Nonmetal That Gains 3 Electrons To Be Stable
+and+Fill+Their+Outer+Shell..jpg)
Hey there, science curious folks! Ever feel like you're just missing something to feel complete? Like, a couple of extra hands, or maybe a really good playlist to get you through the day? Well, guess what? In the wild and wacky world of chemistry, some elements feel the exact same way! And today, we’re going to chat about one in particular, a nonmetal with a bit of a thing for collecting. Yep, we’re talking about the element that’s all about gaining three electrons to achieve peak happiness – that glorious state of being “stable.” Isn't that just the cutest little goal?
Now, before you picture little electrons knocking on an atom’s door with a friendly “Trick or treat!”, let’s get a tiny bit technical. Atoms, you see, are like tiny solar systems. They have a nucleus at the center (that’s the sun!) and electrons whizzing around in orbits (those are the planets!). The number of electrons an atom has is super important. It’s like the outfit it’s wearing to the cosmic party. And just like we all want to look our best, atoms have a strong desire to have a full outer shell of electrons. Think of it as their ultimate fashion statement, their dream outfit!
Why three electrons, you ask? Ah, that’s the magic number for this particular nonmetal. Imagine its outer shell is like a cozy apartment with just a few pieces of furniture. It’s comfortable, sure, but it’s not quite the penthouse suite it craves. It needs three more electron-shaped cushions to feel perfectly complete, perfectly balanced, and, dare I say it, perfectly content. It’s like needing just a few more friends to make your favorite board game complete, or a couple more ingredients to bake that perfect batch of cookies.
Must Read
So, what’s the big deal about being stable? Well, stability in the atomic world is kind of like achieving nirvana. When an atom has a full outer electron shell, it’s super happy. It doesn’t need to go looking for more electrons, and it’s not so eager to give any away. It’s just… chilling. And when an element is stable, it tends to be a lot less reactive. Think of it as being in a state of zen. No drama, no frantic electron-swapping. Just pure, unadulterated peace. Wouldn’t that be nice for us humans sometimes?
This desire for three electrons is a key characteristic of elements belonging to a specific group on the periodic table. Without giving away the whole secret just yet (gotta keep some mystery!), these are elements you’ve probably encountered in everyday life, even if you didn’t realize their electron-gathering aspirations. They’re not usually the flashy, explosive types you might see in sci-fi movies, but they’re the workhorses, the quiet achievers of the chemical world.
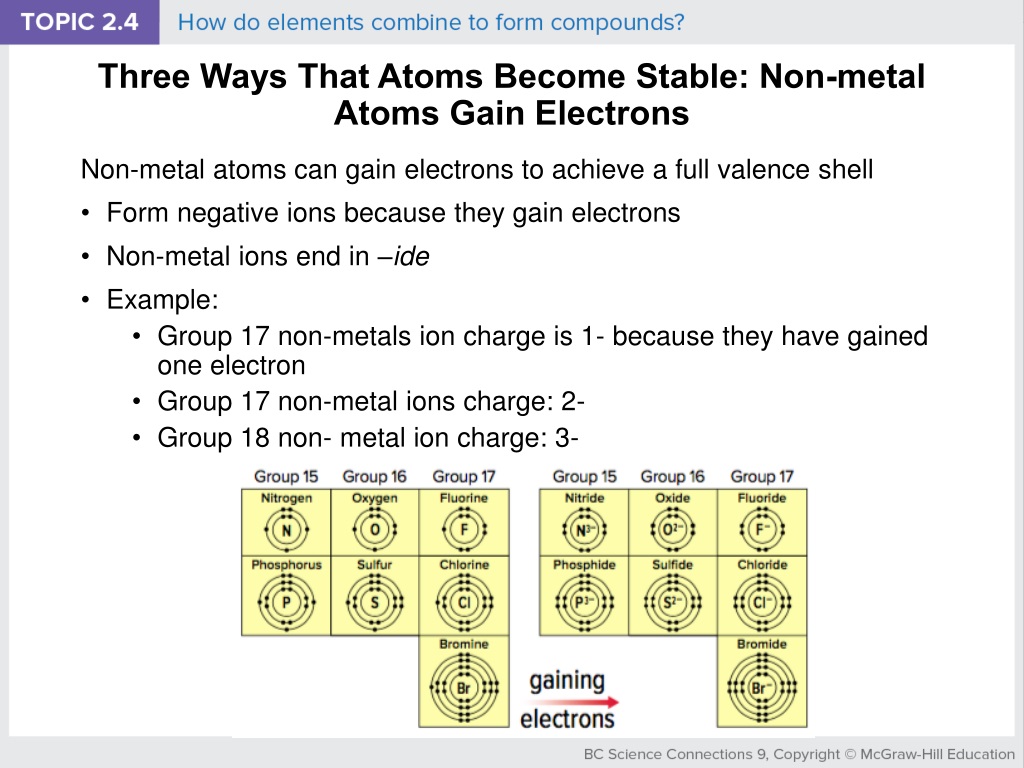
Let’s think about the implications of this electron-grabbing. When an atom needs to gain electrons, it often does so by forming bonds with other atoms. This is how everything around us is formed! Your water bottle? Made of bonded atoms. The air you breathe? Yep, bonded atoms. Even the delicious sandwich you might be planning for lunch is a marvel of atomic bonding. And our three-electron-collecting friend plays a crucial role in forming all sorts of interesting compounds. It’s like it’s the helpful neighbor who always has an extra cup of sugar to lend, and in return, helps create something amazing!
Imagine this: this nonmetal is like that friend who’s always up for trying a new recipe. It sees a gap, a need for three more electrons, and it actively goes out and finds them. This isn't a passive waiting game; it's a proactive pursuit of completeness. This can-do attitude is truly inspiring, don't you think? It’s a reminder that sometimes, to achieve what you want, you have to reach out and grab it (or, in this case, attract it!).

And the beauty of it is, this desire for three electrons allows these elements to form compounds that are essential for life. Think about it. Without these fundamental building blocks and their unique ways of interacting, we wouldn’t have the complex molecules that make us… well, us! It's a beautiful dance of attraction and stability, all driven by a simple desire for a full outer shell. Who knew that a tiny electron deficiency could lead to such grand outcomes?
This element, with its quest for three extra electrons, helps create materials that are both useful and fascinating. They can be found in things that help conduct electricity, or in substances that are used to make things stronger, or even in components that help us see the world around us more clearly. It’s a testament to how fundamental chemical principles can have a profound impact on our daily lives, often in ways we don’t even stop to consider.

So, next time you’re looking at something, anything, consider the atoms that brought it into being. Think about the elements that might have been a little ‘electron-short’ and went out and found their missing pieces to achieve that perfect, stable state. It’s a little bit like our own journey through life, isn’t it? We’re constantly learning, growing, and sometimes, reaching out to fill those gaps within ourselves, striving for our own kind of stability and completeness.
Isn't that a wonderfully uplifting thought? That the very building blocks of the universe are driven by a desire for balance and fulfillment? This nonmetal, with its specific need for three electrons, is a perfect example of how even the smallest, most fundamental processes can lead to incredible complexity and utility. It’s a little bit of science magic happening all around us, all the time!
So, I encourage you, dear reader, to let this little tidbit spark your curiosity! Don’t stop here. Dive deeper! Explore the periodic table. Learn about the amazing personalities of each element. You might just discover that chemistry isn’t just about complicated formulas; it’s about stories of atoms striving for their best selves, and in doing so, creating the vibrant, diverse, and utterly fascinating world we live in. Isn't that a fantastic adventure to embark on? Happy exploring!





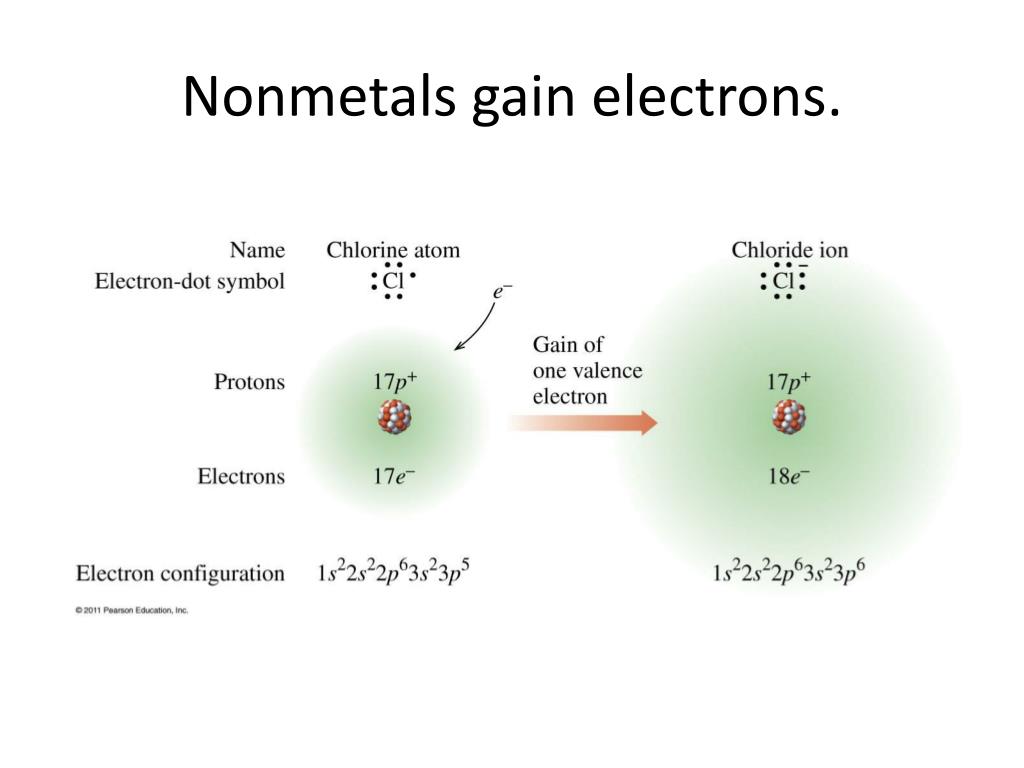
.+gain+electrons+to+achieve+stable+octet..jpg)