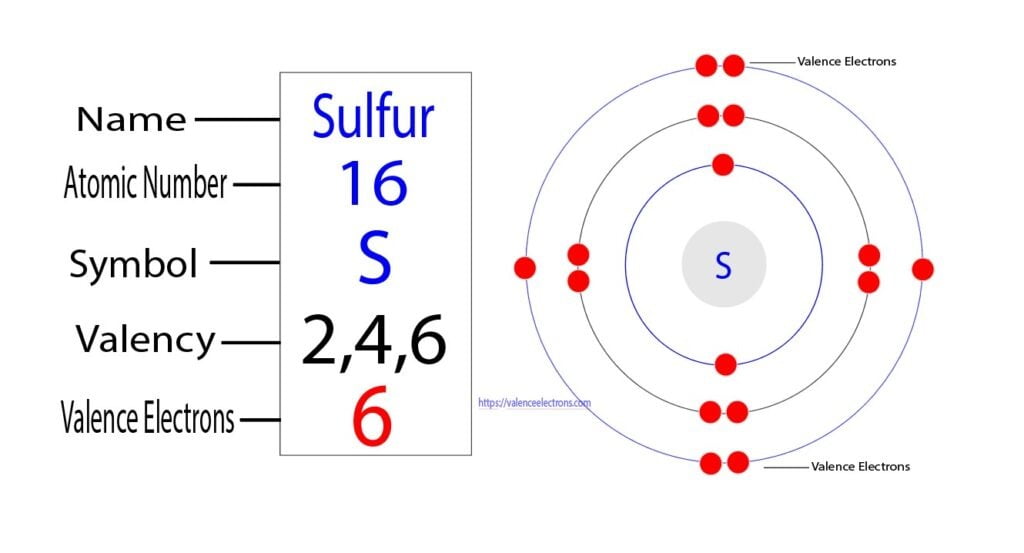
How Many Unpaired Electrons Does Sulfur Have

Ever found yourself staring at a bottle of something, maybe a cleaning product or even just a bag of fertilizer, and wondered about the tiny building blocks that make it all up? It’s a fun little rabbit hole to go down, and today, we’re going to peek into the world of sulfur and answer a seemingly simple question: How many unpaired electrons does it have? Now, this might sound like something straight out of a chemistry lab, but understanding this fundamental aspect of sulfur can unlock some really cool insights into how the world around us works, from the smell of a match to the very air we breathe.
So, why bother with unpaired electrons? Think of electrons as tiny, energetic dancers around an atom's nucleus. Most of the time, these dancers pair up, holding hands, so to speak. But sometimes, a dancer is left all by their lonesome. These are the unpaired electrons, and they’re like the energetic loners of the atomic world. They are incredibly reactive and love to find a partner. This dance of pairing and unpairing is the secret sauce behind how atoms bond together to form everything we see and interact with. For sulfur, knowing its unpaired electrons tells us a lot about its personality – its tendency to form certain kinds of bonds and participate in particular chemical reactions.
The benefits of understanding this are pretty far-reaching. In education, it's a cornerstone for grasping basic chemical principles. For anyone learning chemistry, figuring out unpaired electrons is a crucial step in understanding atomic structure, valency, and how molecules behave. In daily life, it’s the invisible force behind many everyday phenomena. For instance, sulfur's ability to form strong bonds, often facilitated by its unpaired electrons, is key to the rubber in your tires, the vulcanization process that makes it durable. It's also integral to the creation of sulfuric acid, a workhorse chemical used in everything from battery production to fertilizer manufacturing. Even the distinct smell of burning sulfur, associated with matches and volcanic activity, is a direct result of its reactive nature driven by those unpaired electrons.
Must Read
Curious to explore this further? It’s easier than you might think! The most straightforward way is to learn about electron configurations. This is like mapping out where all those electron dancers are supposed to be. For sulfur (which has 16 electrons in total), its electron configuration tells us how they arrange themselves in different energy levels and orbitals. You can find charts and diagrams online that illustrate this. Another simple approach is to look at the periodic table. Sulfur is in Group 16, and elements in the same group often share similar characteristics regarding their outer electrons, which is where those unpaired electrons reside. Many educational websites and even introductory chemistry textbooks have sections dedicated to this. You could even try a simple online search for "sulfur electron configuration" and be amazed by the visual representations that pop up. It's a fascinating glimpse into the hidden mechanics of the universe, all starting with a simple question about one element's dance partners!