How To Calculate The Relative Formula Mass

Hey there, science buddy! Ever looked at a bunch of atoms hanging out together, all chummy-like, and wondered how much "stuff" they collectively weigh? Like, what's the total heft of that little molecular party? Well, get ready, because we're about to dive into the super-duper fun world of calculating Relative Formula Mass! Don't let the fancy name scare you; it's basically just figuring out the total awesome weight of a chemical compound. Think of it as giving a chemical formula its own personal weight tag!
Why is this even cool? Because chemistry isn't just about bubbling beakers and explosions (though that's fun too!). It's about understanding the building blocks of everything. And knowing their weights? That's like knowing how many ingredients go into your favorite cookie recipe. You gotta know the flour, sugar, and chocolate chips to bake that masterpiece, right? Molecules are no different!
So, what's the deal with "relative"? It means we're comparing things. We're not using pesky kilograms or grams just yet. We're using a special unit called the Atomic Mass Unit (amu). Imagine it's like a tiny, universal LEGO brick. All other atomic weights are measured against how many of these tiny bricks they're equivalent to. Pretty neat, huh?
Must Read
Now, to do any of this weight-finding business, you absolutely must have a trusty sidekick: the Periodic Table. This isn't just a poster on a classroom wall; it's like a cheat sheet for the universe! Each element has its own little box, and tucked away inside is its Atomic Mass. This is the number we're hunting for!
Let's Get Down to Business: The Simplest Compounds
Let's start with something easy. Think of a simple molecule, like water. You know, H₂O? That classic. We've got two hydrogen atoms (H) and one oxygen atom (O) doing a little dance together.
First step: find your ingredients. Grab that Periodic Table! Look up Hydrogen (H). See that little number, usually at the bottom? That's its Atomic Mass. For Hydrogen, it's approximately 1.01 amu. We usually round this to 1 for simplicity in basic calculations, but let's keep the 1.01 for a bit of precision. It's like choosing between "about an hour" or "exactly 63 minutes" for your friend’s arrival time. Both are useful!
Next, find Oxygen (O). Its Atomic Mass is roughly 16.00 amu. Again, often rounded to 16. Easy peasy!
Now, let's do the math. Remember, the "2" in H₂O means we have two hydrogen atoms. So, we multiply the Atomic Mass of hydrogen by 2. That's 2 × 1.01 amu = 2.02 amu.

Then, we add the weight of the oxygen atom. We only have one oxygen, so it's just 1 × 16.00 amu = 16.00 amu.
Drumroll please... To get the Relative Formula Mass of water, we add these two parts together: 2.02 amu + 16.00 amu = 18.02 amu. So, one molecule of water weighs about 18.02 of those tiny atomic mass units. Boom! You just calculated the Relative Formula Mass of water!
What About When Things Get a Little More Complicated?
Sometimes, molecules are like bigger, fancier parties. They have more types of atoms, or more of the same type. Let's take Carbon Dioxide (CO₂). We've got one Carbon (C) and two Oxygens (O).
Find Carbon (C) on the Periodic Table. Its Atomic Mass is around 12.01 amu. Let's call it 12.
We already know Oxygen (O) is about 16.00 amu. Let's call it 16.
So, we have one Carbon: 1 × 12 amu = 12 amu.
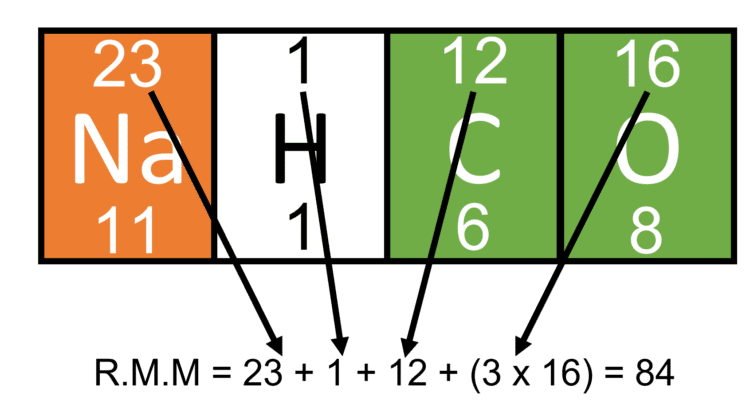
And two Oxygens: 2 × 16 amu = 32 amu.
Add them up: 12 amu + 32 amu = 44 amu. That's the Relative Formula Mass of carbon dioxide. See? You're a natural!
The Quirky Bits and Bobs
Here's a fun fact: Relative Formula Mass is often called Relative Molecular Mass for molecules made of non-metals, like water and carbon dioxide. When we're talking about compounds made of metals and non-metals, especially those that form crystal lattices (think salt!), we tend to use "Relative Formula Mass." It sounds a bit more technical, but it's just about how the atoms are arranged. Don't stress it too much; the calculation is the same!
Another quirky detail: those Atomic Masses on the Periodic Table? They're not always whole numbers! Why? Because most elements exist as different versions called isotopes. These isotopes have the same number of protons but different numbers of neutrons, making them slightly heavier. The number you see is an average weight of all those isotopes. It's like saying the average height of people in a room isn't a perfect whole number of inches. Chemistry is full of averages and delightful complexities!
Why is this stuff important, beyond satisfying your curiosity? Well, scientists use this to figure out how much stuff they need for experiments. If you want to make a certain amount of a new material, knowing the Relative Formula Mass helps you calculate exactly how much of each ingredient you need. It's like a cosmic cookbook!

Let's Tackle a Slightly Trickier One: Magnesium Chloride (MgCl₂)!
Here's where things get a tiny bit more interesting. Magnesium Chloride has one Magnesium (Mg) and two Chlorine (Cl) atoms.
Find Magnesium (Mg) on your Periodic Table. Its Atomic Mass is about 24.31 amu. Let's use 24.3 for a bit of accuracy.
Now, find Chlorine (Cl). Its Atomic Mass is around 35.45 amu. Let's use 35.5.
So, for Magnesium: 1 × 24.3 amu = 24.3 amu.
And for Chlorine: we have two chlorine atoms. So, 2 × 35.5 amu = 71.0 amu.
Add them together: 24.3 amu + 71.0 amu = 95.3 amu. Ta-da! That’s the Relative Formula Mass of magnesium chloride.
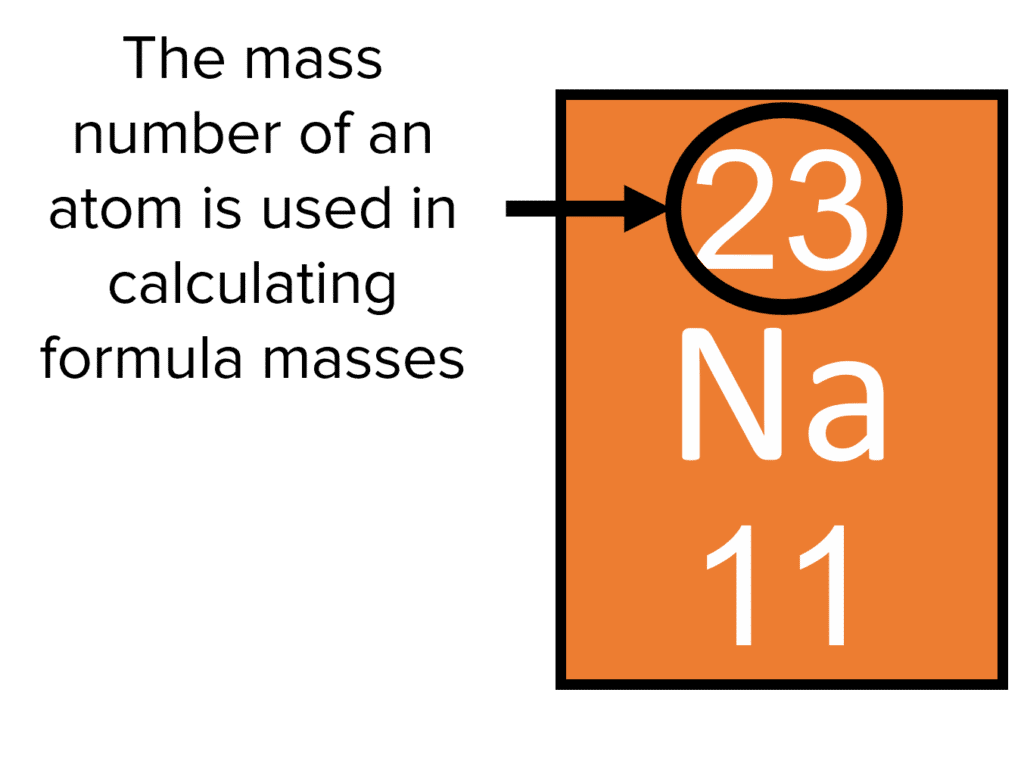
A Note on Units
You might see this number expressed as just a number, without the "amu." This is perfectly fine! When we talk about Relative Formula Mass, the "relative" part means it's a ratio, so the units kind of cancel out. It's like saying one thing is "twice as heavy" as another. You don't necessarily need the grams or pounds for that comparison. So, you might see the answer as 18.02, 44, or 95.3. It all means the same thing!
It's also super helpful to remember that 1 mole of any substance has a mass in grams that is numerically equal to its Relative Formula Mass. So, 1 mole of water weighs 18.02 grams. Mind-blowing, right? But don't let that deep dive scare you! For now, just focus on the calculation and the fun of it.
The Fun Doesn't Stop!
The more atoms in a compound, the more calculations you do. Think of compounds with lots of parentheses, like Calcium Hydroxide, Ca(OH)₂. That little "2" outside the parentheses means you have to multiply everything inside by two. So, you'd have one Calcium, two Oxygens, and two Hydrogens. It’s like a multi-step puzzle!
The key is to always, always, always check your Periodic Table for the Atomic Masses and count how many of each atom you have in the chemical formula. Get those two things right, and you're golden.
So, there you have it! Calculating Relative Formula Mass is your gateway to understanding the weights of the tiny particles that make up our universe. It’s a fundamental skill, yes, but it’s also a little game of numbers and atoms. It’s a peek behind the curtain of how matter works.
Keep practicing, and you’ll be a Relative Formula Mass whiz in no time. Go forth and weigh some molecules! It’s more fun than it sounds, promise!


