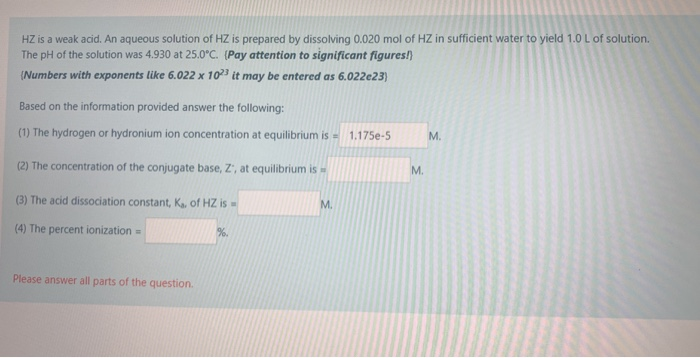
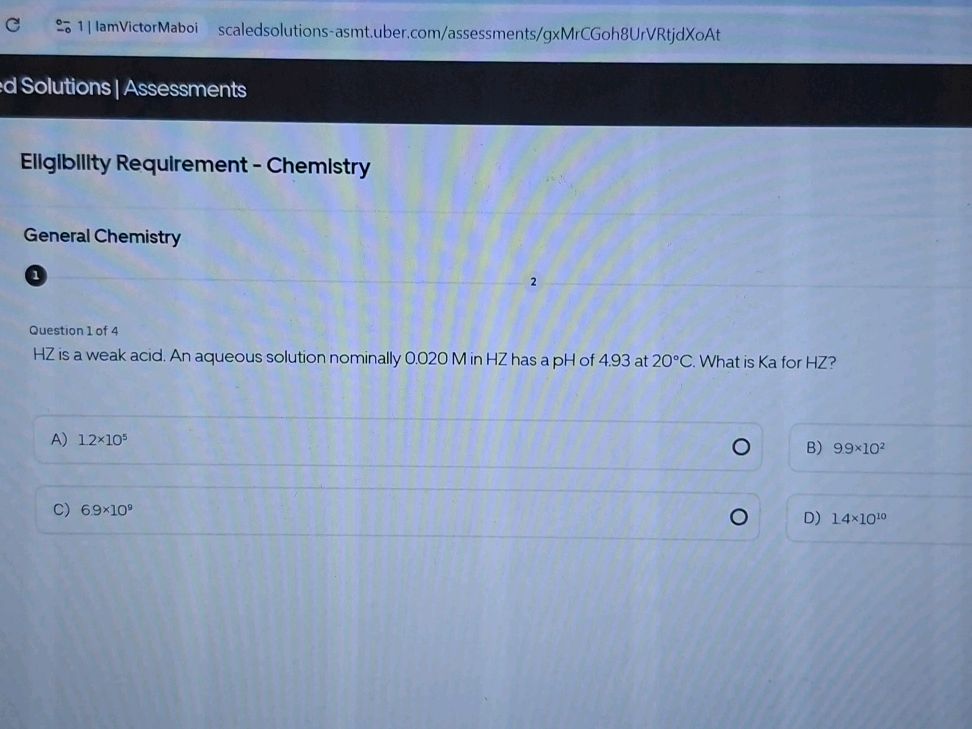
Hz Is A Weak Acid An Aqueous Solution Of Hz
Alright, settle in, grab your latte, and let's chat about something that sounds super boring but is actually kinda… dramatic. We’re talking about Hz, a weak acid. Yeah, I know, “acid” makes you think of bubbling vats and mad scientists, right? But Hz? Oh, Hz is more like the shy kid in the back of the class who might get picked on by the bullies (which are, in this case, other, stronger acids). It’s the acid that whispers rather than shouts, the one that’s just trying its best in a tough neighborhood.
So, what's the deal with Hz being a "weak acid"? Imagine you've got a superpower. Some acids have the superpower of completely dissing water molecules. They go full Hulk smash, ripping apart every single water molecule they touch, which is how they get their strong, "I mean business" vibe. We call these the strong acids. Think of them as the Avengers of the chemical world – big, bold, and always ready for a chemical showdown. They're like, "Water molecules? More like… gone-water molecules!"
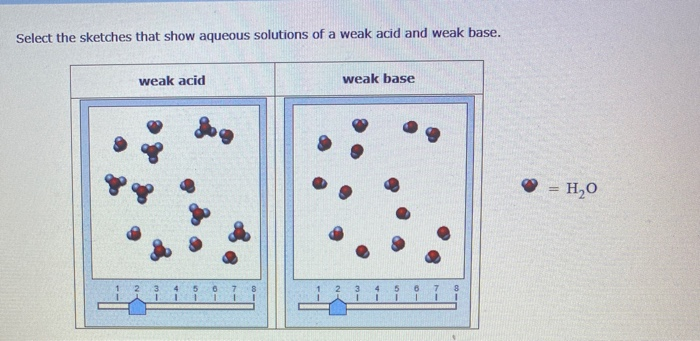
But Hz? Hz is more like… Aquaman on a bad hair day. It can interact with water, it can break apart some water molecules, but it’s not exactly going full Poseidon’s Fury. It’s more of a… polite nudge. It’s like Hz dips its toe in the water and thinks, "Hmm, this is nice," and maybe, just maybe, a couple of water molecules go, "Whoa, what was that?" and decide to hang out with Hz for a bit. The vast majority of Hz, however, are perfectly happy chilling out as their original Hz selves, hanging out with their Hz buddies, and generally minding their own business.
Must Read
This whole "dipping a toe" thing is what makes Hz a weak acid. It doesn’t fully, completely, enthusiastically break apart in water. It’s more of a reluctant agreement. When you plonk Hz into an aqueous solution – that’s fancy talk for "water" – only a small percentage of the Hz molecules decide to get all ionic. They'll donate a tiny proton (which is basically a positively charged hydrogen atom, the superhero sidekick of chemistry) to a water molecule, turning that water molecule into a hydronium ion (H₃O⁺). The remaining Hz molecules? They just shrug and say, "Eh, not today, protons."
Think of it like this: you’re at a party, and there’s a really popular dance move. A strong acid is like the person who instantly nails that dance move, every single time. A weak acid like Hz is like the person who tries it, stumbles a bit, maybe gets a few steps right, but mostly just does their own thing awkwardly in the corner. It's not that they can't do it, it's just… they're not really feeling it. They're more of a "two-step and a nod" kind of acid.

This reluctance to fully commit to breaking apart is why Hz is considered weak. It's not about being ineffective, mind you. Even a weak acid can get things done. It's just that it doesn't go around causing a massive chemical kerfuffle. It's the quiet achiever. You wouldn't use Hz to, say, dissolve a stubborn padlock. You’d probably want something with a bit more… oomph. But for gentler tasks, for a subtle pH adjustment, or for reactions where you don't want a riot of ions? Hz might just be your guy.
Now, let's talk about this aqueous solution of Hz. When we say "aqueous," we're just saying that Hz has been dissolved in water. And because Hz is a weak acid, this solution is going to be a bit of a mixed bag. You'll have some Hz molecules just hanging out, being all Hz-like. You'll also have a small number of those proton-donating Hz molecules, creating those hydronium ions. And the water? Well, the water is still mostly water, just a little bit more excited because some of its buddies have been chatting with Hz.
The pH of this solution is going to be on the acidic side, meaning it'll be less than 7. But it won't be super low. Remember our awkward dancer? They're not going to make the whole dance floor go wild. Hz will bring the pH down, but it won't send it plummeting into the abyss like a hydrochloric acid party. It's like the difference between a lukewarm bath and a hot tub – both are warmer than a cold shower, but one is clearly more… intense.
Here's a fun fact that might blow your mind: some of the most common substances we use every day are weak acids! Think of the vinegar you put on your salad? That's acetic acid, a weak acid. The citric acid in your oranges? Yep, weak acid. Even the lactic acid that makes your muscles burn after a workout is technically a weak acid. So, these "weak" acids are actually super important. They're the unsung heroes of your kitchen and your body!
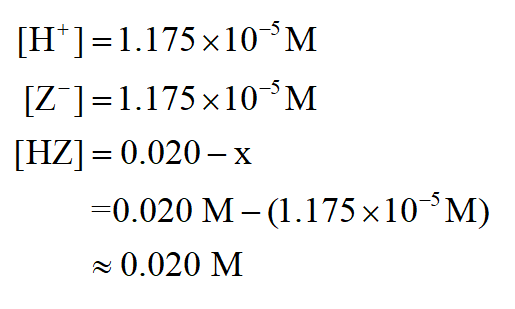
Hz, in its aqueous solution, is like a delicate ecosystem. It's a balance. You’ve got the majority of Hz molecules existing in their un-ionized form. Then you’ve got a smaller number that have decided to play chemical tag with water, forming hydronium ions and their corresponding Hz conjugate base. This equilibrium is what defines Hz as a weak acid. It's not a one-way street; it's more of a reversible handshake.
The strength of an acid is often measured by its acid dissociation constant, or Ka. A really tiny Ka value? That means the acid is a real wimp. It barely dissociates at all. A larger Ka? That acid is a bit more assertive. For Hz, we'd expect a Ka value that’s not astronomically small, but definitely not massive. It's in that sweet spot of "just strong enough to be considered an acid, but not strong enough to start a chemical revolution."
So, the next time you hear about Hz being a weak acid in an aqueous solution, don't picture a dangerous chemical. Picture a shy but friendly participant in the grand ballroom of chemistry. It’s not going to win any awards for acidity, but it definitely has its own unique charm and plays a vital role. It's the quiet hum in the background, the subtle flavor, the gentle nudge that keeps things from getting too out of hand. And honestly? Sometimes, in life, being a little bit weak is exactly what you need to be.


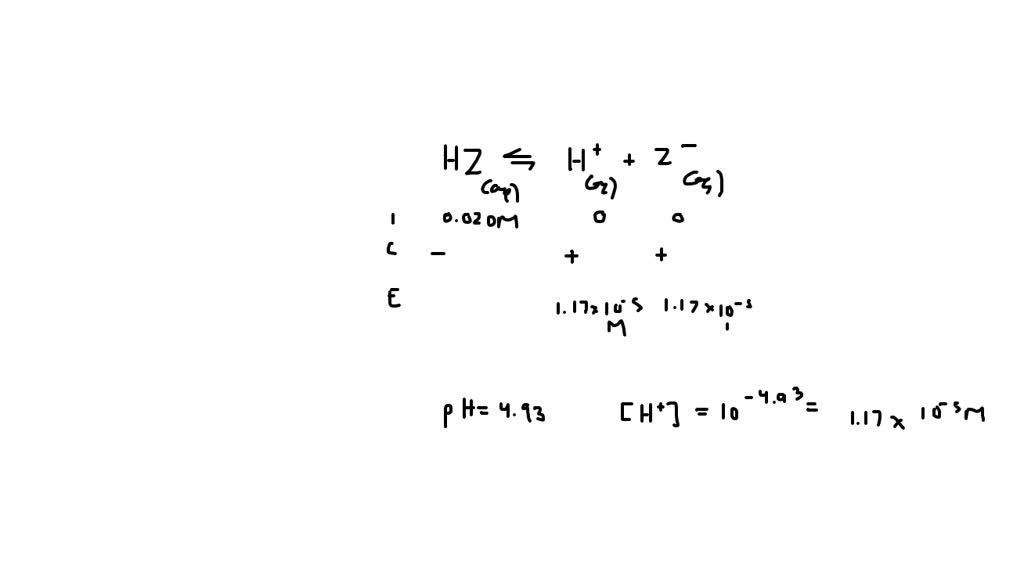
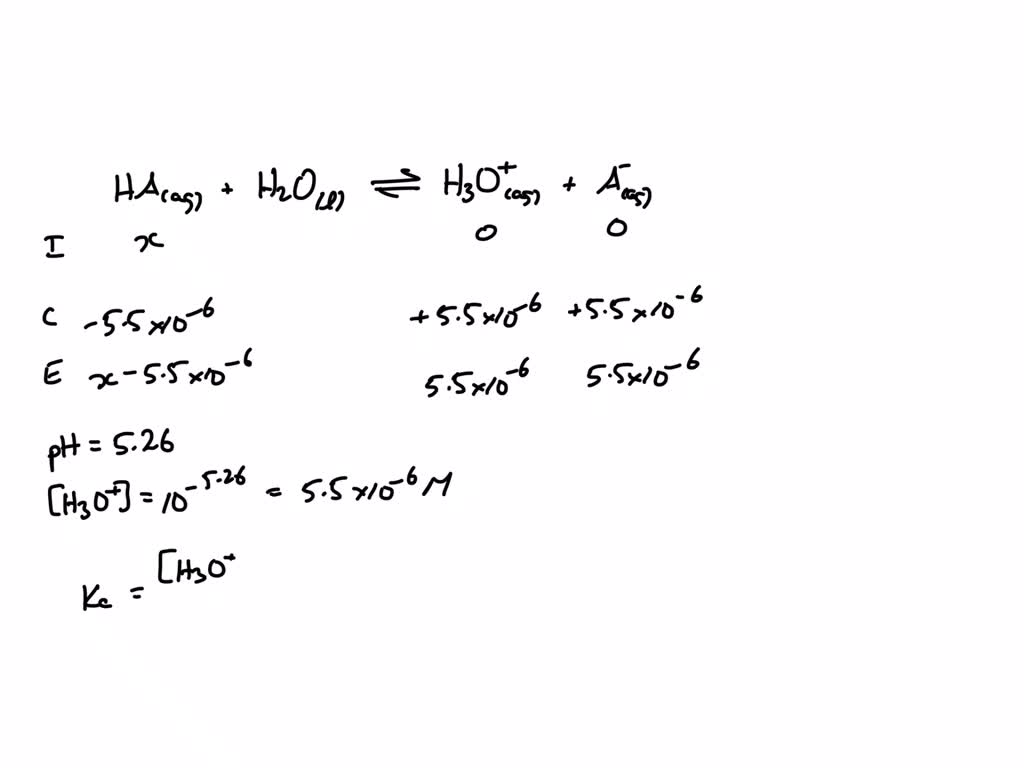

:max_bytes(150000):strip_icc()/list-of-strong-and-weak-acids-603642-v2copy2-5b47abd0c9e77c001a395e55.png)