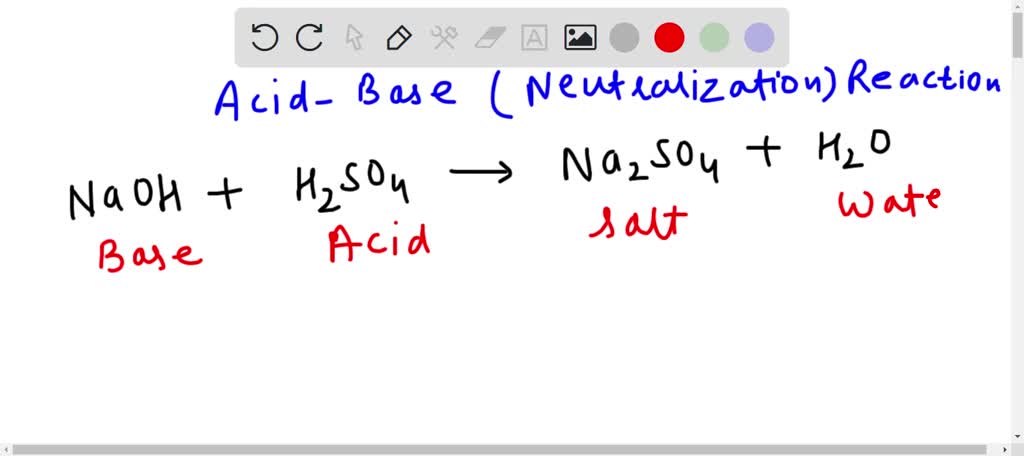
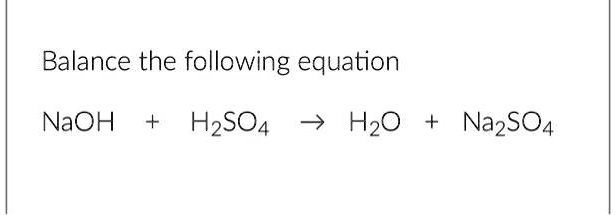
Naoh H2so4 Na2so4 H2o Balance The Equation
Hey there, science enthusiast (or, you know, someone who stumbled onto this page looking for literally anything else to do)! Ever feel like you’re starring in a chemistry play, and all the actors are just… letters? Yeah, me too. Today, we’re diving into a little chemical drama with a cast of characters that sounds like a secret agent’s code: Na, H2SO4, Na2SO4, and H2O. Don’t worry, there’s no need to break out the spy gear. This is going to be way less about espionage and way more about making sure everyone on stage gets an equal spotlight. We're talking about balancing chemical equations, my friends, and it’s not as scary as it sounds. Think of it like a potluck dinner – everyone needs to bring the same number of goodies to the party.
So, what’s the deal with these fancy symbols? Let’s break them down real quick. Na, that’s our pal Sodium. You know, the stuff in salt that makes your fries taste amazing? Yep, that’s sodium. Then we have H2SO4. This one’s a bit more of a heavy hitter. It’s Sulfuric Acid. Handle with care, it’s not exactly a gentle hug. On the other side of the equation, we’ve got Na2SO4. This is Sodium Sulfate. It’s like a calmer, more relaxed version of its acid cousin. And finally, the ever-reliable H2O. Yep, it’s water! The universal solvent, the drink of champions, and the reason we don’t spontaneously combust. Pretty straightforward, right?
Now, the equation itself looks like this: Na + H2SO4 → Na2SO4 + H2O. See that arrow? That’s not just a fancy arrow pointing the way. That’s the “transforms into” arrow. It’s like saying, “What happens when Sodium decides to hang out with Sulfuric Acid?” Well, things get interesting. But before we get too excited about the chemical makeover, we need to make sure our numbers add up. This is where the magic of balancing comes in.
Must Read
Why do we even bother balancing equations? Good question! It all boils down to something called the Law of Conservation of Mass. In simple terms, it means that matter (you know, stuff) can’t just appear out of thin air, and it can’t just vanish into the abyss. In a chemical reaction, the atoms (the tiny building blocks of everything) just rearrange themselves. They don't disappear. So, if you start with, say, 10 apples on one side of a table, and you rearrange them into fruit salad, you’re still going to have 10 apples in that fruit salad. You don’t suddenly get 12 apples, nor do you have only 7. That would be weird, and frankly, a little suspicious. The same goes for our chemical friends.
So, let’s count the atoms on each side of our equation: Na + H2SO4 → Na2SO4 + H2O. Think of the arrow as the middle of a seesaw. We want the weight on both sides to be exactly the same. On the left side (the reactants, for the fancy folks), we have:
- Na: Just one little sodium atom.
- H: Two hydrogen atoms (thanks to that handy little ‘2’ subscript!).
- S: One sulfur atom.
- O: Four oxygen atoms (again, that ‘4’ is our friend).
Now, let’s hop over to the right side (the products, where the magic happens):
- Na: Uh oh. We have two sodium atoms here (the ‘2’ next to Na2SO4). That’s already a mismatch!
- H: Two hydrogen atoms. This side’s looking okay for hydrogen… for now.
- S: One sulfur atom. Still looking good on sulfur.
- O: Four oxygen atoms. And oxygen is keeping pace!
See the problem? Our sodium is out of whack. We have 1 sodium on the left and 2 on the right. That’s like bringing one cookie to a party where everyone else brought two. Not fair! We need to even the playing field. How do we do that?
This is where we get to play with coefficients. Coefficients are those big numbers that go in front of the chemical formulas. They’re like multipliers. If you see a ‘2’ in front of ‘Na’, it means you have 2 * Na. It’s like saying, “Okay, you only brought one sodium, but the party needs two! Let’s get you a buddy!”
So, to get our sodium atoms to match, we need to put a ‘2’ in front of the Na on the left side. Our equation now looks like this: 2Na + H2SO4 → Na2SO4 + H2O.
Let’s recount! On the left side:

- Na: Now we have 2 sodium atoms (thanks to our new coefficient!).
- H: Still 2 hydrogen atoms.
- S: Still 1 sulfur atom.
- O: Still 4 oxygen atoms.
And on the right side:
- Na: We have 2 sodium atoms.
- H: Still 2 hydrogen atoms.
- S: Still 1 sulfur atom.
- O: Still 4 oxygen atoms.
And… voilà! Look at that! Everything matches. We have 2 Na on both sides, 2 H on both sides, 1 S on both sides, and 4 O on both sides. It’s a chemical party where everyone has the same number of party favors. Mission accomplished!
It’s really that simple. You just count the atoms of each element on both sides of the arrow and then adjust the coefficients until the numbers are equal. Easy peasy, lemon squeezy, right? Or in this case, maybe more like easy peasy, sulfuric acid-y?
Sometimes, these balancing acts can be a little more involved. You might have to add a coefficient to one compound, and then find that it messes up another atom, and you have to go back and adjust again. It’s a bit like a puzzle. You move one piece, and it shifts another. But the key is to be methodical. Keep a tally of your atoms on each side, and don’t be afraid to erase and rewrite coefficients. It’s all part of the process!
Let’s think about a slightly trickier scenario, just for practice. Imagine we had something like: Fe + O2 → Fe2O3. This represents iron reacting with oxygen to form iron (III) oxide. Anyone who’s seen a rusty old car knows this reaction well!
Let’s count:
Left side:

- Fe: 1
- O: 2
Right side:
- Fe: 2
- O: 3
Okay, so we have 1 Fe on the left and 2 Fe on the right. Let’s fix the iron first. We’ll put a ‘2’ in front of Fe on the left: 2Fe + O2 → Fe2O3.
New count:
Left side:
- Fe: 2
- O: 2
Right side:
- Fe: 2
- O: 3
Iron is balanced! Yay! But now, oxygen is still a pickle. We have 2 on the left and 3 on the right. Uh oh. This is where it gets a little more creative. We need a number that, when multiplied by 2 (on the left), gives us a number that can also be achieved when multiplied by 3 (on the right). Think common multiples. The least common multiple of 2 and 3 is 6.
So, we want 6 oxygen atoms on both sides. To get 6 oxygen atoms on the left, we need to multiply O2 by 3. So, we put a ‘3’ in front: 2Fe + 3O2 → Fe2O3.
Now let’s check:

Left side:
- Fe: 2
- O: 3 * 2 = 6
Right side:
- Fe: 2
- O: 3
We have 6 oxygens on the left and 3 on the right. We’ve gone from one problem to another! But don’t panic. We’ve balanced the oxygen on the left, but now we need to balance it on the right. To get 6 oxygen atoms on the right, we need to multiply Fe2O3 by 2. So, we put a ‘2’ in front:
2Fe + 3O2 → 2Fe2O3
Let’s do a final count:
Left side:
- Fe: 2
- O: 3 * 2 = 6
Right side:

- Fe: 2 * 2 = 4
- O: 2 * 3 = 6
Whoa, now we have 4 iron atoms on the right and only 2 on the left! Back to the drawing board for iron. This is the fun part, the chemical dance!
We need 4 iron atoms on the left. So, let’s change the coefficient in front of Fe from 2 to 4. Our equation is now: 4Fe + 3O2 → 2Fe2O3.
Final count, take three:
Left side:
- Fe: 4
- O: 3 * 2 = 6
Right side:
- Fe: 2 * 2 = 4
- O: 2 * 3 = 6
And there we have it! 4 iron atoms on both sides, 6 oxygen atoms on both sides. The equation is balanced! See? It’s a bit of a back-and-forth, a chemical tango, but with a little patience and a lot of counting, you can get everyone in sync.
The key takeaways for balancing are:
- Never change the subscripts! Those are part of the chemical identity of the molecule. You can’t just decide water is H3O, that’s a whole different (and probably dangerous) thing. Coefficients are your tools.
- Count meticulously. It’s easy to make a mistake. Double-check your counts after each adjustment.
- Start with the tricky elements. Sometimes elements that appear in only one reactant and one product are easiest to balance first. Elements like oxygen or hydrogen, which often appear in multiple compounds, can be saved for later.
- Treat polyatomic ions as a unit if they appear on both sides unchanged. For example, if you see SO4 on both sides, you can balance the sulfate group as a whole instead of counting S and O separately.
So, the next time you see a chemical equation that looks like it’s been through a blender, take a deep breath. Remember our friends Na, H2SO4, Na2SO4, and H2O. They’re just atoms looking for their perfect partners, and you, my friend, are the matchmaker. You’ve got this! Go forth and balance! May your equations always be neat, your atoms always accounted for, and your scientific adventures always be filled with the joy of perfectly paired elements. Happy balancing!