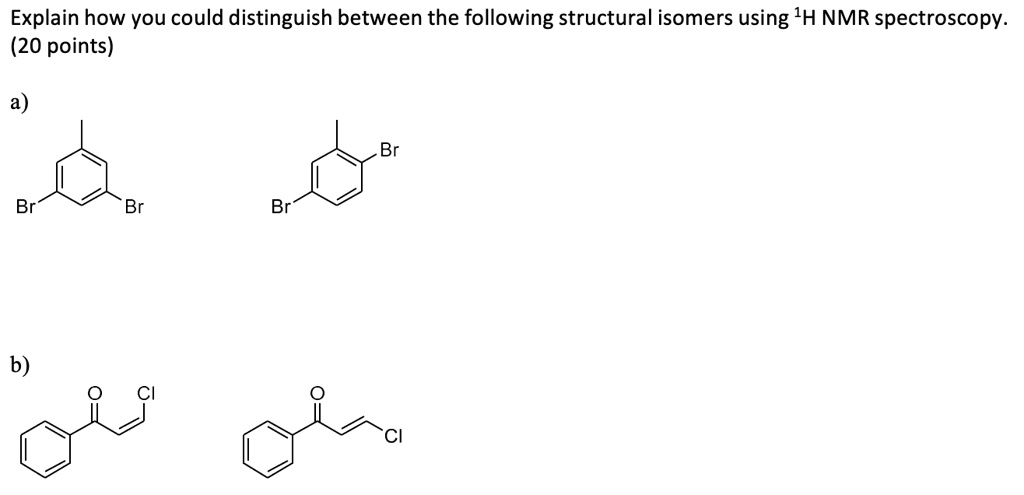
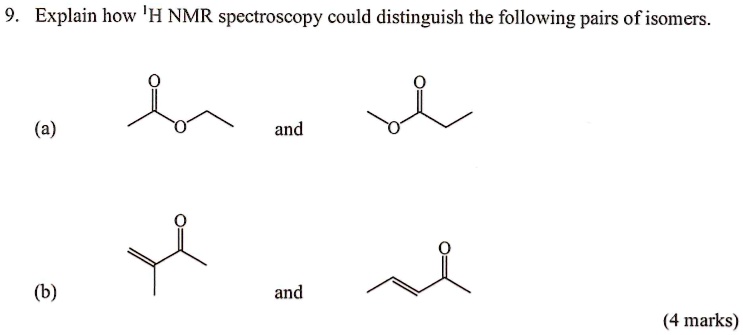
How To Distinguish E And Z Isomers By Nmr
Ever wondered how scientists peek into the secret lives of molecules? It’s like being a detective, but with super-powered vision! Today, we're going to talk about a cool trick called NMR spectroscopy. Think of it as a molecular MRI machine. It lets us see the tiny details of how atoms are arranged.
Specifically, we're diving into the fun world of E and Z isomers. These are like molecular twins that look very similar but have a crucial difference. It’s all about how their parts are arranged in space. And NMR is our special tool to tell them apart.
This might sound a bit technical, but trust me, it’s surprisingly entertaining. It’s like solving a fun puzzle. We get to figure out which twin is which, and NMR gives us the clues. It’s a bit like having a secret handshake for molecules.
Must Read
The Mysterious Case of E and Z Isomers
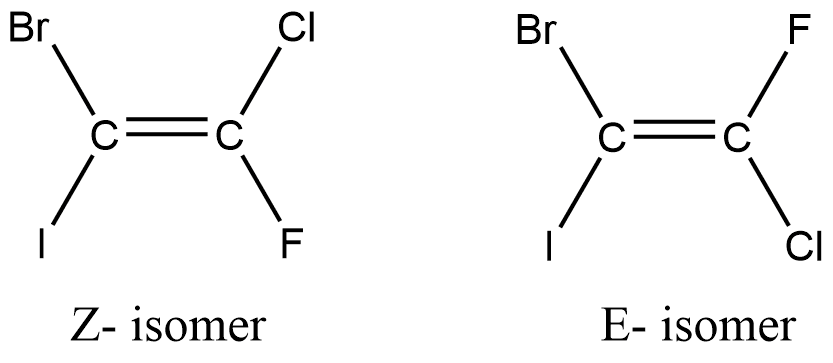
So, what exactly are these E and Z isomers? Imagine you have a molecule with a double bond. This double bond is like a rigid connection. It means the atoms attached to it can't just spin around freely.
Think of it like two people holding hands tightly. They can't easily swap places. The atoms on either side of the double bond are stuck in their positions relative to each other. This leads to different spatial arrangements.
We call these different arrangements isomers. And for molecules with a double bond, we often use the terms E and Z to describe them. It’s a system based on priority rules, which we’ll touch on briefly.
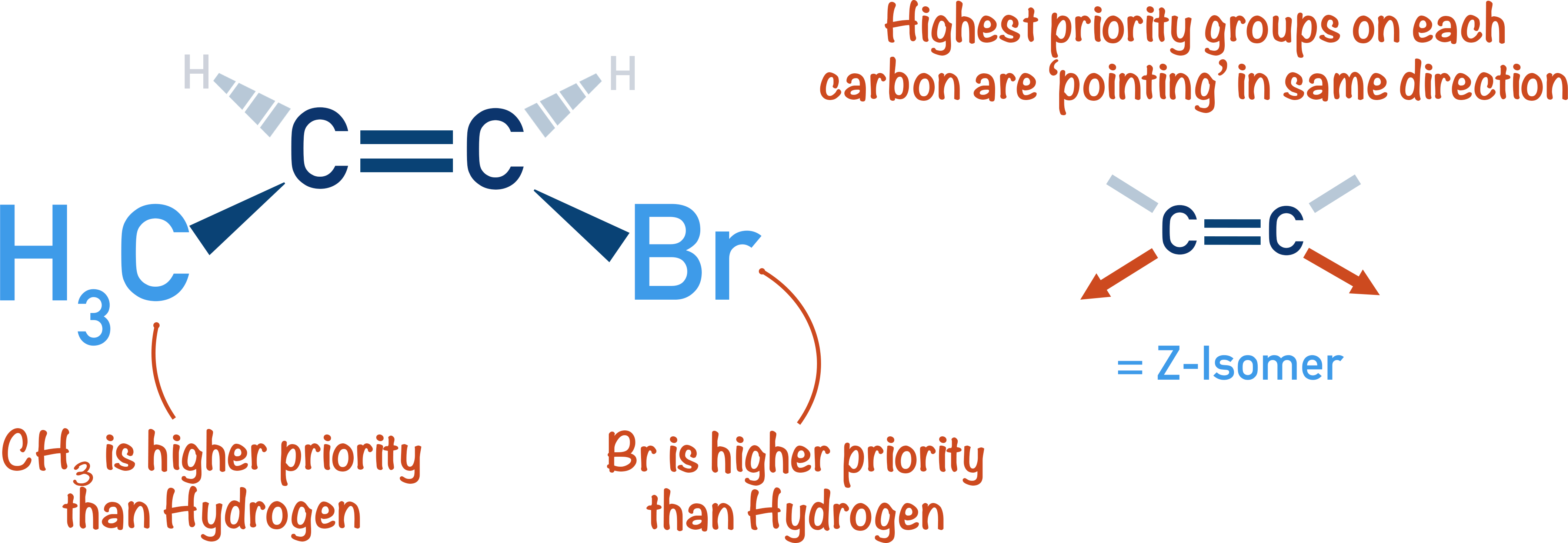
The E comes from the German word entgegen, meaning "opposite." The Z comes from zusammen, meaning "together." So, in E isomers, the higher-priority groups are on opposite sides of the double bond. In Z isomers, they are on the same side.
This difference might seem small, but it can have huge effects on a molecule’s properties. Things like how it reacts, how it smells, or even how it works in your body can change. That’s why distinguishing them is so important.

NMR: Our Molecular Magnifying Glass
Now, how do we actually see this difference using NMR spectroscopy? This is where the magic happens! NMR works by using a strong magnetic field and radio waves. It makes the nuclei of certain atoms, like hydrogen (protons), get excited.
When these excited nuclei relax back down, they give off signals. These signals are like fingerprints for the atoms. The exact signal we get depends on the atom’s environment. And its environment is determined by the other atoms around it and how they are arranged.
This is the key! The arrangement of atoms in E and Z isomers creates slightly different environments for the nuclei. So, their NMR signals will be different. It’s like two identical-looking people wearing slightly different colored shirts. NMR can tell them apart.
For distinguishing E and Z isomers, we often focus on the hydrogen atoms. Specifically, we look at the hydrogen atoms attached to the carbons of the double bond. Their positions relative to each other in space matter.
In an E isomer, the two hydrogen atoms might be further apart from each other. In a Z isomer, they might be closer. This spatial difference affects how they "feel" the magnetic field. It’s like they are whispering to the NMR machine in slightly different ways.
The "How-To" - A Peek into the Spectrograph
So, how do we interpret these whispers? When we run a sample in an NMR spectrometer, we get a spectrum. This spectrum is a graph with signals at different positions. These positions are called chemical shifts.
The chemical shift tells us about the electronic environment of the atom. For hydrogens on a double bond, their chemical shift can be influenced by neighboring groups and their relative positions. This is where the E and Z distinction comes into play.
One of the most common ways to distinguish E and Z isomers using NMR is by looking at the coupling constants. Coupling is a phenomenon where the signal of one nucleus is split by the signal of a nearby nucleus. This splitting pattern gives us information about the distance and orientation between the nuclei.
For hydrogens attached to the carbons of a double bond, the size of the coupling constant between them is highly dependent on their spatial relationship. In E isomers, the hydrogens are further apart, and this leads to a smaller coupling constant. You’ll see a smaller "split" in their signals.
In Z isomers, the hydrogens are closer together. This proximity results in a larger coupling constant. Their signals will be split more noticeably. It's like the closer friends are, the more they influence each other.
Imagine two siblings who are always bickering. They are close, and their arguments are loud and noticeable (large coupling constant). Now imagine two cousins who only see each other at holidays. They are further apart, and their disagreements are quieter and less frequent (small coupling constant).

So, by carefully measuring these coupling constants in the NMR spectrum, we can directly tell if we have an E isomer or a Z isomer. It’s a direct observation of their spatial arrangement.
Beyond coupling constants, sometimes the chemical shifts themselves can offer clues. Depending on the other groups attached to the double bond, the hydrogens in an E isomer might have slightly different chemical shifts compared to those in a Z isomer. This is a bit more subtle and depends on the specific molecule.
Think of it like this: if the two hydrogen atoms are on opposite sides (E), their influence on the electron clouds around them might be different than if they are on the same side (Z). This subtle difference can be picked up by the sensitive NMR machine.
Why This is So Entertaining and Special
What makes this whole process so entertaining? It’s the thrill of the chase! We’re essentially solving a molecular mystery. We have these two seemingly identical molecules, and we need to figure out their secret spatial arrangement.
NMR spectroscopy is our detective kit. It provides the hard evidence. We don't just guess; we have concrete data. The peaks, the splits, the chemical shifts – they are all clues.
It’s special because it allows us to see beyond the simple chemical formula. A formula like C4H8 tells you what atoms are present and how many. But it doesn’t tell you how they are arranged in 3D space. NMR does!

This ability to discern spatial arrangements is crucial. In drug design, for example, the shape of a molecule is everything. A slightly different arrangement can mean the difference between a medicine that works and one that’s completely ineffective, or even harmful.
The elegance of using NMR to distinguish E and Z isomers is also part of its charm. It’s a powerful technique that relies on fundamental physics and chemistry principles. Yet, the interpretation can feel very intuitive once you understand the basics.
It’s like learning a new language. At first, it seems daunting. But then, you start to understand the grammar and vocabulary. Suddenly, you can converse with the molecules! You can ask them, "Are you E or are you Z?" and they will tell you.
And the feeling of cracking the code? It's incredibly satisfying. You've taken raw data from a machine and transformed it into meaningful information about molecular structure. It’s a small victory, but a significant one in the world of chemistry.
So, the next time you hear about NMR spectroscopy and E and Z isomers, remember it's not just dry science. It’s a fascinating detective story playing out at the molecular level. It’s about using incredibly sophisticated tools to uncover the subtle yet important differences that shape the world around us. It’s a testament to human curiosity and our ability to understand the universe, one molecule at a time.
It’s a wonderful way to appreciate the complexity and beauty of the molecular world. And the fact that we can solve these puzzles with such precision is truly something to marvel at. It’s a blend of art and science, where the spectrum is your canvas and the signals are your brushstrokes.