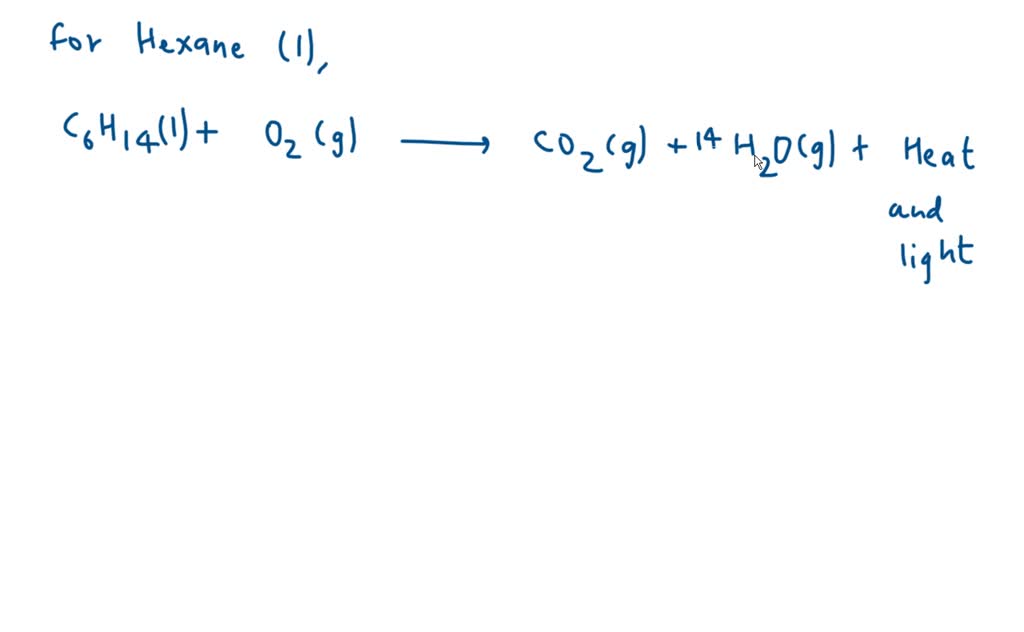Liquid Hexane And Oxygen Balanced Equation
Get ready for a little chemical magic, folks! Today, we're diving headfirst into the wild and wacky world of liquid hexane and its super-powered pal, oxygen. You might be thinking, "Chemicals? Sounds serious!" But trust me, this is more like a culinary explosion waiting to happen, a tiny dance of atoms that's actually pretty darn cool. Imagine your kitchen, but instead of flour and sugar, we've got these two amazing ingredients, ready to put on a show.
So, what exactly is liquid hexane? Think of it as a really, really small building block, like a super-tiny LEGO brick. It's made up of carbon and hydrogen atoms, all linked together in a neat little chain. You might find it chilling in things like glues or even some cleaning products – it's kind of the unsung hero of everyday stuff. Now, when we talk about it being liquid, that just means it's in that pourable, splashy state, like water, but with its own unique personality. It’s not as bubbly as soda, but it’s definitely got a flow!
And then there’s oxygen. Ah, oxygen! The breath of life! The stuff that makes fires roar and makes us all go "Whew!" when we're done with a tough workout. It's in the air we breathe, all around us, just waiting for its moment to shine. In the chemical world, oxygen is like a really enthusiastic party guest, always ready to jump in and get things going. It’s like the energetic friend who’s always suggesting a game of tag, and liquid hexane is like the slightly more laid-back friend who’s happy to join in.
Must Read
Now, here’s where the fun really begins. When liquid hexane and oxygen decide to have a little get-together, things get… exciting! It’s a reaction, a chemical tango, a fiery embrace! Imagine you've got a little bit of liquid hexane, and then you introduce it to a whole bunch of eager oxygen. What happens? Well, it's kind of like throwing a spark on a tiny pile of wood. It ignites! And not just a little flicker, but a full-blown, dazzling display. This reaction is what we call combustion. It’s the process that powers rockets, lights up our stoves, and makes fireworks go BOOM!
Think of it as the ultimate ingredient swap! You start with your hexane and oxygen, and what do you get at the end? A whole new set of goodies! It's like making a cake and ending up with cookies, but way more awesome and energetic.
But what are these "new goodies"? This is where the balanced equation comes in. It’s like a recipe card, but for atoms. It tells us exactly how many of each little building block we start with and how many we end up with. It's all about keeping things fair and square in the chemical universe. For liquid hexane (which has a fancy chemical name we'll wink at for now, let's just call it C6H14 for fun) and oxygen (O2), this recipe looks something like this:

We need to make sure that every atom that goes into the reaction comes out of it. It’s like saying if you put six apples and fourteen bananas into a magical blender, you have to get exactly six apples and fourteen bananas out in a new form. No cheating!
So, for our liquid hexane and oxygen party, the balanced equation is like a carefully orchestrated performance. We have our liquid hexane (C6H14) doing its thing, and it needs a whole bunch of oxygen (O2) to really get the party started. It’s not just one or two molecules of oxygen; oh no! It needs a whole entourage! Imagine giving a shy performer a spotlight – they need the whole stage to shine.
The balanced equation tells us we need 19 molecules of oxygen for every 1 molecule of hexane. That’s a lot of oxygen, right? It’s like inviting your entire neighborhood to a birthday party! This massive influx of oxygen is what allows the hexane to completely burn, to transform into something new and exciting.

And what does it transform into? Ta-da! We get carbon dioxide (CO2) and water (H2O). Yep, the same stuff we exhale and the same stuff that makes up oceans and clouds! It’s like the hexane and oxygen decide to become the building blocks for the very air and water around us. How cool is that? It's a chemical recycling program, but way more spectacular.
So, the whole shebang, the grand finale of this chemical performance, looks like this:
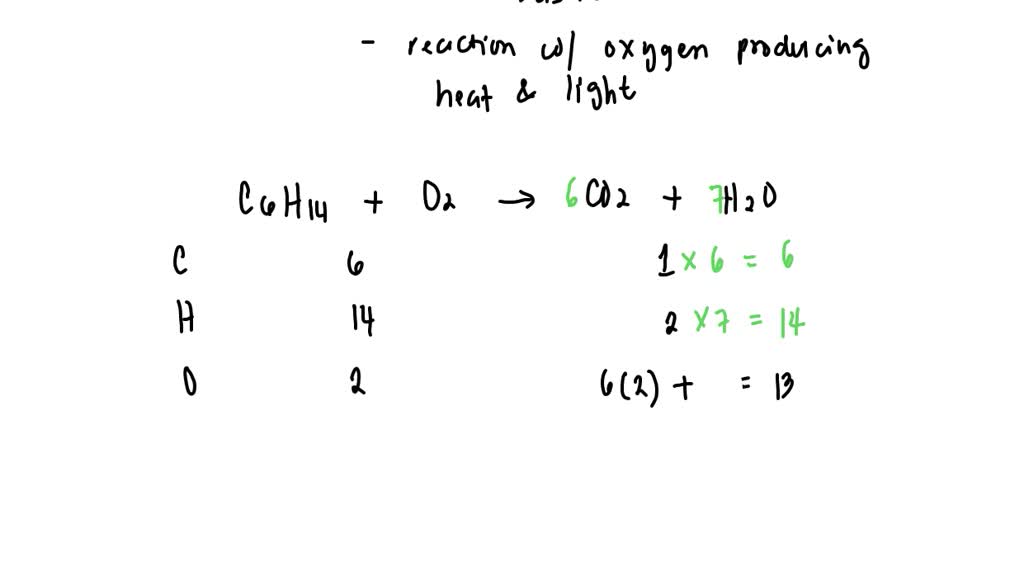
1 molecule of C6H14 + 19 molecules of O2 → 6 molecules of CO2 + 7 molecules of H2O
Isn’t that neat? It’s a perfect balance. For every carbon atom in the hexane, we get a carbon atom in the carbon dioxide. For every hydrogen atom, we get it neatly tucked into the water molecules. It’s a testament to the incredible order and precision that governs the universe, even in something as seemingly simple as burning fuel. It’s like a beautifully choreographed dance where every dancer knows their exact step and position.
So, the next time you see a flame, or think about the air you’re breathing, remember the amazing dance of liquid hexane and oxygen. It’s a story of transformation, energy, and a whole lot of perfectly balanced atoms. It’s proof that even the smallest interactions can lead to something truly spectacular, and that the world of chemistry is full of fun surprises, just waiting to be discovered. It's like finding out your humble LEGO bricks can be used to build not just a house, but a whole new planet! And that, my friends, is something to get excited about!