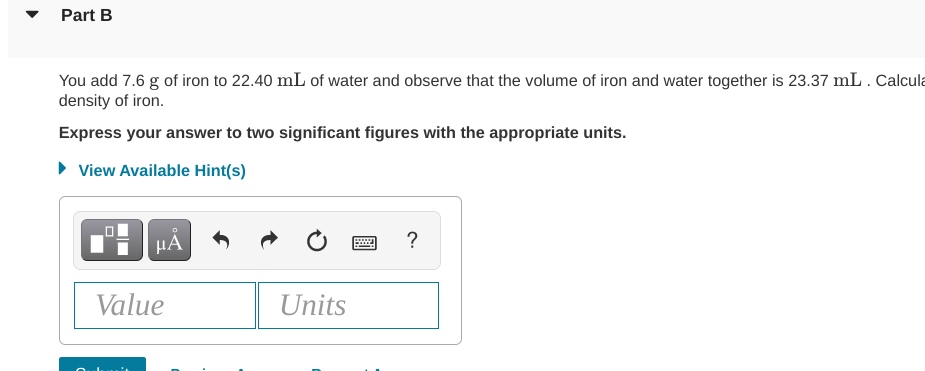
How Much Water Did 15.0 G Of Iron Displace

So, you've got a bit of iron. Just 15.0 grams of it, to be exact. And this little bit of metal decided to take a dip in some water. Now, the burning question, the one that keeps scientists up at night (or maybe just me on a slow Tuesday): how much water did it push out of the way? It's a classic science conundrum, right?
Think of it like this. You're trying to cram yourself into a tub that's already pretty full. What happens? Water goes everywhere! Your amazing body, in all its glory, takes up space. That 15.0 grams of iron? It's doing the same thing in the water. It has its own little volume, its own personal bubble of space it needs.
This isn't about how heavy the iron is, mind you. That's its mass. We're talking about how much room it occupies, its volume. It’s like comparing a fluffy cloud to a tiny pebble. Both might weigh the same, but the cloud takes up way more sky!
Must Read
To figure out how much water our 15.0 grams of iron nudged aside, we need to know the iron's volume. And to get that, we need a little help from its density. Density is kind of like how "packed" something is. Is it super dense and heavy for its size, or is it airy and light?
Now, iron is pretty dense. It's not exactly floating around like a feather. It's more like a little brick, compact and solid. This means a small amount of iron will take up a noticeable amount of space. It’s not shy about occupying its territory.
The density of iron, for the curious souls out there, is approximately 7.87 grams per cubic centimeter. That's a fancy way of saying that if you took a cube of iron that's 1 centimeter on each side, it would weigh 7.87 grams. Pretty dense, right?
So, we have our mass (15.0 grams) and we have our density (7.87 g/cm³). The universe, in its infinite wisdom (and for the sake of this fun little experiment), has given us a way to connect these two: Volume = Mass / Density. It's a simple formula, but it unlocks secrets!
Let's plug in our numbers. We have 15.0 grams divided by 7.87 grams per cubic centimeter. Do the math (or, let's be honest, let your calculator do the math). What pops out?

It’s about 1.91 cubic centimeters. That’s the volume of our 15.0 grams of iron. This is the amount of space that our little iron buddy is taking up. Imagine it as a tiny, invisible blob of iron-ness.
And here’s the magical part, the really satisfying bit of physics that makes you go "aha!" When an object is placed in a liquid, it displaces an amount of liquid equal to its own volume. Archimedes knew this. He was a pretty smart cookie, that Archimedes.
So, the 15.0 grams of iron, with its volume of 1.91 cubic centimeters, has pushed aside exactly 1.91 cubic centimeters of water. It’s a perfect trade! The iron gets its space, and the water has to make way. No arguments, no fuss. Just pure, unadulterated displacement.
It’s such an elegant concept, isn’t it? You can take a solid object, measure it, and then predict precisely how much liquid it will shove out of its path. It’s like a secret handshake between solids and liquids.
Now, 1.91 cubic centimeters might not sound like a lot. You might be picturing a thimbleful, or maybe a very small shot glass. And you’d be pretty much right! It’s not like you’re going to flood your bathroom with just 15 grams of iron.
But the principle is what’s so neat. Whether it’s 15 grams of iron or 15 tons, the concept of displacement remains the same. A ship made of steel, which is much denser than water, floats because of its shape and the volume of water it displaces. It's a giant iron boat taking up a whole lot of ocean space!
![[ANSWERED] If 2 4 g of a metal displace 1 12 litre hydrogen at norm](https://media.kunduz.com/media/sug-question-candidate/20210624121450190450-1461613.jpg?h=512)
Sometimes, in science, the answers are surprisingly straightforward. You think it's going to be some incredibly complicated equation with Greek letters and things you can’t pronounce. But often, it’s just about understanding the fundamental properties of the stuff you're playing with.
So, next time you’re near a body of water and have a bit of iron lying around (which, let’s face it, is probably not a common occurrence for most of us, but a girl can dream!), you can impress your friends with this little tidbit. "Oh, you see that 15 grams of iron? It just displaced 1.91 cubic centimeters of water. Quite the mover, that one!"
It’s a fun little fact that highlights a basic but powerful scientific principle. It’s like the universe’s way of saying, "Hey, if you take up space, something else has to move!" And for our humble 15.0 grams of iron, that something else is a tiny bit of water. A very specific, precisely measured bit of water.
And while we’re on the topic of unpopular opinions in science, here’s one for you: sometimes, the simplest answers are the most elegant. We don’t always need rocket science to understand why a bit of metal makes water slosh. Sometimes, it’s just about good old-fashioned displacement.
So there you have it. 15.0 grams of iron, a little bit of density, and a whole lot of displaced water. It's a tiny victory for physics, and a fun little bit of knowledge for you and me. Go forth and impress your friends with your newfound understanding of iron-induced water acrobatics!
It really makes you appreciate how interconnected everything is. That bit of iron, sitting there, minding its own business, has a direct impact on the water around it. A tiny, measurable impact, but an impact nonetheless. It’s a microcosm of how things work in the big, wide world.
And the best part? This whole calculation, this unveiling of displaced water, is based on the inherent properties of iron. It’s not some arbitrary number we made up. It's dictated by the very nature of the element itself. That's the beauty of science, folks. It's all about understanding the rules of the game.
So, while 15.0 grams of iron might not seem like a lot, its effect on the water is quantifiable and predictable. It’s a testament to the power of scientific principles. Even the smallest objects have a story to tell, and sometimes that story involves a little bit of water getting pushed around.
It’s a fun little mental exercise, and it’s a great way to remember that mass and volume are different beasts. They play by different rules, and understanding that difference is key to unlocking many of the universe's secrets. Even the secret of how much water a tiny bit of iron can displace.
And if you’re anything like me, you’ll probably start looking at all sorts of objects and wondering, "How much water would that displace?" Suddenly, everyday objects become a lot more interesting, don’t they? A rock, a coin, even that stray bolt you found in the garage. They all have their own displacement story.
So, to recap, our 15.0 grams of iron, being the space-hogging entity it is, nudged aside a delightful 1.91 cubic centimeters of water. It's a small number, but it's an exact number, and that's what makes it so wonderfully scientific.
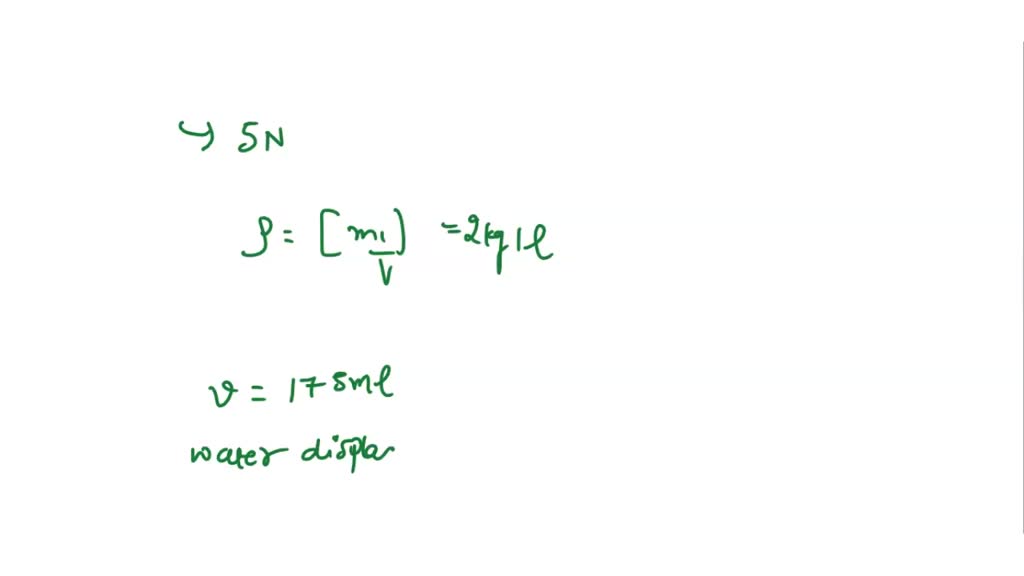
And that, my friends, is the delightful, and perhaps slightly quirky, answer to our question. It’s a reminder that science can be both profound and playfully simple, all at the same time. Who knew that 15 grams of iron could be such a conversation starter?
It's a little piece of the universe explained, one displaced droplet at a time. And isn't that just the most fun way to learn? We’re not just calculating; we’re discovering the hidden dramas of everyday objects interacting with their liquidy environments.
So, the next time you see iron, give it a nod of respect. It’s a metal with a mission: to occupy space and, in doing so, to tell us a little more about the world around us. And that, my friends, is a truly impressive feat for just 15.0 grams.
This is science at its most approachable, its most down-to-earth. It’s about the tangible, the observable, and the delightfully predictable. And for that, I'm eternally grateful to our friend, 15.0 grams of iron, and the water it so expertly displaced.
So let’s raise a (very small, precisely measured) glass of water to the power of density and the charm of displacement. It’s a win-win situation, really. The iron gets its place, and we get a cool fact. Science wins!
And who knows, maybe this knowledge will inspire you to conduct your own little displacement experiments. Just be careful not to flood your kitchen. Remember, 15 grams is a modest amount, but a large object can displace a lot of water indeed!