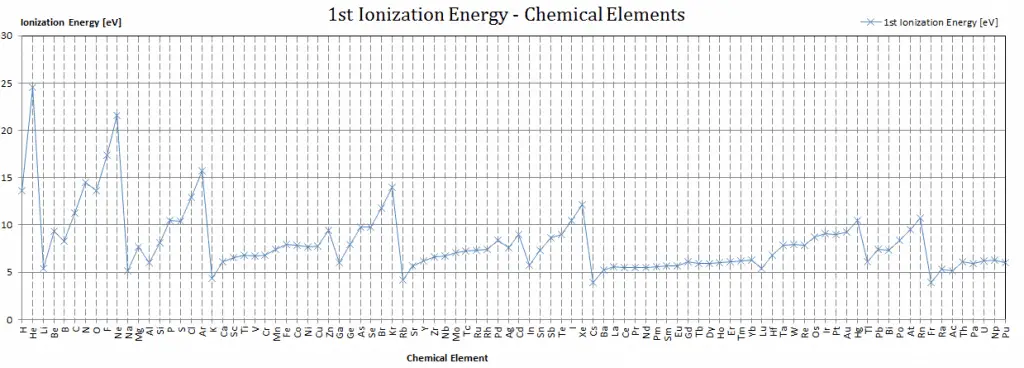
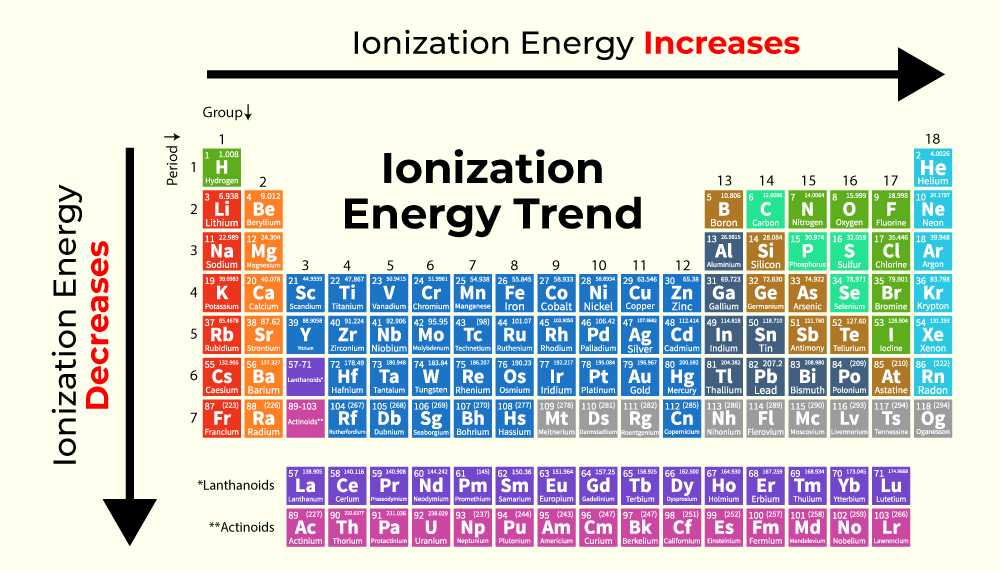
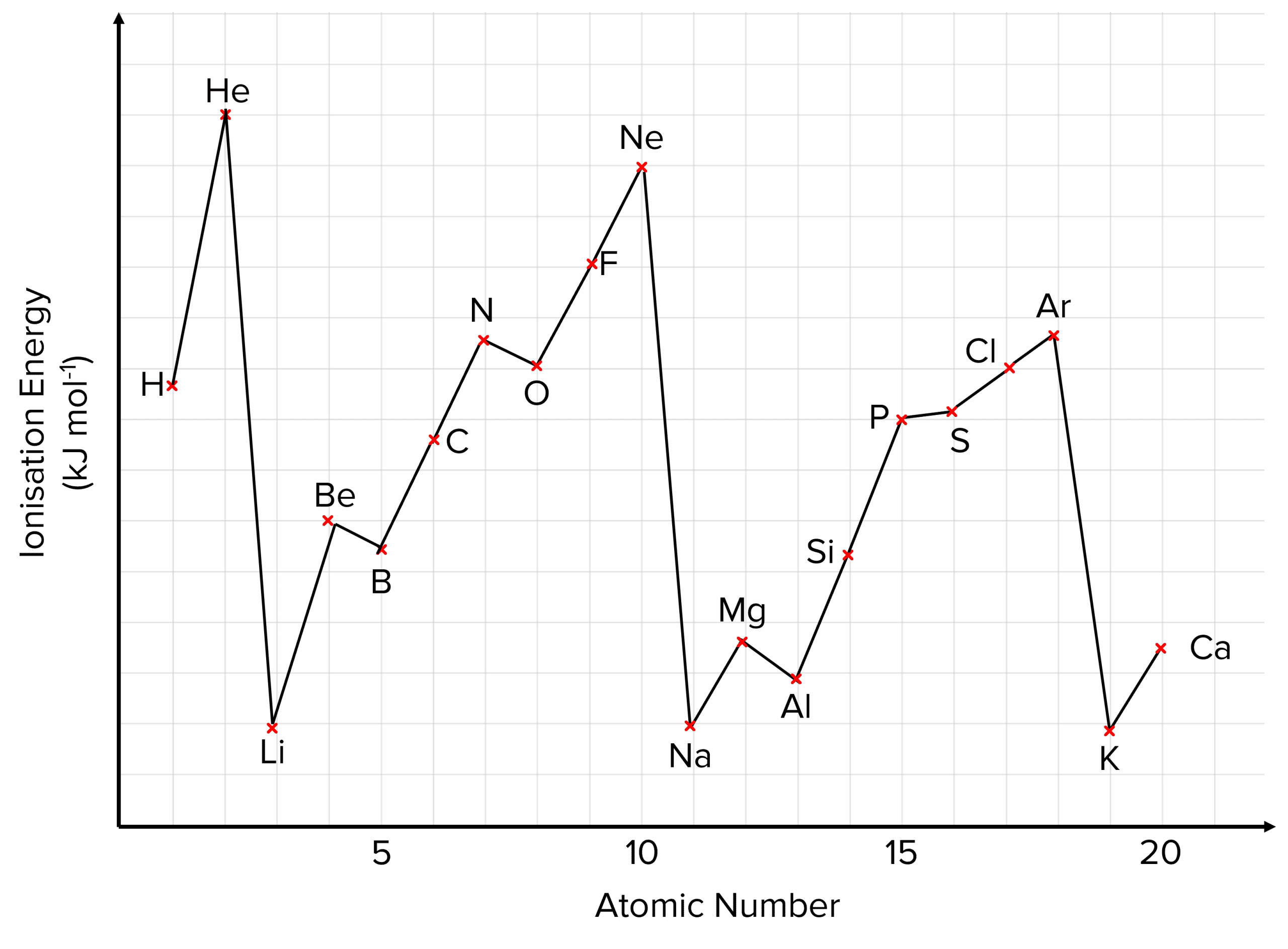
Ionization Energy Of Phosphorus And Sulphur

Imagine the universe is like a giant, bustling playground, and all the elements are the kids playing on it. Each element has its own unique personality, its own way of interacting with others, and its own special quirks. Today, we’re going to zoom in on two of these elemental buddies, Phosphorus and Sulphur, and uncover a little secret about their energy levels. Think of it like figuring out how much energy it takes to get them to share their toys, or perhaps, to convince them to join a new game.
Now, when we talk about "ionization energy," it sounds a bit like a super-secret spy mission, doesn't it? But really, it's just a fancy way of saying how much effort, how much oomph, you need to give an atom to convince it to let go of one of its little electron friends. These electrons are like tiny energetic particles zipping around the atom's core, and they’re pretty attached to their home. Ionization energy is the "come on, let go!" nudge.
Think of it this way: if an atom is a shy kid clutching a favorite teddy bear, ionization energy is the amount of coaxing, tickling, or gentle persuasion it takes to get them to hand over the bear for a moment.
So, let’s meet our first character: Phosphorus. Phosphorus is a bit of a dramatic character on the periodic table. It’s the element that makes those sparklers at parties twinkle and gleam. Without Phosphorus, those magical bursts of light wouldn't happen. It’s also a super important building block of life, found in your DNA and bones! Pretty amazing, right? But Phosphorus, when it's all by itself, is a little bit of a clingy friend when it comes to its electrons. It holds onto them quite tightly.
Now, let’s introduce its neighbor, Sulphur. Sulphur is the element responsible for that distinctive smell of burning matches and, let's be honest, sometimes a less pleasant aroma associated with other things. It's also vital for life, helping to create proteins. Sulphur is a bit more of a free spirit than Phosphorus. It’s more willing to share its electrons, or perhaps, it’s just a little less bothered by the idea of letting one go. This means it takes less energy to pry an electron away from a Sulphur atom compared to a Phosphorus atom.

This might seem like a small detail, a tiny difference in their "sharing" habits. But in the grand scheme of the universe, these little energetic preferences dictate how elements behave. It's like knowing that some kids are happy to share their crayons immediately, while others need a little more convincing. This willingness, or reluctance, to let go of electrons is a fundamental part of how elements bond together to form all the amazing things we see around us.
Imagine you’re trying to build a magnificent castle out of LEGOs. Each LEGO brick is an atom, and how they click together depends on their energetic personalities. Phosphorus, being a bit more protective of its electrons, might not join in a new structure as readily. Sulphur, on the other hand, is more eager to connect and form new shapes. This difference in their ionization energy is like having bricks that are slightly easier or harder to snap together. It influences the kinds of structures, or molecules, that can be built.
So, the next time you see a sparkler fizzing or catch a whiff of something distinctly… sulphurous, remember these two elemental buddies. Remember that even in the seemingly abstract world of atoms and energy, there’s a story of personality, of quirks, and of how readily they share their tiny energetic companions. Phosphorus holds on a little tighter, a bit more possessive. Sulphur is a bit more relaxed, a touch more open to new connections. It’s these subtle differences, these small energetic nudges, that allow the universe to be the incredible, diverse, and sometimes wonderfully smelly place that it is.
It’s a heartwarming thought, really, that the fundamental building blocks of everything are governed by such relatable traits. They’re not just cold, hard facts; they’re the energetic dance of atoms, each with its own reason for holding on or letting go. And the story of Phosphorus and Sulphur’s ionization energy is just one tiny, yet fascinating, chapter in this grand, elemental tale.