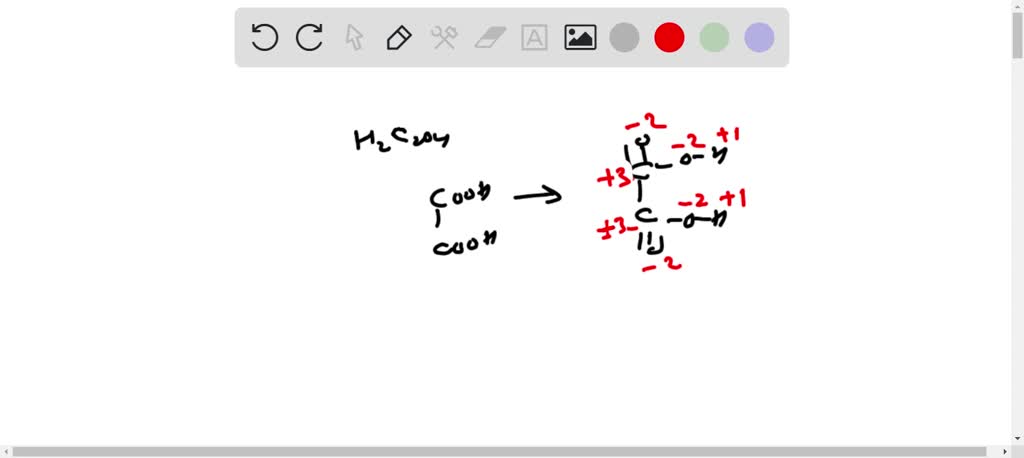
This simplifies to: (Total oxidation number of carbons) - 6 = 0.
Which means, the total oxidation number of the carbons is +6. But wait! There are two carbon atoms. So, if the total is +6, and we assume, because of the symmetrical structure, that both carbon atoms are doing the same kind of thing (chemically speaking), then we can divide that +6 by 2. And voilà!
Each carbon atom in oxalic acid has an oxidation number of +3. Pretty neat, huh?
Why Does This Matter? (Besides Proving You Remembered Chemistry)
So, we’ve established that the carbon in oxalic acid is rocking a +3. Why should you care? Well, besides being a fun little mental workout, understanding oxidation numbers helps us see how molecules behave. Carbon with a +3 oxidation number is in a relatively oxidized state. This means it's bonded to more electronegative atoms (like oxygen) and has effectively “lost” more electrons compared to, say, carbon in methane (CH₄), where it has an oxidation number of -4.

This +3 state makes oxalic acid a decent reducing agent. This means it can donate electrons to other substances in a chemical reaction, causing them to be reduced (gain electrons). It's like the giving friend in the electron exchange. You’ll often see oxalic acid used in titrations to determine the concentration of oxidizing agents. It’s a workhorse in the lab, and who knew it started with a salad?
Think about it: the carbon in your spinach, chilling out as part of oxalic acid, is ready to participate in redox reactions. It’s not just sitting there being green; it’s got chemical potential! It’s a little bit of a scientific thriller happening in your digestive system, but don't worry, your body is pretty good at handling it.
It’s also interesting to compare this to other carbon-containing compounds. Carbon in carbon dioxide (CO₂) has an oxidation number of +4. Carbon in methane (CH₄) has an oxidation number of -4. And carbon in carbon monoxide (CO) has an oxidation number of +2. See the range? Carbon is a true master of disguise when it comes to oxidation states. It’s like the James Bond of the periodic table, able to adopt so many different personas!
The +3 for oxalic acid is right there in the middle of its typical range. This implies a certain level of reactivity, but not extreme. It’s willing to play ball in chemical reactions, but it’s not going to be an uncontrolled explosion.
The "Trick" to the Trick Question
Sometimes, when you’re looking at organic molecules with multiple carbons, you might be tempted to just apply the rules to the whole molecule and get an average. But here’s the cheeky part, the little twist that keeps chemistry interesting: not all carbons in a molecule necessarily have the same oxidation number. Sometimes they can be different!
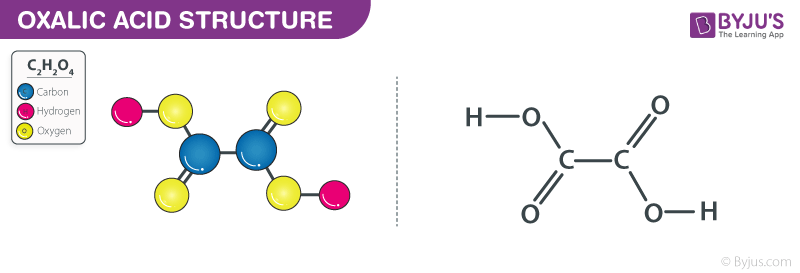
However, in the case of oxalic acid, the molecule is symmetrical. The two carbon atoms are in chemically equivalent environments. They are bonded to the same types of atoms in the same kinds of ways. This symmetry is why we can confidently say that each carbon atom has the same oxidation number, and that the average oxidation number is indeed the oxidation number for each individual carbon. It's a nice, clean situation.
If the molecule were more complex, or if the carbons were bonded differently, we’d have to be more careful. We might need to look at which atoms each specific carbon is bonded to and how many bonds it has to more electronegative atoms. This is where things can get a little… hairy. But for oxalic acid, we’re golden.
So, next time you’re enjoying your kale, or perhaps your rhubarb (another oxalic acid heavyweight!), you can impress your friends with your newfound knowledge. You can casually mention, “Ah yes, the carbon in this delectable dish is currently sporting an oxidation number of +3, making it a rather versatile player in the world of redox reactions.” They’ll either be utterly fascinated or slowly back away. Either way, you’ve won!
It’s these little details, these numerical assignments that unlock the behavior of molecules, that make chemistry so captivating. It’s not just about memorizing formulas; it’s about understanding the underlying principles that govern how matter interacts. And who knew that a simple dietary component could lead us down such an interesting chemical rabbit hole?
The journey from a spinach salad to the intricate world of oxidation numbers is a testament to how interconnected everything is, even at the molecular level. It’s a reminder that even the most mundane aspects of our lives can hold fascinating scientific stories, just waiting to be discovered. So, keep your eyes peeled, your curiosity piqued, and your chemistry knowledge sharp. You never know when a salad will inspire your next great scientific revelation!