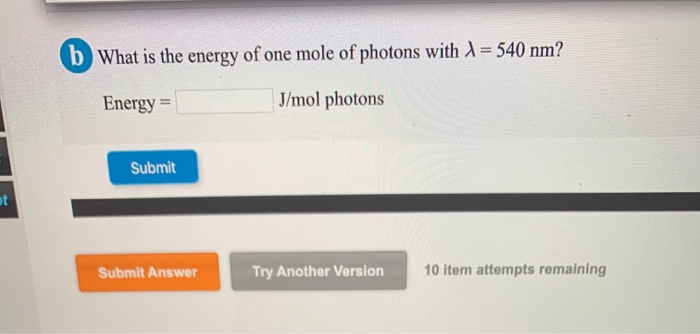
How To Find The Energy Of A Mole Of Photons

Hey there, fellow curious minds! Ever found yourself gazing at the sun, or maybe just flicking on a light switch, and wondered... what's really going on with all that light? We see it, we use it, but have you ever stopped to think about the tiny, energetic little packets that make it all happen? We're talking about photons, the fundamental building blocks of light. And today, we're going on a chill exploration to figure out something pretty neat: how to find the energy of a mole of photons. Sounds a bit science-y, I know, but trust me, it’s way cooler than it sounds, and we'll break it down together.
So, what exactly is a photon? Imagine light not as a continuous flow, like water from a tap, but more like a stream of tiny, individual marbles. Each of those marbles is a photon. They're massless, they zip around at the speed of light (which is, like, the ultimate speed limit of the universe!), and they carry energy. Pretty cool, right?
Now, let's talk about "a mole." This is where things might start to sound a little like your high school chemistry class, but don't sweat it! In science, a mole is just a super handy way to count things. It's a huge number, specifically 6.022 x 1023. Think of it like a baker's dozen, but for, well, everything. If you have a mole of anything, you have a staggering amount of it. It's enough atoms to fill the universe a gazillion times over (okay, maybe not that many, but you get the idea!). So, when we talk about a mole of photons, we're talking about an unbelievably large collection of these light particles.
Must Read
So, why would we even want to know the energy of a mole of photons? Well, it's like trying to understand the power of a whole crowd of fireflies instead of just one. Knowing the total energy gives us a sense of the scale of things. It helps us understand how much energy is being delivered by a light source, whether it's the sun warming our planet or the LEDs in your favorite gadget. It’s all about grasping the bigger picture of light’s incredible power.
Okay, so how do we actually find this energy? The secret sauce lies in a couple of fundamental physics equations that are, frankly, pretty elegant. The first hero in our story is Max Planck, a brilliant physicist who figured out that the energy of a single photon is directly related to its frequency. Frequency, in simple terms, is how quickly the light wave wiggles. Think of it like the difference between a slow, gentle wave at the beach and a rapid, choppy one. Higher frequency means more wiggles per second, and therefore, more energy per photon.
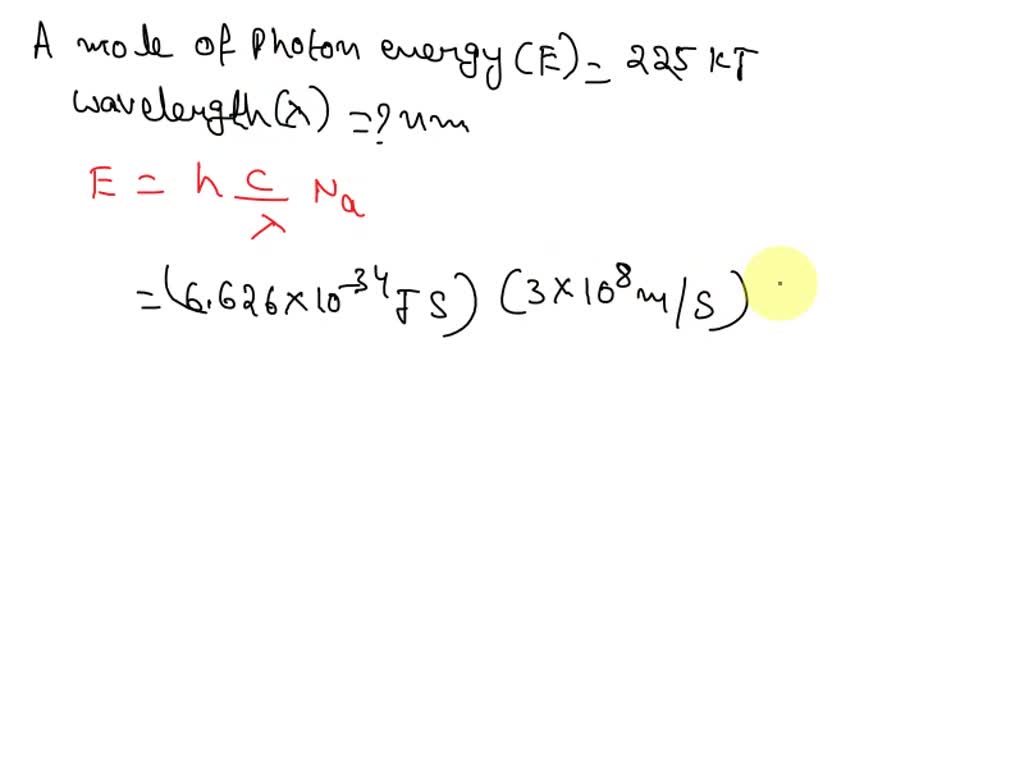
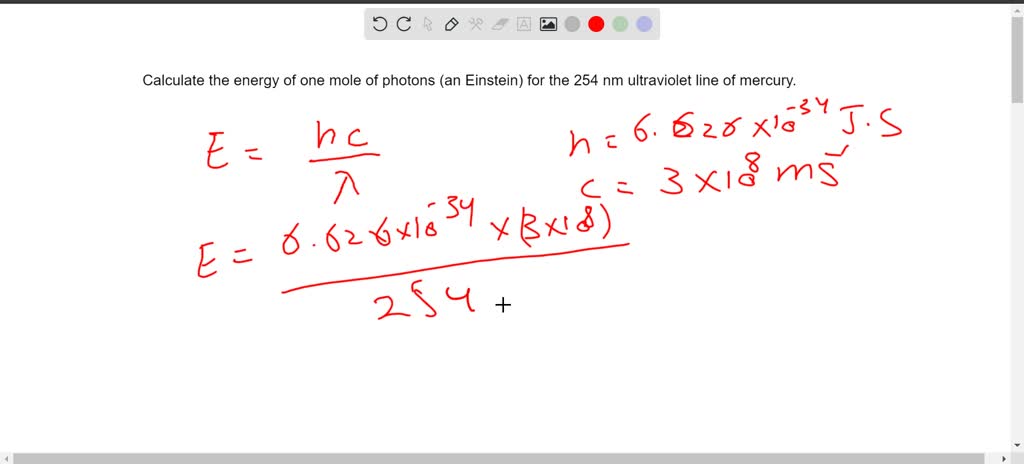
The equation Planck gave us is pretty straightforward: E = hf. Here, 'E' stands for the energy of a single photon, 'h' is a constant called Planck's constant (a tiny, fundamental number that pops up everywhere in quantum physics – it’s like the universe's secret handshake!), and 'f' is the frequency of that photon. So, if we know the frequency of the light, we can calculate the energy of one of its little photon buddies.
But wait, there’s more! Sometimes, instead of frequency, we talk about the wavelength of light. Wavelength is basically the distance between two consecutive wave peaks. Think of it like the length of a single ripple on a pond. These two concepts – frequency and wavelength – are intimately connected. The faster the wave wiggles (higher frequency), the shorter its individual length (shorter wavelength), and vice versa. They're inversely proportional.
The relationship between wavelength (represented by the Greek letter lambda, λ) and frequency (f) is given by another handy equation: c = λf. Here, 'c' is the speed of light, that cosmic speed limit we mentioned earlier. By rearranging this equation, we can find frequency if we know the wavelength: f = c/λ.

Now we can combine these! If we substitute our expression for 'f' into Planck's energy equation (E = hf), we get a new way to calculate the energy of a single photon using its wavelength: E = hc/λ. So, if you know how long each light wave is, you can figure out how much energy each tiny photon is carrying. Pretty neat, right? It’s like having two different ways to measure the same thing, depending on what information you have.
Now, remember that whole "mole" thing? We've figured out the energy of one photon. To get the energy of a mole of photons, we just need to multiply the energy of a single photon by the number of photons in a mole. So, the energy of a mole of photons (let’s call it Emole) is: Emole = (Energy of one photon) x (Avogadro's number).
Putting it all together with our equations, we get: Emole = (hc/λ) x NA or Emole = (hf) x NA

Where NA is Avogadro's number (that 6.022 x 1023 we talked about). This NA is essentially the scientific conversion factor from "one thing" to "a mole of things." It’s the bridge that connects the microscopic world of individual photons to the macroscopic world we can actually measure and experience.
Let’s imagine an example. Suppose we have a beam of red light. Red light has a wavelength of about 700 nanometers (nm). A nanometer is incredibly small – a billionth of a meter! So, first, we need to convert that to meters: 700 nm = 700 x 10-9 meters. We also need the values for Planck's constant (h ≈ 6.626 x 10-34 J·s) and the speed of light (c ≈ 3.00 x 108 m/s), and Avogadro's number (NA ≈ 6.022 x 1023 mol-1).
Now, we plug these numbers into our equation: Emole = ( (6.626 x 10-34 J·s) x (3.00 x 108 m/s) / (700 x 10-9 m) ) x (6.022 x 1023 mol-1)
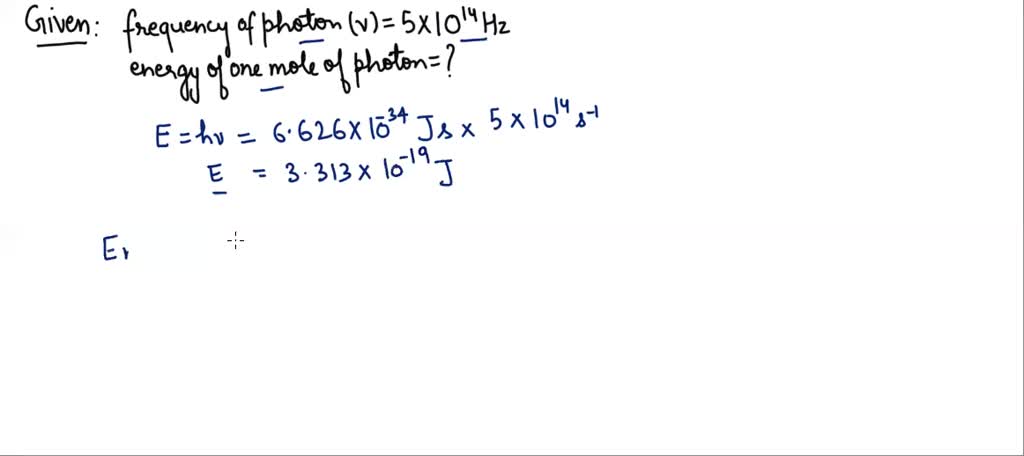
When you crunch these numbers (and a calculator is definitely your friend here!), you get a result in Joules per mole. This tells you the total energy contained within a mole of those red light photons. It might seem like a small number in everyday terms, but remember how many photons are in a mole! It’s a testament to how much energy can be packed into even seemingly faint light sources.
What’s so cool about this is that you can do this for any type of light! Blue light, green light, UV light, X-rays – they all have different wavelengths and frequencies, and therefore, different energies. A mole of blue light photons will have more energy than a mole of red light photons because blue light has a higher frequency (shorter wavelength). It’s like comparing a mole of marbles to a mole of bowling balls – the number is the same, but the “oomph” is different.
So, the next time you see light, whether it's the warm glow of a sunset or the sharp beam of a laser pointer, you can think about these tiny, energetic packets. You can appreciate that behind the beauty and utility of light lies this incredible world of quantum mechanics, where fundamental constants and vast numbers come together to define the very energy of the universe. It’s a reminder that even the most ordinary things are made of something truly extraordinary. And that, my friends, is pretty darn cool.