If The Reaction Quotient Is Greater Than K

Hey there, science curious folks! Ever feel like you're in the middle of a really good party, and things are just happening? Well, imagine a microscopic party, a chemical one, where everything is buzzing and the energy is super high. Sometimes, in these tiny chemical shindigs, things can get a little... out of hand. It's like the guests at the party are having so much fun, they're trying to do more than the usual amount of mingling.
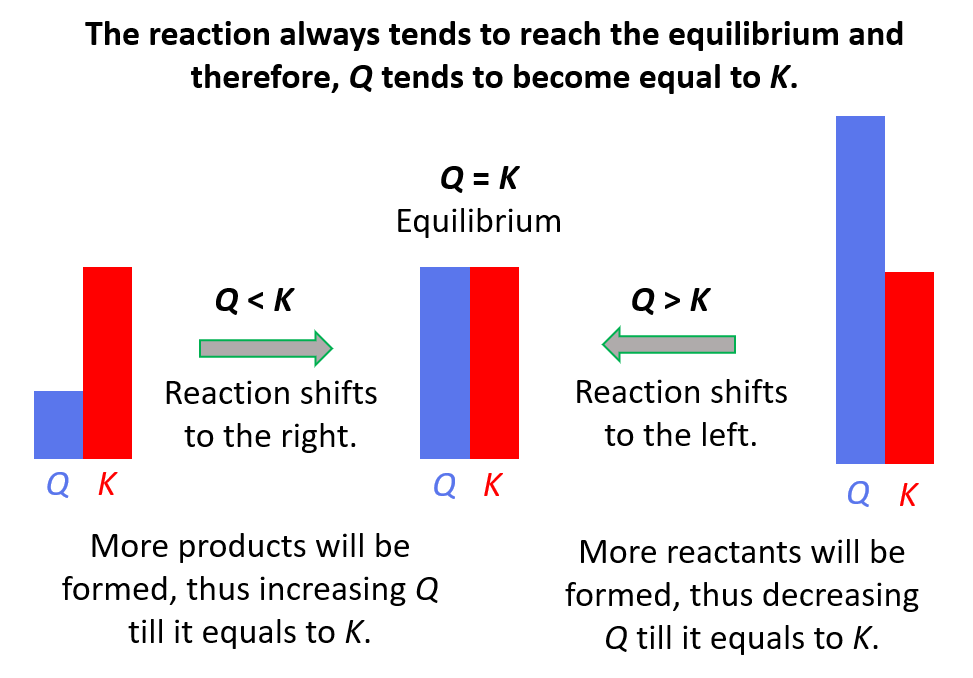
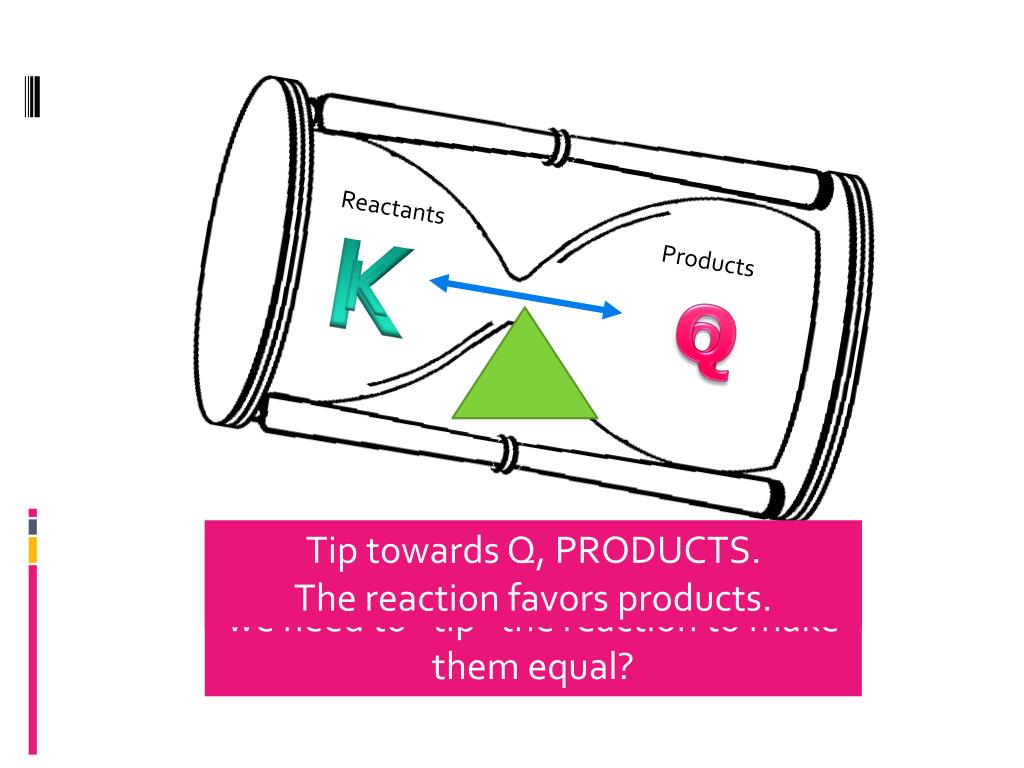
In the world of chemistry, we have this neat idea called equilibrium. Think of it like a perfectly balanced seesaw. On one side, you have your reactants – the ingredients you start with. On the other, you have your products – what you end up with after the reaction has done its thing. Equilibrium is that sweet spot where both sides are equally busy, and the seesaw is perfectly level. It's a state of balance, and we have a special number to describe this balance called K, or the equilibrium constant. It tells us how much product is favored over reactant at that perfect balance point.
But what happens when the party gets a little too wild? What if, for some reason, the chemical reaction has made way, way more product than it's supposed to at equilibrium? This is where our story gets exciting! It's like our seesaw has tipped way over to the product side. We have a special tool to measure this, and it's called the reaction quotient, or Q. Think of Q as a snapshot of the reaction right now, at any given moment. It's like taking a quick photo of the party and seeing who's doing what.
Must Read
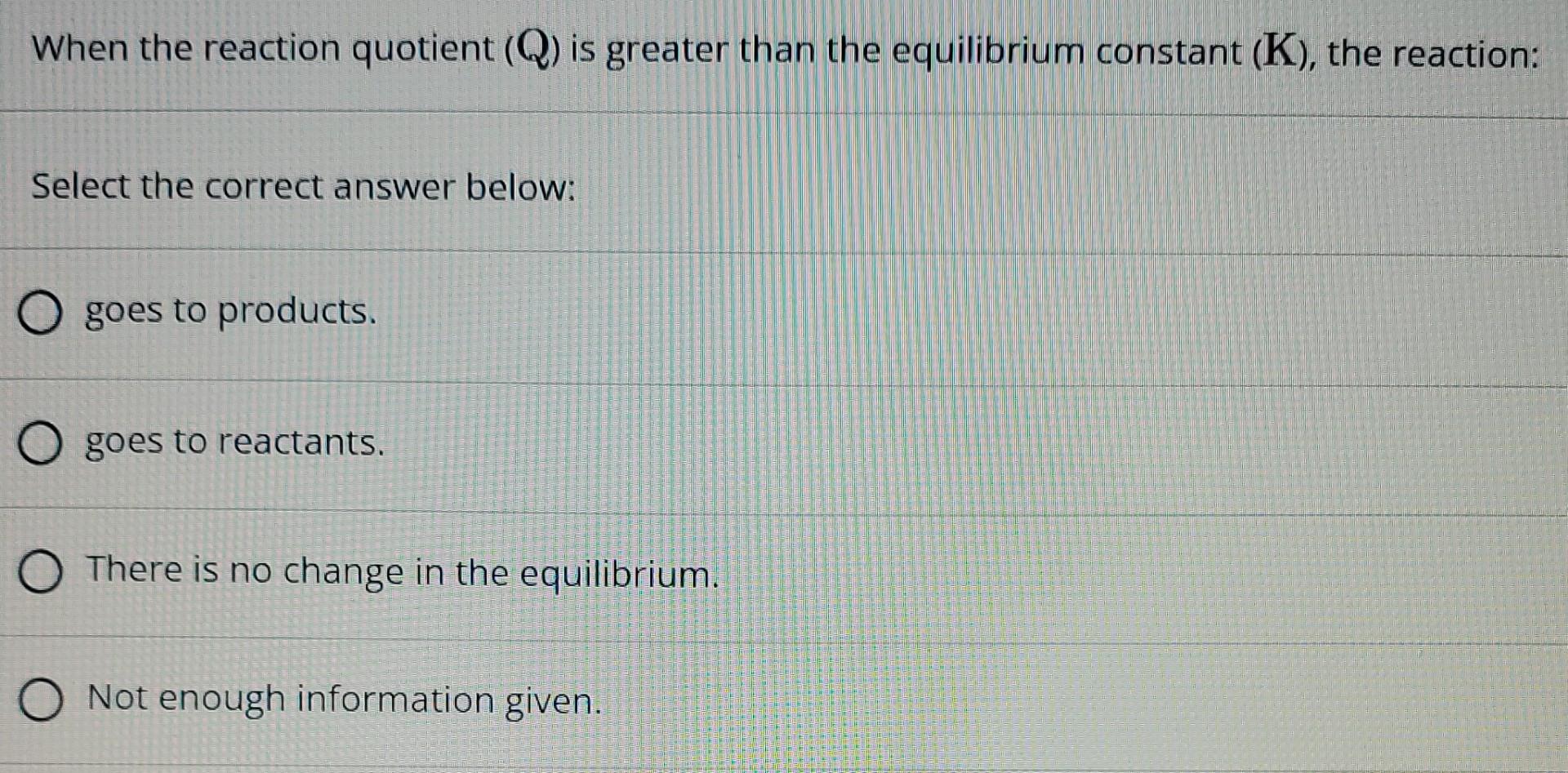
Now, here’s the fun part. What if our snapshot, our Q, shows that there are a lot more products hanging around than there should be at the perfect, balanced party (that's what K tells us)? In other words, what if Q is greater than K? This is where the real drama unfolds! It's like our party is just overflowing with products. The chemistry is basically saying, "Whoa there! We've got too much of this stuff!"
So, what does chemistry do when it finds itself in this sticky situation? It doesn't just shrug its shoulders and let the party run wild forever. Oh no. Chemistry is all about finding that balance, that equilibrium. When Q is greater than K, it's a clear signal that the reaction needs to chill out and re-adjust. It's like the party host stepping in and saying, "Okay everyone, let's dial it back a notch!"
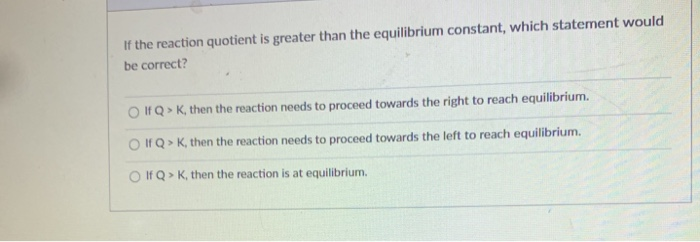
The reaction, in its own clever chemical way, will start to undo some of its recent work. It's going to start converting those excess products back into reactants. Imagine the partygoers who were having too much fun making new things suddenly deciding to un-make them and go back to being their original selves. This process is called the reaction shifting to the left, or favoring the reverse reaction. It's a beautiful, natural tendency towards balance.
Why is this so entertaining? Because it shows us that even at the microscopic level, there's a constant drive for order and balance. It's like a tiny, invisible dance of creation and destruction, all in pursuit of a perfect equilibrium. It’s a cosmic tug-of-war, and when Q is greater than K, you know the tug is on!

It’s a reminder that things are always in motion, always adjusting. Even when it seems like a reaction has settled, it's still working, still fine-tuning. This concept of Q vs. K isn't just some abstract equation; it's a fundamental principle that governs how chemical reactions behave. It's the unseen hand that guides the chemical world towards its most stable, balanced state.
Think about it like this: imagine you’re baking cookies. You mix your ingredients (reactants) and bake them (reaction) to get delicious cookies (products). If you somehow ended up with way, way more cookie dough than you ever planned, you’d probably try to use that extra dough to make more cookies, right? Or maybe you'd even think about how to un-mix some of the ingredients if you could! Well, chemistry does something similar. When there's "too much product," it starts to convert that product back into reactants to reach that perfect balance.

It’s like a self-correcting mechanism built into the universe. When things get a little too lopsided, nature steps in and nudges them back into place. This is what makes studying chemistry so fascinating. You’re not just memorizing facts; you’re learning about the underlying rules that govern the entire physical world. You're peeking behind the curtain to see how everything is supposed to work.
So, the next time you hear about Q being greater than K, don't just think of it as a number. Think of it as a tiny chemical party that's gotten a little too enthusiastic. It's a moment of imbalance, a signal for a correction, and a beautiful illustration of the relentless pursuit of equilibrium. It's chemistry in action, and honestly, it's pretty darn entertaining!
It’s a fundamental concept that explains so much about how reactions proceed, and understanding it is like unlocking a secret code to the chemical world. It’s not just about what happens, but why it happens and how it all tries to get back to that ideal state of balance. Pretty cool, right?
So, if you ever find yourself wondering what happens when the chemical scales tip a little too far, remember the tale of Q > K. It's a story of a chemical system taking a deep breath, looking around, and saying, "Alright, time to get back to normal!" It’s a reminder of the elegant, dynamic nature of chemistry, always striving for that perfect moment of equilibrium.