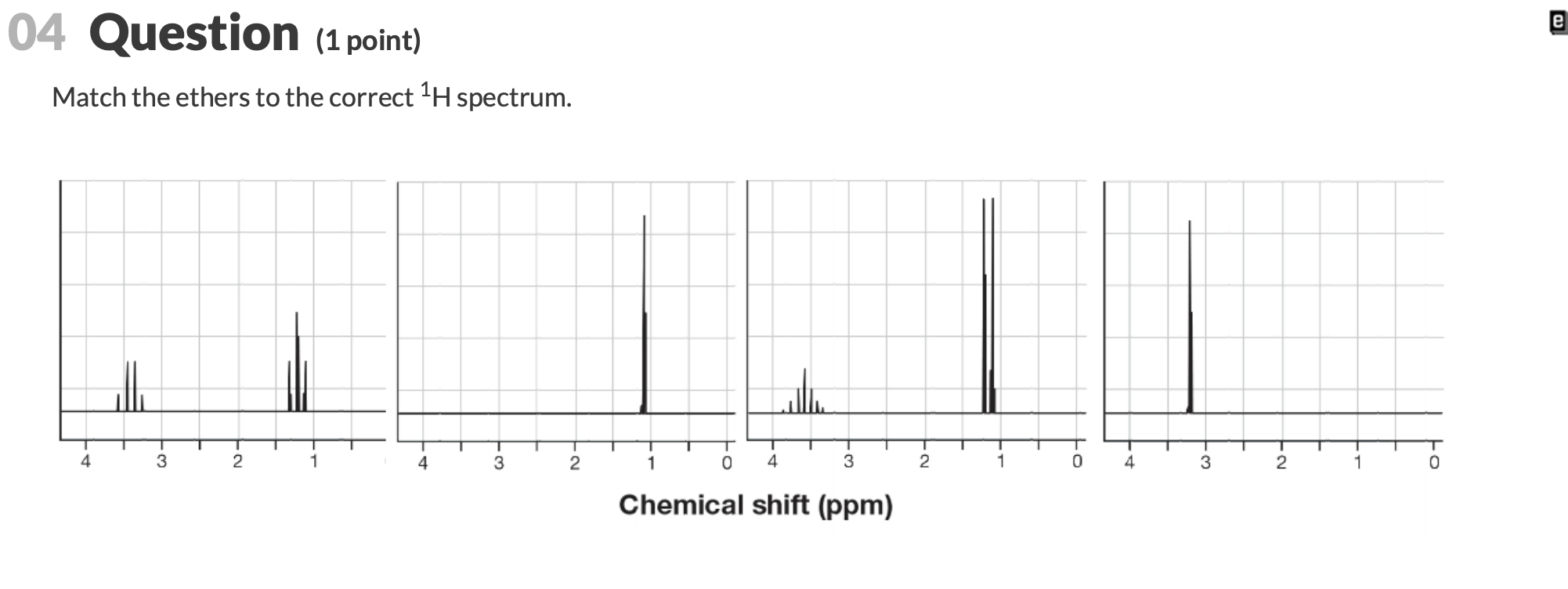
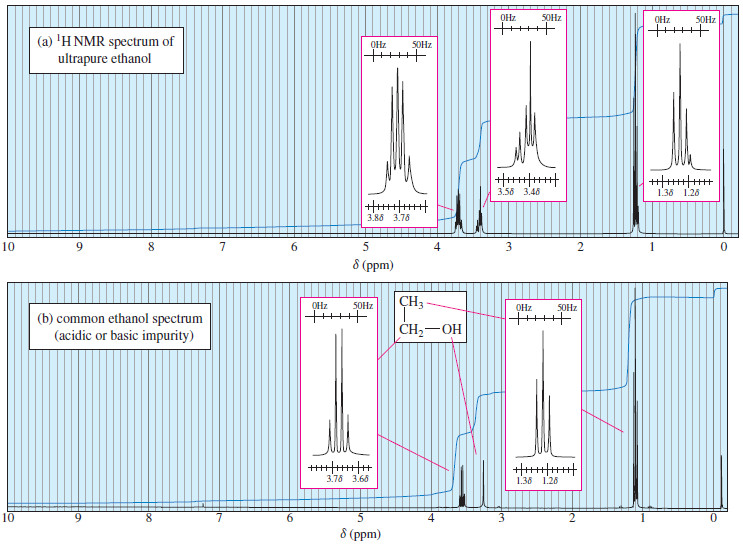
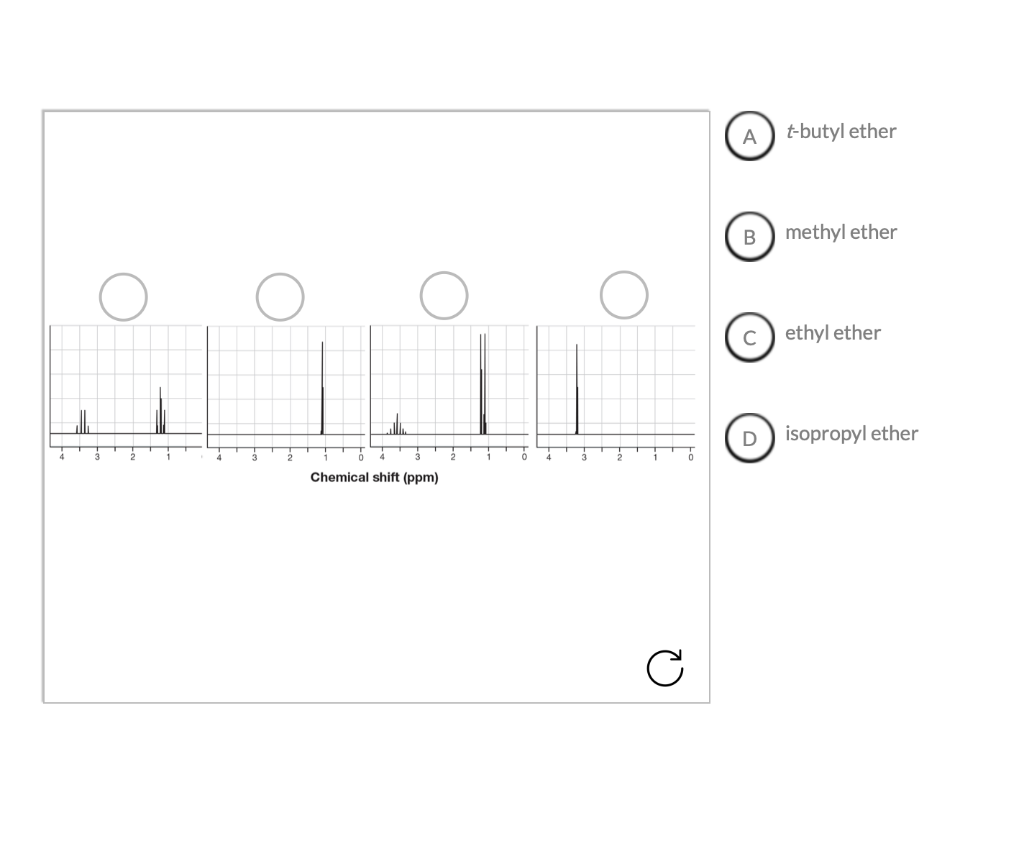
Match The Ethers To The Correct 1h Spectrum.
Ever wondered what makes different substances tick on a molecular level? It’s a bit like a detective story, and one of the coolest tools in our investigative kit is something called Nuclear Magnetic Resonance (NMR) spectroscopy, specifically looking at 1H NMR spectra. Now, don't let the fancy name scare you! Think of it as deciphering a unique fingerprint for each molecule. In this case, we're going to focus on a particular kind of fingerprint: the one belonging to ethers. Matching these fingerprints, the 1H NMR spectra, to their correct ether structures can be a surprisingly rewarding and fun puzzle.
So, why is this relevant or fun? Well, understanding these spectral "fingerprints" is absolutely crucial in chemistry. It allows scientists to identify unknown substances, confirm the structure of newly synthesized compounds, and even study how molecules interact. For anyone with even a passing interest in how the world around us is built, it’s a fascinating glimpse into the hidden world of atoms and their arrangement. It’s like learning a secret language that molecules speak!
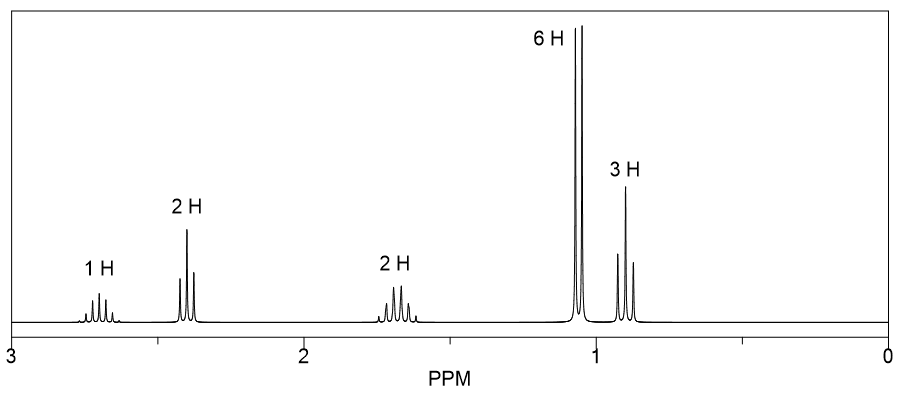
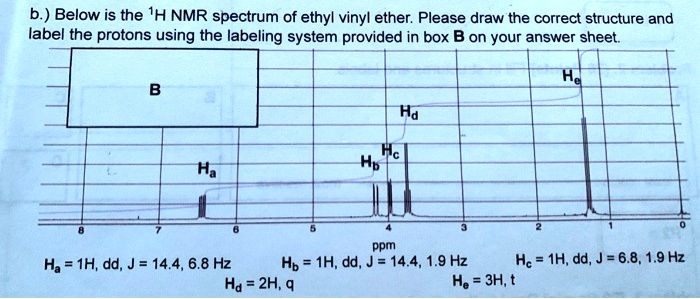
The main purpose of matching 1H NMR spectra to ethers is to validate molecular structures. Imagine you've made a new compound in your lab – how do you know you actually made what you intended? The NMR spectrum acts as a definitive proof. Each signal in the spectrum tells us about the type of hydrogen atoms present in the molecule and their environment. By analyzing the number of signals, their positions (called chemical shifts), their shapes (splitting patterns), and their intensities, we can build a detailed picture of the molecule. The benefits are huge: it saves time and resources by confirming structures quickly, and it’s an indispensable tool for research and development in fields ranging from pharmaceuticals to materials science.
Must Read
In education, this is a staple of undergraduate organic chemistry courses. Students learn to interpret these spectra, which is a foundational skill for any aspiring chemist. Beyond the classroom, this technique is used daily in industrial settings. Pharmaceutical companies use it to ensure the purity and identity of drug compounds. Environmental scientists might use it to identify pollutants. Even in the food industry, it can be used to verify the composition of ingredients.
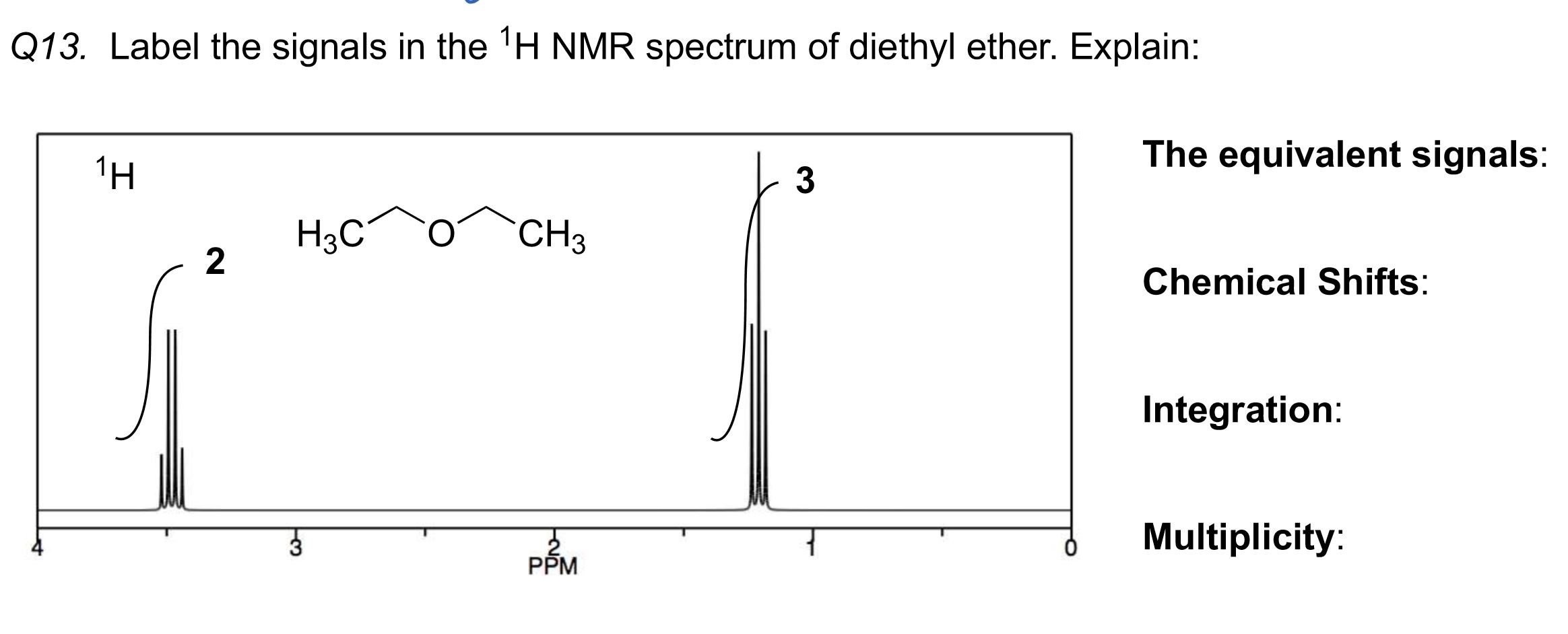
Ready to dip your toes into this spectral detective work? The best way to start is by looking at some simple examples. You can find plenty of 1H NMR spectra of common ethers online. For instance, consider diethyl ether (CH3CH2OCH2CH3) versus dimethyl ether (CH3OCH3). You’ll notice distinct differences in their spectra that directly relate to the different hydrogen atoms in each molecule. A tip for exploration: start with molecules that have only one or two different types of hydrogen atoms. Look at how the number of signals corresponds to the number of unique hydrogen environments. Then, try to understand the splitting patterns – they give clues about neighboring hydrogen atoms. It’s a bit like solving a jigsaw puzzle, where each spectral feature is a piece that fits into the overall molecular picture. Don't be afraid to look up resources and guides; there are many excellent online tutorials and textbooks that break down these concepts in an accessible way.