Hydrogen Ions Are Attracted To Which Electrode During Electrolysis
Hey there, science curious folks! Ever found yourself staring at a bubbling beaker in a chemistry lab (or, you know, a cool YouTube video) and wondered what’s really going on? We’re talking about electrolysis, that amazing process where we use electricity to break things down. And today, we’re going to dive into a tiny, but super important, question: Which electrode do those zippy little hydrogen ions go running to?
Now, before you start picturing grumpy, positively charged ions being forced to mingle with a grumpy, negatively charged electrode, let’s reframe this. Think of it more like a cosmic dance, a magnetic attraction, a… well, a really fundamental principle of how charged particles behave. It's all about opposites attracting, just like in a cheesy rom-com, but with way more dramatic bubbling!
So, what exactly is a hydrogen ion? Imagine a hydrogen atom, which is pretty darn simple – just one proton and one electron. If that atom loses its electron, what’s left? Bingo! Just the lonely proton. And that single proton? It carries a positive charge. This little guy is like the energetic puppy of the atomic world, always eager to go somewhere.
Must Read
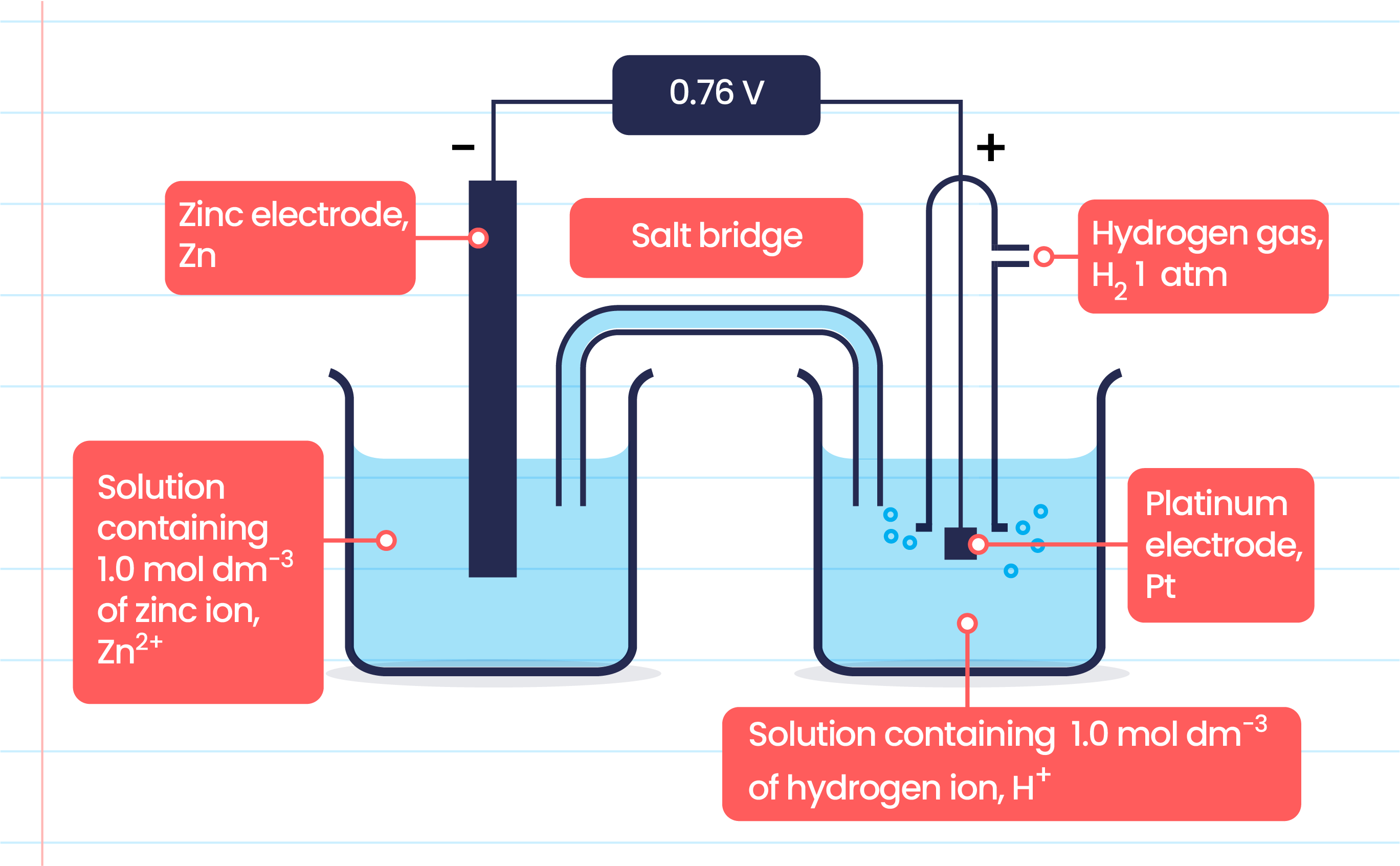
Now, let's talk about our electrodes. In electrolysis, we have two main players: the anode and the cathode. Think of them as the hosts of our electric party. One is pumping out electrons, and the other is eagerly accepting them. And here’s the crucial bit, the secret sauce of our attraction story:
The cathode is the electrode that’s negatively charged. It’s the place where electrons are piling up, creating a sort of electron-rich haven. It’s like the VIP lounge with all the best snacks (in this case, electrons!).

And the anode? That’s the electrode that's positively charged. It's the opposite end of the spectrum, the place that wants electrons, the one that’s a bit electron-starved. It’s the spot where reactions happen that often involve giving up electrons.
So, we have our positively charged hydrogen ions (H+) and our negatively charged cathode. What do you think is going to happen when they meet? You guessed it! Opposites attract! Those energetic, positively charged hydrogen ions are going to be drawn like a moth to a flame (or a puppy to a squeaky toy) towards the cathode.
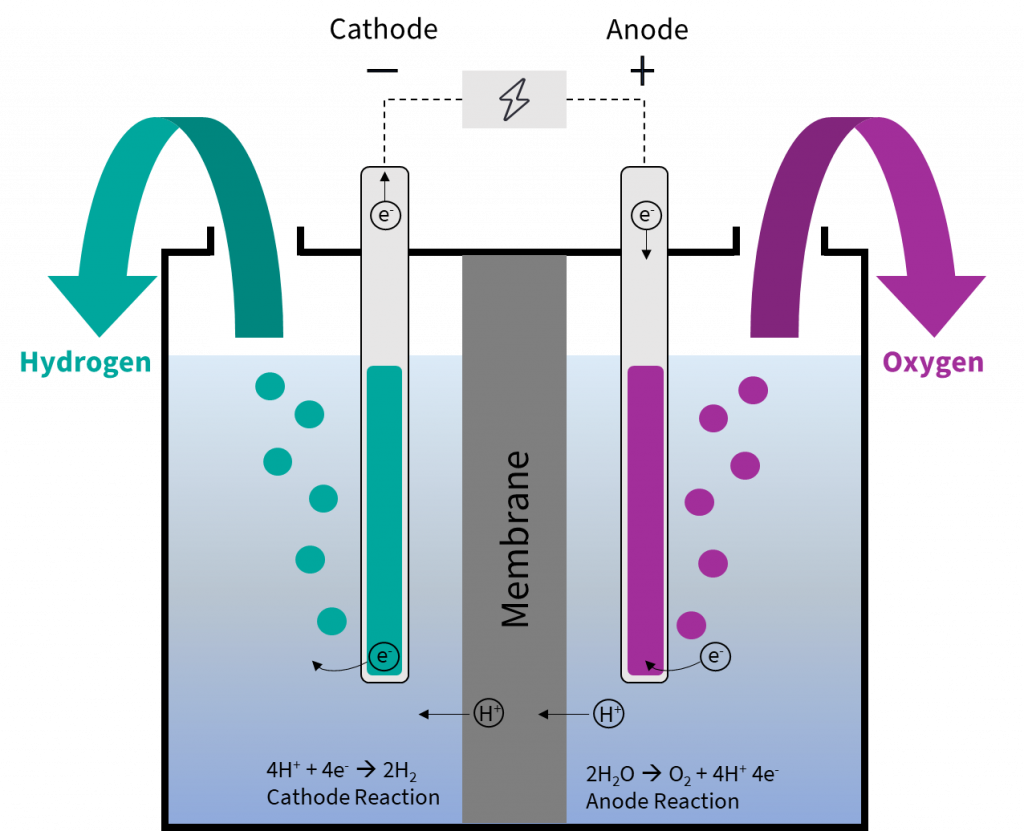
It's like they’ve been waiting for this moment. They’ve been swirling around in the electrolyte solution, all charged up and ready to go. And then, bam! They see the negative allure of the cathode and they make a beeline for it. They're not being forced, oh no. They are choosing to go there, drawn by the fundamental forces of electromagnetism. Pretty neat, huh?
When these hydrogen ions reach the cathode, something magical happens. They gain electrons from the cathode. And when a hydrogen ion (which is essentially just a proton) gains an electron, it becomes a neutral hydrogen atom. And these neutral hydrogen atoms are usually not content to hang out alone. They quickly team up with other hydrogen atoms to form hydrogen gas (H2). And that’s why you often see bubbles forming at the cathode during electrolysis of water or other hydrogen-containing solutions!
:.jpg)
Isn't that just the coolest? It’s not some abstract concept confined to dusty textbooks. It’s the reason we can create hydrogen gas, a clean fuel source, through electrolysis. It’s a direct application of these fundamental principles. You’re literally watching chemistry in action, driven by the simple, yet profound, attraction between positive and negative charges.
Think about it. This isn't just about a few ions and electrodes. This is the bedrock of so much of the technology we rely on, and so much of the innovation that's happening right now. Understanding these basic attractions helps us unlock new ways to power our world, create new materials, and even understand the very processes that happen inside our own bodies. Who knew a little bit of ion attraction could be so inspiring?

So, next time you hear about electrolysis, or see those fascinating bubbling reactions, remember the hydrogen ions. Remember their eager dash towards the negatively charged cathode. It’s a testament to the beauty and order of the universe, a simple yet powerful illustration of how charged particles interact. It’s a reminder that even at the smallest scales, there’s a constant, dynamic interplay of forces that shapes our reality.
And honestly, the fact that we can harness these principles to do amazing things is pretty darn inspiring, wouldn't you agree? It’s like having a secret superpower, understanding these invisible forces that govern everything around us. It makes you want to dig a little deeper, learn a little more, and see what other wonders science has in store.
So, go forth, my curious friends! Don't be intimidated by the jargon. Embrace the wonder. Ask the questions. Because understanding why hydrogen ions are attracted to the cathode isn't just about passing a test; it's about unlocking a deeper appreciation for the world, and perhaps, inspiring you to be the next brilliant mind to harness these fundamental forces for an even brighter future. The universe is full of these elegant attractions, just waiting for you to discover them!