How Many Valence Electrons Does S-32 Have
Let's talk about atoms. Specifically, let's ponder a rather peculiar specimen: Sulfur-32. Now, you might be thinking, "Valence electrons? Sounds like homework." But stick with me, because this is going to be fun. We're on a little adventure, and the destination is a number. A very specific, yet somehow elusive, number.
So, how many of these so-called valence electrons does our friend Sulfur-32 actually have floating around? It's a question that might keep some folks up at night. Others might just shrug and go back to their Netflix binges. I'm firmly in the "slightly amused by the question" camp.
Imagine atoms as tiny little solar systems. At the center, you've got your nucleus, like the sun. Then, zipping around in their own little orbits, are the electrons. These are like planets. Some electrons are closer to the sun, some are further out. It's a whole cosmic dance happening at an impossibly small scale.
Must Read
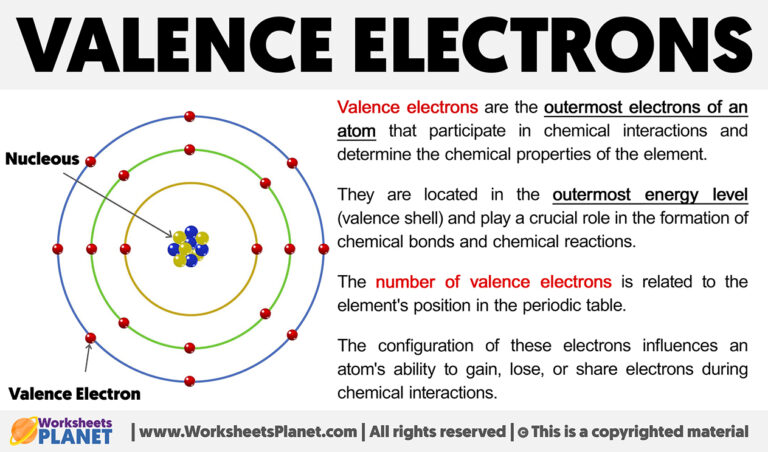
Now, the ones we're interested in for this whole "valence electron" thing are the outermost ones. Think of them as the planets on the very edge of the solar system. These are the players in the grand game of chemistry. They decide if an atom is going to be chatty and make friends, or be a bit of a loner.
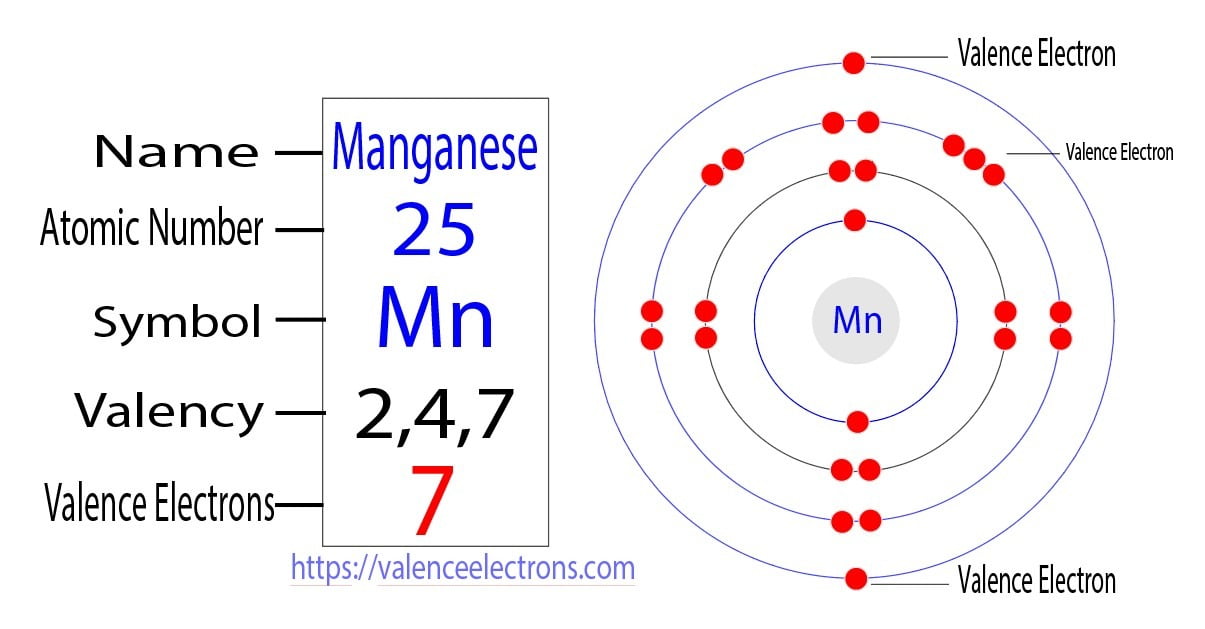
Our star of the show is Sulfur. And this isn't just any old sulfur. This is the common, garden-variety Sulfur-32. The "32" part? That's like its weight class. It tells us how many protons and neutrons are chilling in its nucleus. For our electron quest, though, the "Sulfur" part is the main event.
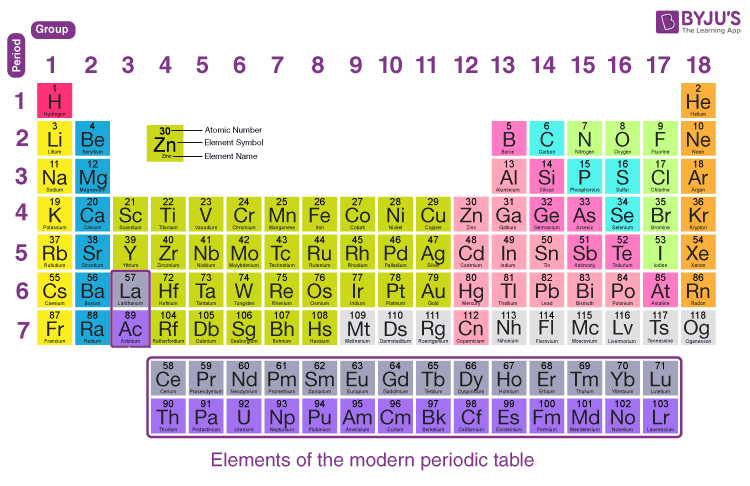
You see, the number of valence electrons an atom has is usually tied to its position on that fancy chart called the Periodic Table. It's like a cosmic cheat sheet for atoms. If you know where an atom hangs out on that table, you can get a pretty good idea of its electron personality.
So, where does Sulfur reside on this esteemed table? It's in the group often referred to as the chalcogens. Sounds fancy, right? Like something you'd find in an ancient potion. But really, it just means they like to form bonds, sort of like a group of friends who always do things together.
And this group, this family of chalcogens, they all share a certain number of those important outer electrons. It's like a family trait, passed down through generations of atoms. A little bit like how some families all have curly hair, or a knack for telling bad jokes.
So, if we know Sulfur is a chalcogen, and we know what the general rule is for chalcogens, we're getting closer to our answer. We're like detectives, piecing together clues. Except our clues are subatomic particles and a chart designed by very clever people.
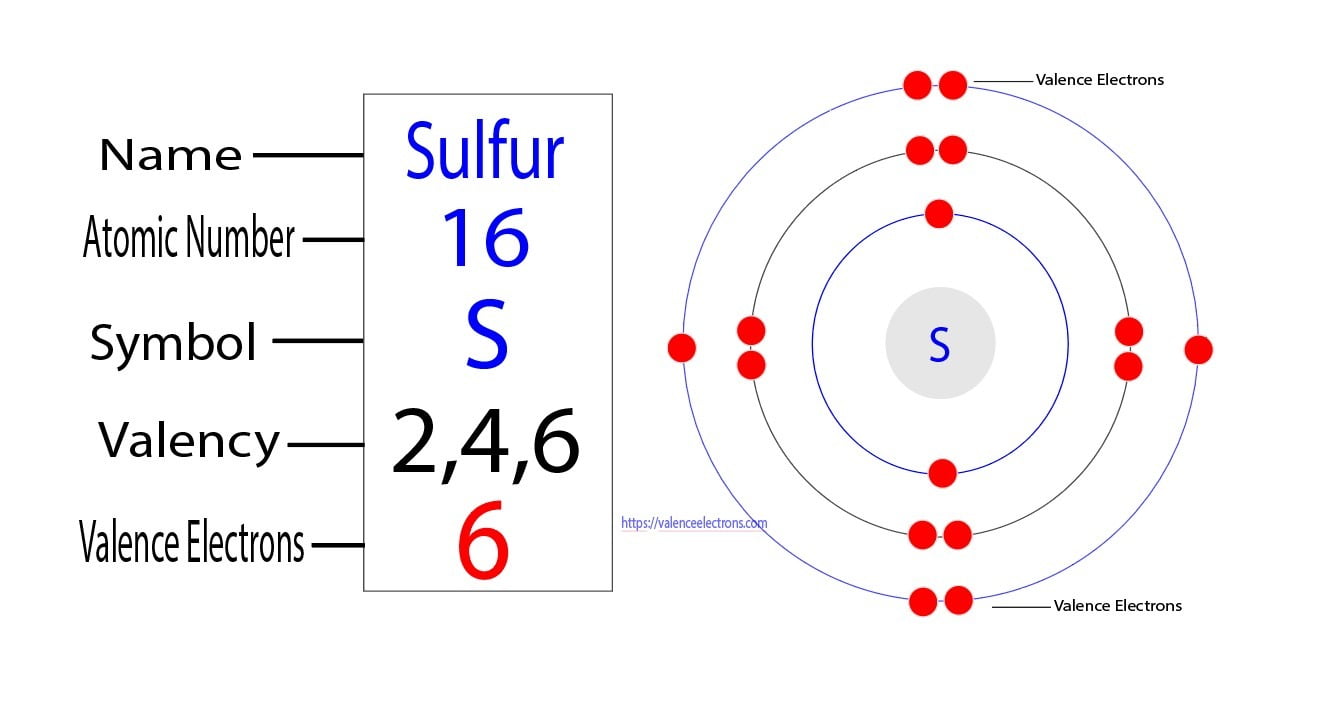
The Periodic Table is organized into rows called periods and columns called groups. The groups are where the real family resemblances start to show. And Sulfur, bless its atomic heart, is in Group 16. This is a significant detail in our thrilling investigation.
Now, for the elements in the main blocks of the Periodic Table, the group number often gives you a direct hint about the number of valence electrons. It’s a bit of a shortcut, a helpful little nudge from the universe of chemistry.
For elements in Group 16, the number of valence electrons is generally six. Yes, a neat and tidy number. Six little electron planets orbiting on the outer edge, ready for action. This is where the magic, or at least the chemistry, happens.
So, our particular specimen, Sulfur-32, being a form of Sulfur, will follow this rule. The "32" doesn't change its fundamental electron configuration. It's like having the same model of car, just with a slightly different engine size. The passenger capacity, the number of doors – those things stay the same.
Therefore, and this is where we might start to feel a sense of smug satisfaction, Sulfur-32 has six valence electrons. There you have it. The grand mystery is solved. It wasn't that complicated, was it? You might even feel a tiny bit of pride in your newfound atomic knowledge.
Now, I have an unpopular opinion. Do we really need to be so hung up on the exact number? I mean, sure, it's important for doing actual chemistry calculations. But for simply appreciating the sheer wonder of it all, does it matter if it's six or seven or five and a half? Probably not.
Think about it. Atoms are doing their thing, making bonds, forming molecules, creating everything around us. And they're doing it with these little electron buddies. It's a constant party of interactions. The exact number of guests at the party is less important than the fact that the party is happening and it's producing awesome results.
My theory is that the universe is a bit more forgiving than textbooks sometimes make it out to be. It's more about the general vibe, the overall electron eagerness. Is the atom ready to share? Is it looking to grab? That's the real story.
So, while we’ve diligently established that Sulfur-32 has six valence electrons – and I salute your dedication for sticking with me this far – let’s also acknowledge the larger picture. This is a snapshot of a much grander, much more dynamic reality.
These valence electrons are the atomic equivalent of social butterflies. They're the ones who get out there and mingle. They're the ones who make things happen. Without them, the world as we know it would be a very different, and frankly, a much less interesting place.
So next time you encounter a molecule, or a compound, or even just a plain old element like Sulfur, take a moment to appreciate its valence electrons. Give them a little mental nod. They're the real MVPs, the unsung heroes of the atomic universe.
And as for Sulfur-32, it’s got its six trusty valence electrons, ready to play. Whether it’s forming acids, or contributing to that distinct eggy smell, it’s all thanks to those little guys on the outside. So, yeah, six. But also, wow, atoms are cool.
It's funny how a simple number can lead us down such an interesting path. We started with a question, a bit of scientific jargon, and ended up contemplating the very fabric of existence. All thanks to Sulfur-32 and its six valence electrons.
I encourage you, from this day forward, to view valence electrons with a sense of wonder. They are the spark, the connection, the building blocks of everything. They are the reason why you can enjoy a perfectly fried egg, or why batteries work, or why your phone screen is made of glass.

And if anyone ever asks you about Sulfur-32's valence electrons, you can confidently say "six." But then, perhaps, with a knowing wink, you can add, "But it's really more about the amazing chemistry they enable, don't you think?" That's the real, entertaining answer.
The actual number is six. But the journey to get there is far more entertaining than the destination itself.
So, there you have it. The great valence electron count of Sulfur-32. It's six. A humble number, perhaps, but one that underpins so much of the universe. It’s a testament to the elegant simplicity and profound complexity that nature offers us every single day.
And if you’re anything like me, you’ve learned something new, had a little chuckle, and maybe even developed a newfound appreciation for the humble atom. The world of chemistry isn't so scary after all, is it? It's just a big, energetic party, and everyone's invited.
The key takeaway here, besides the number six, is that even seemingly dry scientific facts can be explored with a light heart and a curious mind. It's about making connections, finding the humor, and realizing that science is for everyone.
So, give yourself a pat on the back. You’ve just demystified the valence electrons of Sulfur-32. And who knows what other atomic mysteries await your playful investigation?