N-alkylation Of Amines With Alkyl Halides

Ever wondered how those delightful fragrances in perfumes are created, or how the very building blocks of life, like DNA, are tweaked? Well, a little bit of chemical magic called N-alkylation of amines with alkyl halides might be the unsung hero behind it all! It sounds a bit fancy, but at its heart, it’s a wonderfully creative and surprisingly useful chemical reaction that’s accessible to anyone with a curious mind and a little willingness to experiment (safely, of course!). Think of it like snapping LEGO bricks together, but with molecules! We're taking a molecule with a nitrogen atom (an amine) and attaching a carbon chain (an alkyl group) to that nitrogen, using a molecule that's practically a "handle" for this attachment (an alkyl halide).
Why dive into this seemingly niche topic? For the absolute beginner chemist, it’s a fantastic entry point into understanding how new molecules are built. It's a foundational reaction that pops up in many more complex syntheses, so grasping it early makes future learning much smoother. For families looking for an engaging science activity, this can be a way to explore chemical transformations that lead to tangible results, perhaps even creating simple compounds with interesting properties (always under adult supervision and with proper safety precautions!). Hobbyists in areas like soap making, candle crafting, or even creating custom cleaning solutions might find N-alkylation opens doors to new product possibilities, allowing them to tailor the properties of their creations. Imagine creating a scent that’s uniquely yours or a cleaner with just the right level of effectiveness!
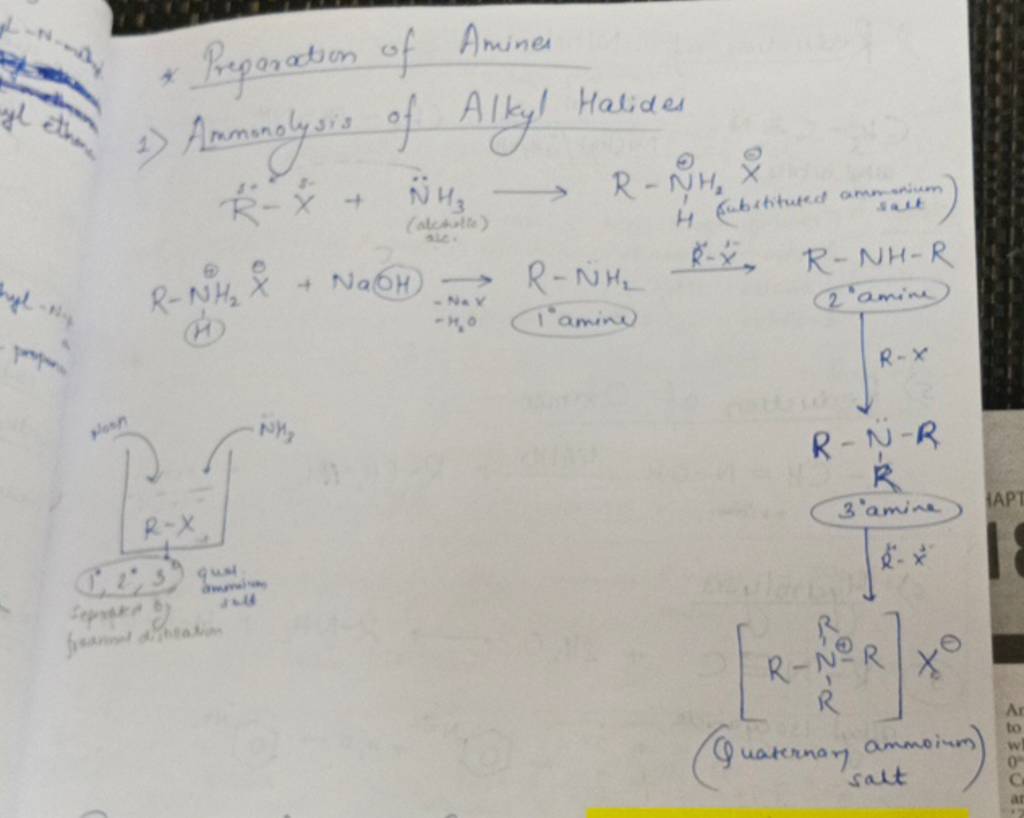
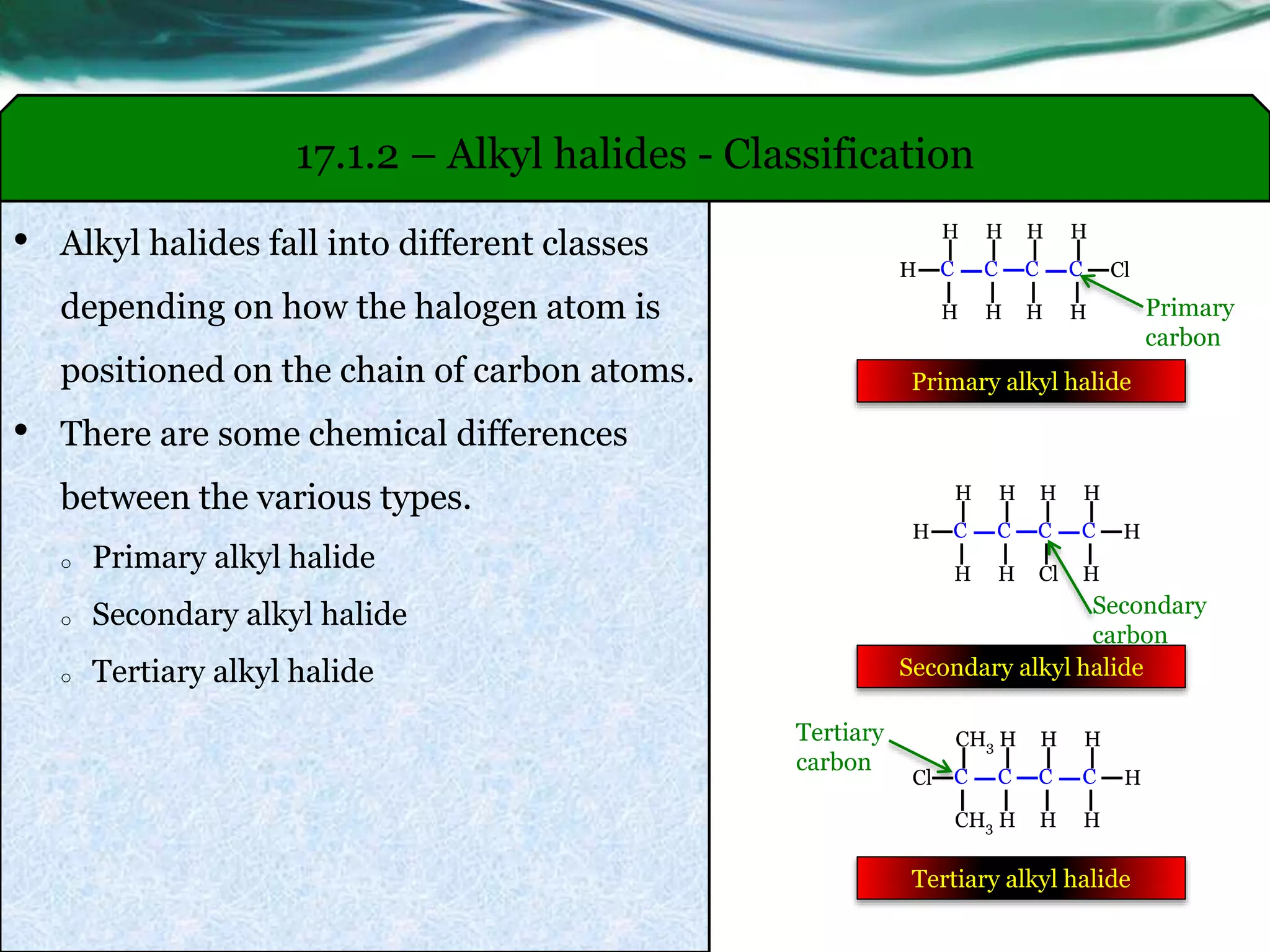
The beauty of N-alkylation lies in its versatility. You can start with a simple amine, like ammonia (though that's a bit trickier to handle), or a more common one like methylamine (CH₃NH₂), and attach different alkyl halides. For instance, using methyl iodide (CH₃I) on methylamine would give you dimethylamine ((CH₃)₂NH). Keep going, and you can even make trimethylamine ((CH₃)₃N)! This is how we build up complexity. Think about the different amino acids that make up our proteins; their structures are often variations on this theme. Even the creation of certain pharmaceuticals and dyes relies on carefully controlled N-alkylation reactions.
Must Read
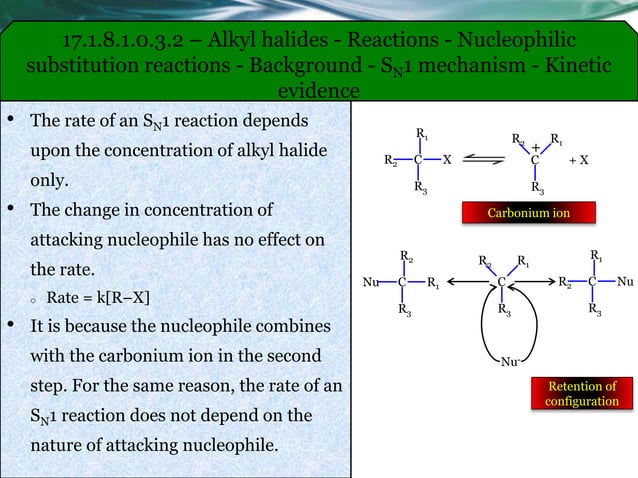
Getting started might sound daunting, but it doesn’t have to be. For a safe, introductory exploration at home (again, with adult supervision!), you could look into experiments that use readily available materials. For example, observing how a mild amine reacts with a carefully chosen alkyl halide in a controlled environment can demonstrate the principle. Many educational chemistry kits offer guided experiments that touch upon these concepts. The key is to start with simple reagents, understand the reaction conditions (like temperature and solvent), and always prioritize safety equipment like gloves and eye protection. Reading up on the basic mechanism – how the electron-rich nitrogen atom attacks the slightly positive carbon atom in the alkyl halide – can also demystify the process.
Ultimately, exploring N-alkylation of amines with alkyl halides is about understanding how we can creatively modify molecules to achieve desired outcomes. It’s a journey into the heart of organic chemistry that can be both educational and surprisingly fun, opening up a world of molecular possibilities for the curious mind.