Identify The True Statements About Colloids.

So, picture this: I'm at my aunt Mildred's house, right? Aunt Mildred, bless her cotton socks, makes the best homemade whipped cream. Seriously, it's like a cloud of pure, unadulterated joy. But one time, she was telling me about how she was trying to make it for a big party, and it just… wouldn't whip. It was this sad, watery mess. She was about to chuck it all, muttering about the humidity and evil spirits in the kitchen. I, being the ever-so-helpful niece, piped up, "Aunt Mildred, maybe it's something to do with... science?" She gave me a look that could curdle milk, but eventually, after a bit of poking around online (and maybe a stern word with the electric mixer), we figured it out. Turns out, the fat globules in the cream weren't stable enough. It was a bit of a colloid crisis, if you will.
And that, my friends, is how I stumbled headfirst into the wonderfully weird world of colloids. You know, those things that are sort of here, sort of there, and definitely not what you expect them to be. They're all around us, doing their own mysterious thing, and once you start noticing them, you can't unnotice them. It's like a secret superpower for spotting the less-obvious wonders of everyday life.
So, let's ditch the dusty textbooks and the terrifyingly complex equations for a moment. We're going to have a chinwag about colloids. Think of this as a friendly chat, fueled by curiosity and maybe a bit of coffee. We're going to try and pick out the true statements about these enigmatic mixtures. Ready to dive in? Because I am!
Must Read
Colloids: The Ultimate "In-Betweeners"
First things first, what exactly is a colloid? If you're picturing a bunch of particles just chilling randomly, you're not entirely wrong, but it's a little more nuanced than that. Imagine a soup. You can see the bits of carrot and peas floating around, right? That's a suspension. The bits are big enough to see and they'll eventually settle out if you leave it alone. Now, think of salt dissolved in water. That's a true solution. You can't see the salt anymore; it's completely dispersed. Colloids? They're that awkward middle child. They're not quite a suspension, and they're definitely not a solution. The particles are bigger than in a solution, but not big enough to settle out easily.
This "in-between" nature is key to understanding them. The dispersed particles in a colloid are typically larger than molecules in a true solution, but still too small to be seen with the naked eye. We're talking sizes between 1 nanometer and 1 micrometer. Think of them as being suspended in a continuous medium, but with a bit more stability than a regular suspension. They don't settle out readily because they're constantly being bombarded by the molecules of the medium, which keeps them dancing around.
And this dancing? It’s quite energetic! This movement is called the Brownian motion, named after a botanist who noticed pollen grains jiggling around in water under a microscope. It’s like they’re perpetually doing a little jig, never quite finding a place to rest. This constant movement is what prevents them from settling. Pretty neat, huh? It’s one of the defining characteristics, and you can often see this jiggling if you shine a beam of light through a colloid – a phenomenon called the Tyndall effect. Ever seen a shaft of sunlight cut through a dusty room? That's basically the same principle, but with dust particles instead of colloid particles. Pretty cool, right?
Debunking the Myths: What Colloids AREN'T
Now, before we get too carried away with their fascinating properties, let's clear up some common misconceptions. It's easy to get colloids confused with other types of mixtures, but understanding the differences is crucial for identifying the true statements.

Firstly, colloids are not true solutions. This is a big one. In a true solution, the solute particles are completely dissolved and indistinguishable from the solvent. Think of sugar in water. You can't see the sugar molecules. In a colloid, however, the dispersed particles are larger and can, under certain conditions, scatter light or even be separated by physical means (though it's usually more difficult than separating a suspension). So, if someone tells you milk is a true solution, you can politely (or not so politely, depending on your caffeine levels) correct them. Milk is a classic colloid – a dispersion of fat globules and protein in water.
Secondly, colloids are not necessarily opaque. While many colloids are opaque or translucent (like milk or paint), some can appear clear. The key is the size of the dispersed particles and their interaction with light. If the particles are small enough and don't scatter light intensely, the colloid might look clear. So, just because something looks clear doesn't mean it's a true solution. It could be a colloid with very finely dispersed particles!
Thirdly, the dispersed particles in a colloid do not settle out easily. This is the defining feature that separates them from suspensions. While gravity is always there, the constant bombardment by solvent molecules keeps the colloid particles in perpetual motion (Brownian motion, remember?). So, that foggy window in your car after a rainstorm? That's probably not a suspension of dirt settling; it's more likely a colloid of oil or other residue dispersed in water. If it were a suspension, you'd see the dirt collecting at the bottom of the puddle.
The Nitty-Gritty: True Statements About Colloids
Alright, enough with the what-ifs and what-not. Let's get down to the brass tacks. What are the absolute, no-doubt-about-it, true statements about colloids?
Statement 1: Colloids exhibit the Tyndall effect.
This is a biggie, and we've touched on it. The Tyndall effect is the scattering of light by particles in a colloid or suspension. When a beam of light passes through a colloid, the dispersed particles scatter the light in all directions, making the beam visible. This is why you can see the path of a headlight beam through fog, or why sunlight streaming through a window illuminates dust particles. In a true solution, the particles are too small to scatter light in this way, so the beam of light would pass through unseen.
Think about it: if you shine a flashlight through a glass of water with salt dissolved in it, you won't see the beam. But if you shine it through a glass of milk, you absolutely will. That's the Tyndall effect in action. It's a simple, yet powerful, way to differentiate between a colloid and a true solution. So, if you see light scattering like that, you can confidently say, "Ah, a colloid!"
Statement 2: The dispersed particles in a colloid are in constant, random motion (Brownian motion).
We've already sung the praises of Brownian motion. This random zigzag movement of the dispersed particles is a direct result of their collisions with the molecules of the dispersion medium. These collisions are happening constantly, from all directions, and they impart enough kinetic energy to the colloid particles to keep them suspended and prevent them from settling under gravity. It's this perpetual dance that gives colloids their stability.
Imagine a bunch of tiny billiard balls (the colloid particles) being constantly nudged and bumped by an invisible swarm of even tinier, super-energetic ping-pong balls (the medium molecules). They can't just sit still; they're always being pushed around. This isn't some gentle sway; it's a vigorous, chaotic jiggling that keeps them from grouping up and sinking to the bottom. So, if you're observing particles that are visibly jiggling around in a fluid and not settling, you're likely witnessing Brownian motion, a sure sign of a colloid.

Statement 3: Colloids can have electrical charges on their dispersed particles.
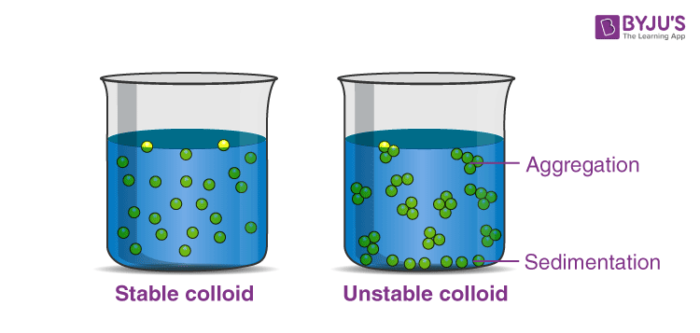
This one's a bit more subtle, but super important for understanding how colloids behave, especially in things like stability and flocculation (which is basically clumping up). The surfaces of colloid particles often carry an electrical charge. This charge can be positive or negative, and it arises from a variety of factors, like adsorption of ions from the surrounding medium or ionization of surface groups. What's really interesting is that, in a stable colloid, all the dispersed particles tend to carry the same type of charge.
Why is this a good thing? Because, as you probably remember from your basic science classes, like charges repel! So, these like charges on the colloid particles keep them from clumping together. They're constantly pushing each other away, which helps maintain their dispersed state. This electrostatic repulsion is a major factor in the stability of many colloidal systems. If you were to add something that neutralized these charges, you might find your colloid starting to settle or clump up – like Aunt Mildred's whipped cream crisis!
Statement 4: Colloids are heterogeneous mixtures.
Even though they might look uniform to the naked eye, colloids are indeed heterogeneous mixtures. This means they consist of two or more distinct phases that are not uniformly distributed throughout the mixture. In a colloid, you have the dispersed phase (the particles) and the dispersion medium (the substance in which the particles are dispersed). While the particles are too small to be seen individually, they are still physically distinct from the medium.
Think back to milk. It looks pretty uniform, right? But under a powerful microscope, you can see the individual fat globules and protein micelles dispersed within the watery serum. They haven't chemically bonded with the water; they're just floating around in it. This is the hallmark of a heterogeneous mixture. Unlike a homogeneous mixture (like a true solution), where the components are indistinguishable, in a colloid, there's still a structural difference, even if it's at a microscopic level. So, even if it looks like one thing, it's actually made of different parts that haven't completely merged.

Statement 5: Colloids are more stable than suspensions but less stable than true solutions.
This is the essence of their "in-between" nature. Colloids are more stable than suspensions because the particles don't settle out readily due to Brownian motion and electrostatic repulsion. You can leave a cup of milk on the counter for days and it won't separate into distinct layers. However, colloids are less stable than true solutions. In a true solution, the solute particles are so small and so uniformly dispersed that they are essentially inseparable by simple physical means. They're chemically integrated, not just physically suspended.
So, while a colloid offers a good degree of stability, it's not as permanent as a true solution. With the right conditions (like changing the pH, temperature, or adding electrolytes), you can often disrupt the colloidal state and cause the particles to aggregate or settle. It’s like the difference between a carefully constructed sandcastle that might withstand a gentle breeze (colloid) and a puddle of water that’s just… water (true solution). The sandcastle has structure and some resilience, but it’s not as fundamental as the water itself.
Beyond the Basics: Why Do We Care?
You might be thinking, "Okay, this is all well and good, but why should I care about colloids?" Well, beyond the sheer fascination of understanding the world around us, colloids are incredibly important in countless everyday applications and natural phenomena. Think about it:
- Food: Whipped cream (our Aunt Mildred's nemesis turned friend!), mayonnaise, butter, jelly, ice cream – all are colloids.
- Medicine: Many drugs are administered in colloidal form for better absorption. Antacids are often colloidal dispersions.
- Industry: Paints, inks, glues, cosmetics, and even water purification processes rely heavily on colloidal chemistry.
- Nature: Fog, mist, clouds, smoke, blood, and the very structure of soil all involve colloidal systems.
So, the next time you’re enjoying a frothy latte, marveling at a sunset through a dusty haze, or even just looking at a carton of milk, remember the unsung heroes of the mixture world: the colloids. They’re the masters of the in-between, the perpetual dancers, and the reason why so many of the things we take for granted actually work. Keep your eyes peeled, and you’ll start seeing them everywhere!
And who knows, maybe you’ll even be able to help a relative out of a whipped cream crisis. You’ll be the science guru of the kitchen, all thanks to understanding the true statements about colloids. How cool is that?