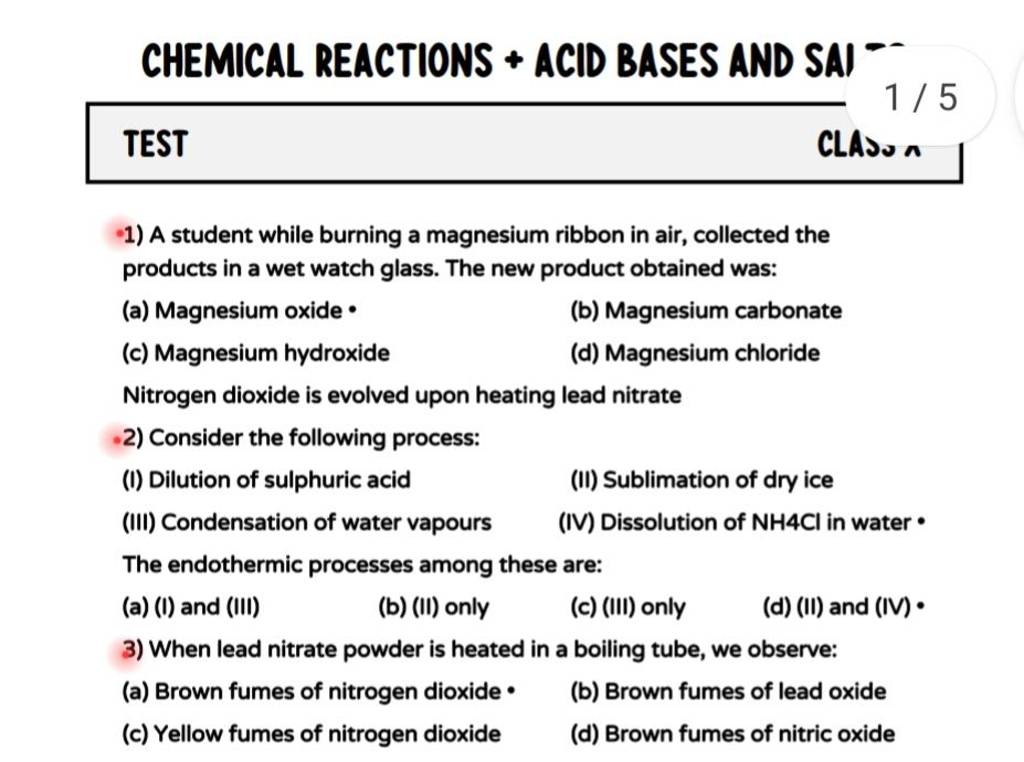
Lead Nitrate Can Be Decomposed By Heating
Hey there, science curious folks! Ever wondered what happens when you take something, give it a good old heating, and it completely changes its tune? Today, we're going to chat about something pretty neat: lead nitrate and how it can be decomposed by heating. Sounds a bit fancy, right? But trust me, it's actually quite a chill concept, like watching a caterpillar transform into a butterfly, only with a bit more drama and some interesting gases involved.
So, what exactly is lead nitrate? Think of it as a chemical compound, a bit like a recipe for a specific kind of salt. Its formula is Pb(NO₃)₂, if you're into the chemical shorthand. The key players here are lead (Pb), a metal, and nitrate (NO₃), which is made of nitrogen and oxygen. When these guys are together, they form a nice, crystalline solid. Usually, it looks like white crystals, pretty much like sugar or salt, but please, don't taste it! This stuff isn't for snacking.
Now, let's get to the exciting part: decomposition by heating. What does that even mean? Well, imagine you've got a complex LEGO castle. If you heat it up (hypothetically, of course!), you might imagine the bricks just getting warmer. But in the chemical world, heating can actually break things apart. It's like taking that LEGO castle and, with a little energetic nudge from heat, it splits into individual bricks, maybe some wheels, and even some tiny little people. Lead nitrate is a bit like that LEGO castle. When you apply enough heat, it doesn't just get warm; it actually starts to fall apart into simpler substances.
Must Read
Why is this cool? Because it shows us that even seemingly stable compounds have hidden potential for change. It's a fundamental principle in chemistry, but seeing it happen with something like lead nitrate is pretty captivating. It’s a reminder that the world around us is constantly in flux, even at a microscopic level. Think of it like baking a cake. You start with flour, sugar, eggs, and butter – all separate ingredients. But when you apply heat (the oven!), they come together to create something entirely new and delicious. Heating lead nitrate is kind of the reverse; you start with one thing, and heat breaks it down into other things.
So, what exactly are these simpler substances that lead nitrate breaks down into? When you heat solid lead nitrate to a high enough temperature, it doesn't just melt or evaporate. Oh no, it undergoes a chemical transformation. It breaks down into lead(II) oxide, which is a yellowish solid, and then releases two gases: nitrogen dioxide and oxygen.

The Products of the Heat Party
Let's break down these products a little, because they're the real stars of the show here. First up, we have lead(II) oxide, sometimes just called lead monoxide. This stuff is a bit different from the original lead nitrate. It's a solid, and it's often described as having a yellowish or reddish color, depending on its specific form. Imagine your white sugar crystals turning into a sandy yellow powder when you heat them – that's the vibe. It’s a much simpler compound, representing a part of the original lead nitrate that’s still hanging around, just in a different form.
Then we have the gases. This is where things get really interesting and, dare I say, a little dramatic. We get nitrogen dioxide. This gas is notorious for being a brown, noxious gas. It's the kind of stuff that makes you wrinkle your nose. If you've ever smelled a slightly acrid or metallic scent, you might have encountered something similar. It’s an important gas in atmospheric chemistry, and its presence tells us that a significant chemical change has occurred. It’s like when you pop popcorn, and you get that burst of steam and aroma – here, we get a burst of brown gas!

And finally, we have oxygen. Yes, the very same oxygen that we breathe! It's kind of poetic, isn't it? That this process, which breaks down a compound, also releases something essential for life. It's a good reminder that chemistry isn't just about complicated reactions; it's about the fundamental building blocks of our world, and sometimes, those reactions give back something precious.
Why Does This Happen? The Science-y Bits (But Keep it Chill)
Now, you might be asking, "Why does the heat do this?" It all comes down to the stability of the molecules. The nitrate group (NO₃) in lead nitrate is a bit like a fragile structure. When you add energy in the form of heat, you're essentially making the atoms within the lead nitrate molecule vibrate more and more violently. Eventually, these vibrations become so strong that they overcome the bonds holding the nitrate group together and to the lead atom.
Think of it like holding hands with a few friends. If everyone is standing calmly, you're all connected. But if everyone starts jumping and spinning, eventually, someone's going to let go! The heat energy is like that energetic dance, causing the bonds to break. The nitrate group itself breaks down into oxygen and nitrogen dioxide, and the lead combines with the oxygen to form lead(II) oxide.
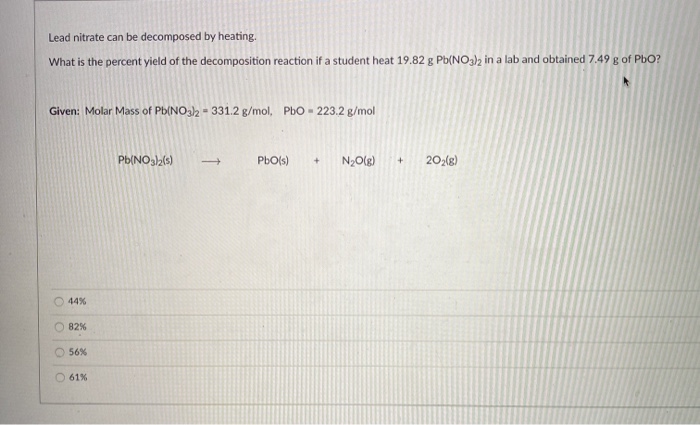
The whole process can be summarized with a chemical equation, which is just a fancy way of writing down what happens: 2Pb(NO₃)₂ (s) → 2PbO (s) + 4NO₂ (g) + O₂ (g). Don't let the symbols scare you! The (s) just means solid, and (g) means gas. So, solid lead nitrate turns into solid lead(II) oxide and two gases, nitrogen dioxide and oxygen.
This decomposition is actually a really important concept in analytical chemistry. Scientists use these kinds of reactions to identify and quantify substances. By observing the products and how much of them are formed, they can learn a lot about the original compound. It’s like being a detective, but instead of clues, you’re looking at chemical fingerprints.
Another fun comparison: imagine a really intricate clockwork mechanism. When you heat it up, some of the smaller, more delicate gears might break off, the springs might unwind, and you're left with a simpler, less functional, but still recognizable set of parts. Lead nitrate is like that clockwork; heating it reveals its constituent components in a new arrangement.
It's also worth noting that lead compounds, including lead nitrate, are toxic. So, while this is a fascinating chemical reaction, it's something that should be handled with extreme care and proper safety precautions in a laboratory setting. We're talking about learning and understanding here, not about trying this at home with your kitchen stove!
So, there you have it. Lead nitrate, a seemingly simple white crystal, can be coaxed by heat to reveal its hidden components: a yellowish solid and two intriguing gases, one of which is vital for us to breathe. It’s a small glimpse into the vast and dynamic world of chemistry, where even a little bit of heat can cause a world of change. Pretty cool, right?